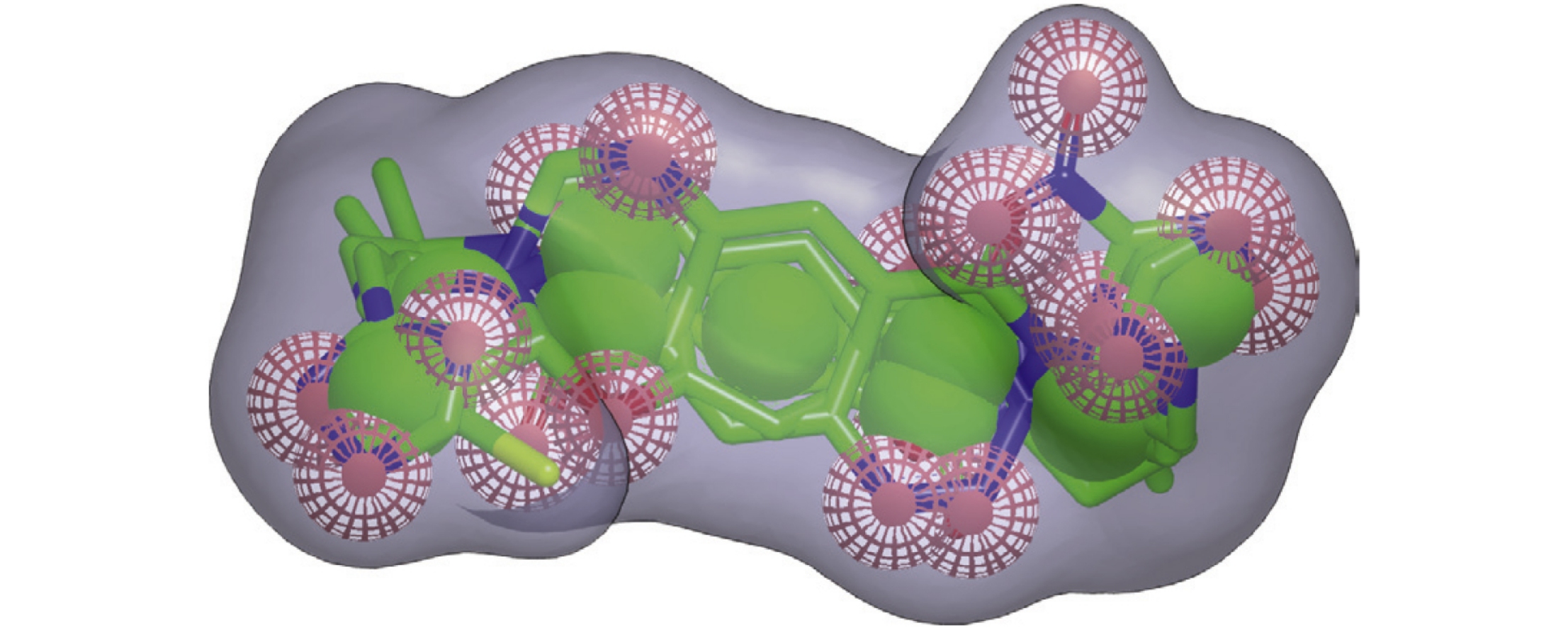
Abstract
Positive allosteric modulators of AMPA receptor are prospective for the development of safe and efficacious agents for the correction of disruption of the central nervous system functions. Joint application of molecular docking, MFTA and CoMFA QSAR modelling, and pharmacophore analysis affords a consistent picture of structural requirements for molecular design of novel promising bivalent modulators with high activity, including overall twisted-staple shape of a molecule, hydrophobic central core, and polar hydrogen bond acceptor groups in peripheral parts of a molecule.
References
1.

Traynelis S.F., Wollmuth L.P., McBain C.J., Menniti F.S., Vance K.M., Ogden K.K., Hansen K.B., Yuan H., Myers S.J., Dingledine R.
Pharmacological Reviews,
2010
2.

Grigoriev V.V., Proshin A.N., Kinzirsky A.S., Bachurin S.O.
Russian Chemical Reviews,
2009
3.

