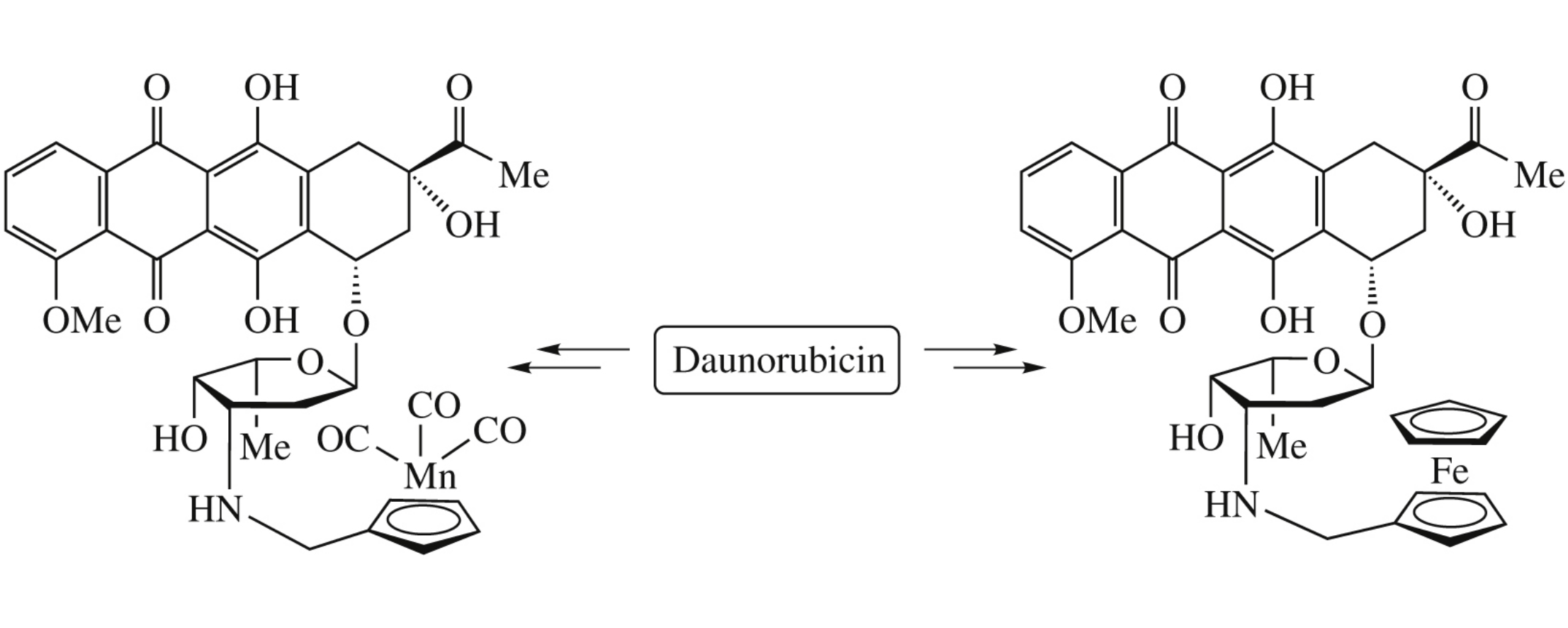
Abstract
Hybrid molecules wherein five-membered rings of ferrocene or cymanthrene are covalently bonded with the amino group of daunorubicin through a methylene linker, have been synthesized by reductive amination of the corresponding aldehydes [M]C5H4CHO {[M] = CpFe or (CO)3Mn} using daunorubicin as an amine component.
References
1.

van Staveren D.R., Metzler-Nolte N.
Chemical Reviews,
2004
2.

Hartinger C.G., Dyson P.J.
Chemical Society Reviews,
2009
3.

Patra M., Gasser G., Metzler-Nolte N.
Dalton Transactions,
2012
4.

Patra M., Gasser G.
ChemBioChem,
2012
5.
10.1016/j.mencom.2017.11.023_bib0015
Bioorganometallics,
2006
6.

