Abstract
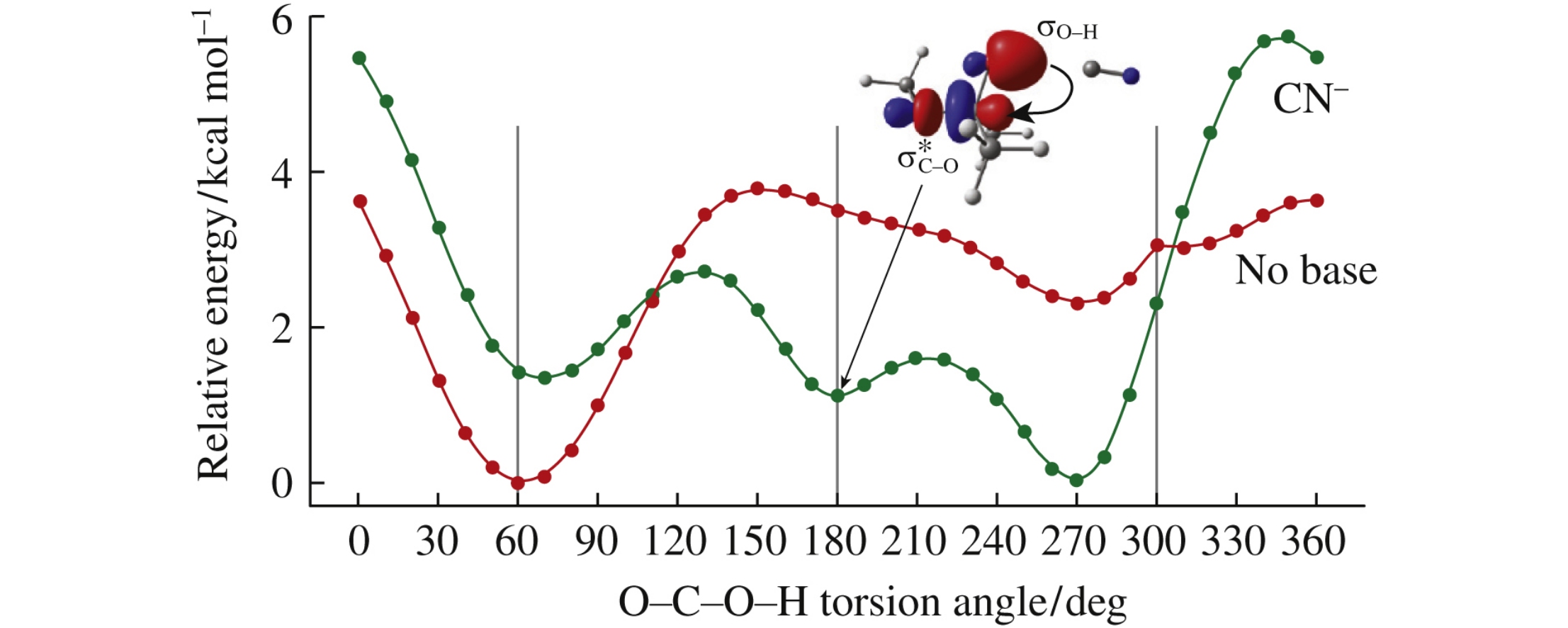
Hemiketals are important targets for crystal prediction and molecular modeling. The supramolecular stereoelectronic effect (SSE) recently found in carboxylic acid associates occurs in hemiketals: the presence and nature of an H-bond acceptor affect the conformational preference of hemiketals. To provide a structural basis for the multitude of biological roles played by hemiketal-containing structures, it is important to accurately model their spatial and dynamic properties, so the SSE in hemiketals should be explicitly implemented in future force fields.
References
1.

Price S.L.
Chemical Society Reviews,
2014
2.

3.

Yilmazer N., Korth M.
International Journal of Molecular Sciences,
2016
4.

Pyrkov T.V., Ozerov I.V., Balitskaya E.D., Efremov R.G.
Russian Journal of Bioorganic Chemistry,
2010
5.