Destot-Wong K., Liang K., Gupta S.K., Favrais G., Schwendimann L., Pansiot J., Baud O., Spedding M., Lelièvre V., Mani S., Gressens P.
Neuropharmacology,
2009
4.

Bloss E.B., Hunter R.G., Waters E.M., Munoz C., Bernard K., McEwen B.S.
Experimental Neurology,
2008
5.

Lauterborn J.C., Palmer L.C., Jia Y., Pham D.T., Hou B., Wang W., Trieu B.H., Cox C.D., Kantorovich S., Gall C.M., Lynch G.
Journal of Neuroscience,
2016
6.

Partin K.M.
Current Opinion in Pharmacology,
2015
7.

Tozzi A., Sclip A., Tantucci M., de Iure A., Ghiglieri V., Costa C., Di Filippo M., Borsello T., Calabresi P.
Neurobiology of Aging,
2015
8.

Machado-Vieira R., Henter I.D., Zarate Jr. C.A.
Progress in Neurobiology,
2017
9.

Ward S.E., Pennicott L.E., Beswick P.
Future Medicinal Chemistry,
2015
10.

Lavrov M.I., Grigor’ev V.V., Bachurin S.O., Palyulin V.A., Zefirov N.S.
Doklady Biochemistry and Biophysics,
2015
11.
Zefirov N.S., Palyulin V.A.
Mendeleev Communications,
2010
12.

Karlov D.S., Lavrov M.I., Palyulin V.A., Zefirov N.S.
Russian Chemical Bulletin,
2016
13.

Mueller R., Li Y., Hampson A., Zhong S., Harris C., Marrs C., Rachwal S., Ulas J., Nielsson L., Rogers G.
Bioorganic and Medicinal Chemistry Letters,
2011
14.

Mueller R., Rachwal S., Tedder M.E., Li Y., Zhong S., Hampson A., Ulas J., Varney M., Nielsson L., Rogers G.
Bioorganic and Medicinal Chemistry Letters,
2011
15.

Mueller R., Rachwal S., Lee S., Zhong S., Li Y., Haroldsen P., Herbst T., Tanimura S., Varney M., Johnson S., Rogers G., Street L.J.
Bioorganic and Medicinal Chemistry Letters,
2011
16.

Mueller R., Rachwal S., Varney M.A., Johnson S.A., Alisala K., Zhong S., Li Y., Haroldsen P., Herbst T., Street L.J.
Bioorganic and Medicinal Chemistry Letters,
2011
17.

Fourches D., Muratov E., Tropsha A.
Journal of Chemical Information and Modeling,
2016
18.
Instant JChem 17.3.27, ChemAxon Ltd., 2017, http://www.chemaxon.com.
19.

Palyulin V.A., Radchenko E.V., Zefirov N.S.
Journal of Chemical Information and Computer Sciences,
2000
20.
10.1016/j.mencom.2017.11.029_bib0100
Radchenko
Royal Society of Chemistry,
2008
21.

Cramer R.D., Patterson D.E., Bunce J.D.
Journal of the American Chemical Society,
1988
22.

McGann M.
Journal of Chemical Information and Modeling,
2011
23.
OEDocking 3.2.0.2, OpenEye Scientific Software, 2015, http://www.eyesopen.com.
24.

Timm D.E., Benveniste M., Weeks A.M., Nisenbaum E.S., Partin K.M.
Molecular Pharmacology,
2011
25.

Hawkins P.C., Skillman A.G., Warren G.L., Ellingson B.A., Stahl M.T.
Journal of Chemical Information and Modeling,
2010
26.
OMEGA 2.5.1.4, OpenEye Scientific Software, 2013, http://www.eyesopen.com.
27.
SZYBKI 1.9.0.3, OpenEye Scientific Software, 2016, http://www.eyesopen.com.
28.
Sybyl-X 2.1, Certara L.P., 2013, http://www.certara.com.
29.

Hawkins P.C., Skillman A.G., Nicholls A.
Journal of Medicinal Chemistry,
2006
30.
vROCS 3.2.1.4, OpenEye Scientific Software, 2015, http://www.eyesopen.com.