Snegur L.V., Nekrasov Y.S., Sergeeva N.S., Zhilina Z.V., Gumenyuk V.V., Starikova Z.A., Simenel A.A., Morozova N.B., Sviridova I.K., Babin V.N.
Applied Organometallic Chemistry,
2008
7.
Vatsadze S.Z., Shulga D.A., Loginova Y.D., Vatsadze I.A., Wang L., Yu H., Kudryavtsev K.V.
Mendeleev Communications,
2016
8.

Meggers E., Atilla-Gokcumen G., Bregman H., Maksimoska J., Mulcahy S., Pagano N., Williams D.
Synlett,
2007
9.

Williams D.S., Atilla G.E., Bregman H., Arzoumanian A., Klein P.S., Meggers E.
Angewandte Chemie - International Edition,
2005
10.

Policar C., Waern J.B., Plamont M., Clède S., Mayet C., Prazeres R., Ortega J., Vessières A., Dazzi A.
Angewandte Chemie - International Edition,
2010
11.

N′Dongo H.W., Neundorf I., Merz K., Schatzschneider U.
Journal of Inorganic Biochemistry,
2008
12.

Neundorf I., Hoyer J., Splith K., Rennert R., Peindy N’Dongo H.W., Schatzschneider U.
Chemical Communications,
2008
13.
García J.J., Flores-Alamo M., Vega M.A., Klimova E.I.
Mendeleev Communications,
2014
14.

Schweitzer B.A., Koch T.H.
Journal of the American Chemical Society,
1993
15.

Zhang S., Dong J., Wang Y.
Synthetic Communications,
2003
16.

Piorecka K., Radzikowska E., Kurjata J., Rozga-Wijas K., Stanczyk W.A., Wielgus E.
New Journal of Chemistry,
2016
17.
10.1002/1520-6017(200007)89:7<910::AID-JPS8>3.0.CO;2-#
18.

Zhao P., Lu J., He J., Chen W., Chen P., Chen D., Bin Q.
Nucleosides, Nucleotides and Nucleic Acids,
2014
19.

Zhao P., Jin S., Lu J., Lv J., Wu G., Chen P., Tan C., Chen D.
Spectrochimica Acta - Part A: Molecular and Biomolecular Spectroscopy,
2015
20.

N. Jungheim L., A. Shepherd T., K. Kling J.
Heterocycles,
1993
21.

Israel M., Potti G.
Journal of Medicinal Chemistry,
1982
22.

Zhang G., Fang L., Zhu L., Sun D., Wang P.G.
Bioorganic and Medicinal Chemistry,
2006
23.
![An improved synthetic approach to 7-[3-amino-4-O-(α-l-mycarosyl)-2,3,6-trideoxy-α-l-lyxo-hexopyranosyl]daunorubicinone and its interaction with human serum albumin](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Dang X., Liu Q., Cui F., Qin L., Zhang G., Yao X., Du J.
Carbohydrate Research,
2011
24.

Matsuda F., Matsumoto T., Ohsaki M., Terashima S.
Bulletin of the Chemical Society of Japan,
1991
25.

Csorvási A., Kövér K.E., Menyhárt M.M., Sztaricskai F., Dobrynin Y.V., Nikolaeva T.G.
Archiv der Pharmazie,
1998
26.
J.-B., Ducep, D., Farge, G., Ponsinet, D., Reisdorf,J.-B. Ducep, D. Farge, G. Ponsinet and D. Reisdorf, US Patent 4185111, 1980.
27.

Bakina E., Wu Z., Rosenblum M., Farquhar D.
Journal of Medicinal Chemistry,
1997
28.

Ghirmai S., Mume E., Tolmachev V., Sjöberg S.
Carbohydrate Research,
2005
29.

Masquelier M.
European Journal of Medicinal Chemistry,
2000
30.

Sato M., Kono H., Shiga M., Motoyama I., Hata K.
Bulletin of the Chemical Society of Japan,
1968
31.

Tong G.L., Wu H.Y., Smith T.H., Henry D.W.
Journal of Medicinal Chemistry,
1979