Abstract
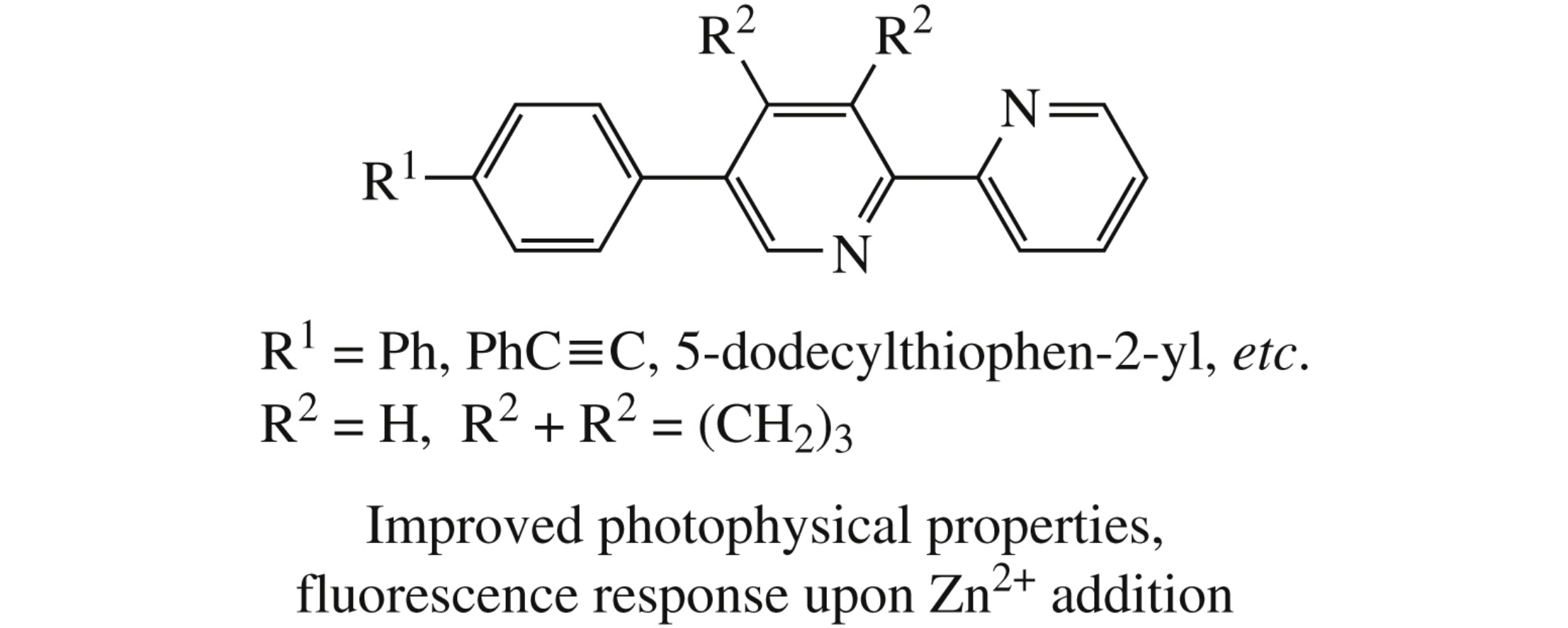
A straightforward and convenient synthesis of new fluorescent 5-aryl-2,2’-bipyridines with the extended conjugation system involves the ‘1,2,4-triazine’ methodology followed by Pd- catalyzed cross-coupling or Wittig reactions using 5-(4-bromo- phenyl)- or 5-(4-bromomethylphenyl)-2,2’-bipyridines as the starting materials. For these compounds, a pronounced bathochromic shift was observed in both absorption and emission spectra compared to previously reported 5-phenyl- 2,2’-bipyridine. For the one of the products, adsorption and fluorescent titrations were fulfilled, fluorescent response on the addition of metal cations was examined.
References
1.
10.1016/j.mencom.2017.11.021_sbref0005a
von Zelewsky
Stereochemistry of Coordination Compounds,
1996
2.
10.1016/j.mencom.2017.11.021_sbref0005b
1996
3.

Kaes C., Katz A., Hosseini M.W.
Chemical Reviews,
2000
4.

Hancock R.D.
Chemical Society Reviews,
2013
5.
Sakhapov I.F., Gafurov Z.N., Babaev V.M., Rizvanov I.K., Dobrynin A.B., Krivolapov D.B., Khayarov K.R., Sinyashin O.G., Yakhvarov D.G.
Mendeleev Communications,
2016
6.

Photophysical Properties of Ru(II) Bipyridyl Complexes Containing Hemilabile Phosphine−Ether Ligands
Angell S.E., Zhang Y., Rogers C.W., Wolf M.O., Jones W.E.
Inorganic Chemistry,
2005
7.

Bünzli J.G., Charbonnière L.J., Ziessel R.F.
Journal of the Chemical Society Dalton Transactions,
2000
8.

