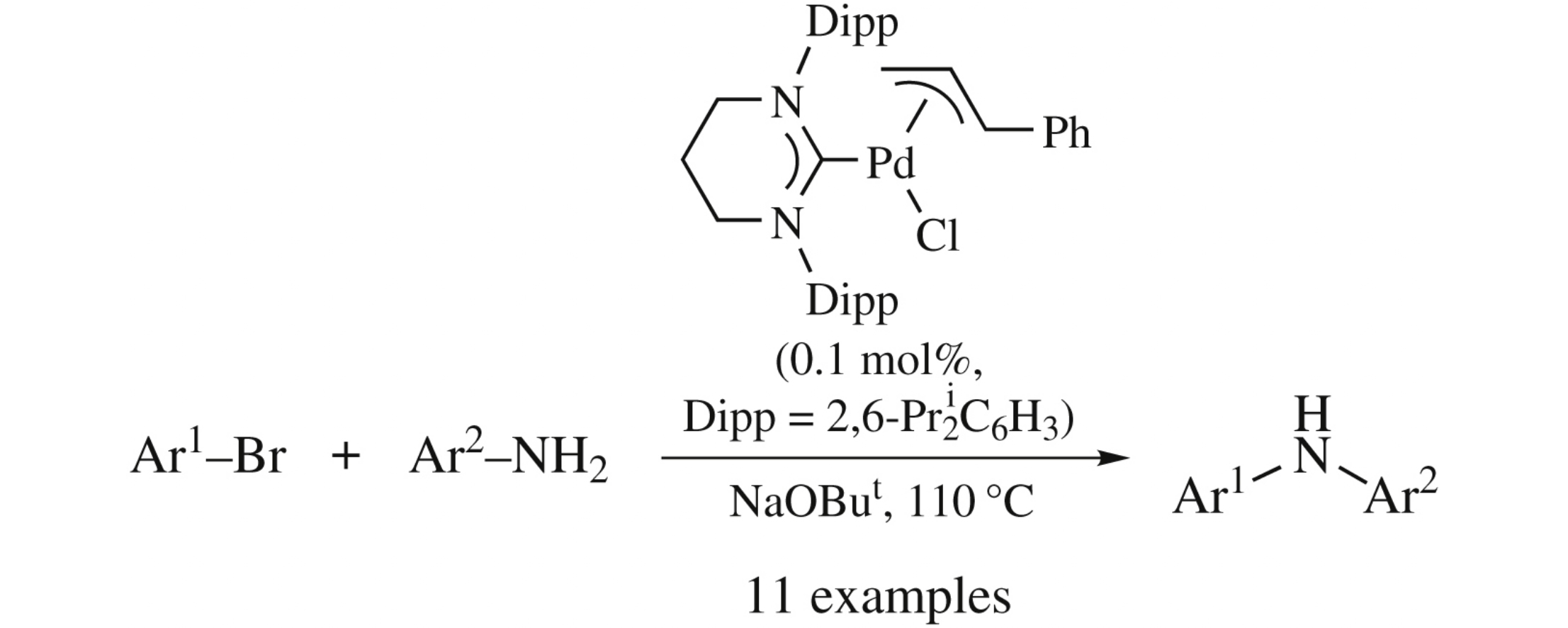
Abstract
A highly efficient ‘green’ solvent-free monoarylation of primary anilines with aryl bromides mediated by the expanded-ring N-heterocyclic carbene palladium complex (THP-Dipp)Pd(cinn)Cl [THP-Dipp is 1,3-bis(2,6-diisopropylphenyl)-3,4,5,6-tetrahydropyrimidin-2-ylidene; cinn is cinnamyl] can be performed at low catalyst loadings (0.1 mol%) to provide excellent yields and remarkable selectivities for various substrates.
References
1.

Tang C.W., VanSlyke S.A.
Applied Physics Letters,
1987
2.

Yu T., Liu L., Xie Z., Ma Y.
Science China Chemistry,
2015
3.

Bellmann E., Shaheen S.E., Thayumanavan S., Barlow S., Grubbs R.H., Marder S.R., Kippelen B., Peyghambarian N.
Chemistry of Materials,
1998
4.
Kaplunov M.G., Yakushchenko I.K., Krasnikova S.S., Echmaev S.B.
Mendeleev Communications,
2016
5.

Topchiy M.A., Dzhevakov P.B., Rubina M.S., Morozov O.S., Asachenko A.F., Nechaev M.S.
European Journal of Organic Chemistry,
2016
6.

Surry D.S., Buchwald S.L.
Journal of the American Chemical Society,
2007
7.

Yamamoto T., Nishiyama M., Koie Y.
Tetrahedron Letters,
1998
8.

Hirai Y., Uozumi Y.
Chemical Communications,
2010
9.

Sadighi J.P., Harris M.C., Buchwald S.L.
Tetrahedron Letters,
1998
10.
![Synthesis of diarylamines in the benzo[b]thiophene series bearing electron donating or withdrawing groups by Buchwald–Hartwig C–N coupling](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Ferreira I.C., Queiroz M.R., Kirsch G.
Tetrahedron,
2003
11.

Wüllner G., Jänsch H., Schubert F., Boche G.
Chemical Communications,
1998
12.

Ananikov V.P., Khemchyan L.L., Ivanova Y.V., Bukhtiyarov V.I., Sorokin A.M., Prosvirin I.P., Vatsadze S.Z., Medved'ko A.V., Nuriev V.N., Dilman A.D., Levin V.V., Koptyug I.V., Kovtunov K.V., Zhivonitko V.V., Likholobov V.A., et. al.
Russian Chemical Reviews,
2014
13.

Melvin P.R., Nova A., Balcells D., Dai W., Hazari N., Hruszkewycz D.P., Shah H.P., Tudge M.T.
ACS Catalysis,
2015
14.

Kolychev E.L., Asachenko A.F., Dzhevakov P.B., Bush A.A., Shuntikov V.V., Khrustalev V.N., Nechaev M.S.
Dalton Transactions,
2013
15.

16.

Rataboul F., Zapf A., Jackstell R., Harkal S., Riermeier T., Monsees A., Dingerdissen U., Beller M.
Chemistry - A European Journal,
2004
17.

Shen H., Zhang Z., Li J.
Journal of Chemical Research,
2010
18.

Ackermann L., Spatz J.H., Gschrei C.J., Born R., Althammer A.
Angewandte Chemie - International Edition,
2006
19.

Budén M.E., Rossi R.A.
Tetrahedron Letters,
2007
20.

21.

Fors B.P., Davis N.R., Buchwald S.L.
Journal of the American Chemical Society,
2009
22.

Xie Y., Liu S., Liu Y., Wen Y., Deng G.
Organic Letters,
2012
23.

24.

Chen J., Song G., Pan C., Li X.
Organic Letters,
2010