Chen J., Gao G., Di B., Luo Y., Zeng X., Qiu L., He L., Liu S., Wen H.
Journal of Coordination Chemistry,
2016
9.
![The effects of ligand substitution and deuteriation on the spectroscopic and photophysical properties of [Ru(LL)(CN)4]2−complexes](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Kovács M., Ronayne K.L., Browne W.R., Henry W., Vos J.G., McGarvey J.J., Horváth A.
Photochemical and Photobiological Sciences,
2007
10.

Loren J.C., Siegel J.S.
Angewandte Chemie - International Edition,
2001
11.

Fei X., Shuang-Qing W., Li-Ming H., Sha-Yu L., Quan G., Guo-Qi Z., Yi L., Guo-Qiang Y.
Chinese Journal of Chemistry,
2005
12.

Kozhevnikov V.N., Shabunina O.V., Kopchuk D.S., Ustinova M.M., König B., Kozhevnikov D.N.
Tetrahedron,
2008
13.

Joshi H.S., Jamshidi R., Tor Y.
Angewandte Chemie - International Edition,
1999
14.

Younes A.H., Zhang L., Clark R.J., Zhu L.
Journal of Organic Chemistry,
2009
15.

Ajayaghosh A., Carol P., Sreejith S.
Journal of the American Chemical Society,
2005
16.

Prokhorov A.M., Kozhevnikov V.N., Kopchuk D.S., Bernard H., Le Bris N., Tripier R., Handel H., Koenig B., Kozhevnikov D.N.
Tetrahedron,
2011
17.

Kopchuk D.S., Prokhorov A.M., Slepukhin P.A., Kozhevnikov D.N.
Tetrahedron Letters,
2012
18.
Kozhevnikov V.N., Shabunina O.V., Sharifullina A.R., Rusinov V.L., Chupakhin O.N., König B.
Mendeleev Communications,
2005
19.

Dai F., Wu W., Wang Q., Tian H., Wong W.
Dalton Transactions,
2011
20.

Nitschke J.R., Zürcher S., Tilley T.D.
Journal of the American Chemical Society,
2000
21.

Sakamoto J., Schlüter A.D.
European Journal of Organic Chemistry,
2007
22.

Kozhevnikov V.N., Kozhevnikov D.N., Nikitina T.V., Rusinov V.L., Chupakhin O.N., Zabel M., König B.
Journal of Organic Chemistry,
2003
23.

Kozhevnikov D.N., Shabunina O.V., Kopchuk D.S., Slepukhin P.A., Kozhevnikov V.N.
Tetrahedron Letters,
2006
24.

Pabst G.R., Pfüller O.C., Sauer J.
Tetrahedron,
1999
25.

Rykowski A., Branowska D., Kielak J.
Tetrahedron Letters,
2000
26.

Prokhorov A.M., Kozhevnikov D.N.
Chemistry of Heterocyclic Compounds,
2012
27.
Kovalev I.S., Kopchuk D.S., Khasanov A.F., Zyryanov G.V., Rusinov V.L., Chupakhin O.N.
Mendeleev Communications,
2014
28.
Khasanov A.F., Kopchuk D.S., Kovalev I.S., Taniya O.S., Zyryanov G.V., Rusinov V.L., Chupakhin O.N.
Mendeleev Communications,
2015
29.

Gonsalves A.M., Pinho e Melo T.M., Gilchrist T.L.
Tetrahedron,
1992
30.

Kopchuk D.S., Nikonov I.L., Zyryanov G.V., Kovalev I.S., Rusinov V.L., Chupakhin O.N.
Chemistry of Heterocyclic Compounds,
2014
31.
![(Benzo[h])Quinolinyl-Substituted Monoazatriphenylenes: Synthesis and Photophysical Properties](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Kopchuk D.S., Khasanov A.F., Kovalev I.S., Kim G.A., Nikonov I.L., Zyryanov G.V., Rusinov V.L., Chupakhin O.N.
Chemistry of Heterocyclic Compounds,
2014
32.

Ławecka J., Olender E., Piszcz P., Rykowski A.
Tetrahedron Letters,
2008
33.

Krinochkin A.P., Kopchuk D.S., Kozhevnikov D.N.
Polyhedron,
2015
34.

Kopchuk D.S., Krinochkin A.P., Kozhevnikov D.N., Slepukhin P.A.
Polyhedron,
2016
35.

Pawar S., Akula M., Labala S., Venuganti V.V., Bhattacharya A., Nag A.
ChemPlusChem,
2016
36.

Xu Z., Yoon J., Spring D.R.
Chemical Society Reviews,
2010
37.

Núñez C., Santos S.M., Oliveira E., Santos H.M., Capelo J.L., Lodeiro C.
ChemistryOpen,
2014
38.

Dawson W.R., Windsor M.W.
The Journal of Physical Chemistry,
1968