Arcon J.P., Defelipe L.A., Modenutti C.P., López E.D., Alvarez-Garcia D., Barril X., Turjanski A.G., Martí M.A.
Journal of Chemical Information and Modeling,
2017
6.

Perilla J.R., Goh B.C., Cassidy C.K., Liu B., Bernardi R.C., Rudack T., Yu H., Wu Z., Schulten K.
Current Opinion in Structural Biology,
2015
7.

Vanommeslaeghe K., Guvench O., MacKerell A.D.
Current Pharmaceutical Design,
2014
8.
10.1016/j.mencom.2017.11.019_bib0040
Chiu
The 19th Annual Inter- national IEEE Symposium on Field-Programmable Custom Computing Machines,
2011
9.

Medvedev M.G., Bushmarinov I.S., Lyssenko K.A.
Chemical Communications,
2016
10.
Ananikov V.P., Galkin K.I., Egorov M.P., Sakharov A.M., Zlotin S.G., Redina E.A., Isaeva V.I., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2016
11.

Reed A.E., Curtiss L.A., Weinhold F.
Chemical Reviews,
1988
12.
10.1016/j.mencom.2017.11.019_bib0060
Bader
Atoms in Molecules: A Quantum Theory,
1994
13.

Allen F.H.
Acta Crystallographica Section B Structural Science,
2002
14.

15.
10.1016/j.mencom.2017.11.019_bib0075
Frisch
Gaussian 09, Revision D.01,
2009
16.

Adamo C., Barone V.
Journal of Chemical Physics,
1999
17.

Grimme S., Antony J., Ehrlich S., Krieg H.
Journal of Chemical Physics,
2010
18.

Dunning T.H.
Journal of Chemical Physics,
1989
19.

Peverati R., Truhlar D.G.
Philosophical Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences,
2014
20.

Mardirossian N., Head-Gordon M.
Molecular Physics,
2017
21.

Medvedev M.G., Bushmarinov I.S., Sun J., Perdew J.P., Lyssenko K.A.
Science,
2017
22.

Lu T., Chen F.
Journal of Computational Chemistry,
2011
23.
10.1016/j.mencom.2017.11.019_bib0115
Keith
AIMA II,Version 16.01.09, TK Gristmill Software,
2015
24.

Solís D., Bovin N.V., Davis A.P., Jiménez-Barbero J., Romero A., Roy R., Smetana K., Gabius H.
Biochimica et Biophysica Acta - General Subjects,
2015
25.
10.1016/j.mencom.2017.11.019_bib0125
Alabugin
Stereoelectronic Effects: A Bridge between Structure and Reactivity,
2016
26.

Kasaei G.A., Nori-Shargh D., Yahyaei H., Mousavi S.N., Pourdavoodi E.
Molecular Simulation,
2012
27.
10.1016/j.mencom.2017.11.019_bib0135
Gavezzotti
Computational Approaches in Supra- molecular Chemistry,
1994
28.

Alabugin I.V., Zeidan T.A.
Journal of the American Chemical Society,
2002
29.

Espinosa E., Molins E.
Journal of Chemical Physics,
2000
30.

Ananyev I.V., Karnoukhova V.A., Dmitrienko A.O., Lyssenko K.A.
Journal of Physical Chemistry A,
2017
31.

Ananyev I.V., Bushmarinov I.S., Ushakov I.E., Aitkulova A.I., Lyssenko K.A.
RSC Advances,
2015
32.

Langan P., Sangha A., Wymore T., Parks J., Yang Z., Hanson B.L., Fisher Z., Mason S., Blakeley M., Forsyth V.T., Glusker J., Carrell H., Smith J., Keen D., Graham D., et. al.
Structure,
2014
33.

Stroganov O.V., Novikov F.N., Zeifman A.A., Stroylov V.S., Chilov G.G.
Proteins: Structure, Function and Genetics,
2011
34.

Stroganov O.V., Novikov F.N., Stroylov V.S., Kulkov V., Chilov G.G.
Journal of Chemical Information and Modeling,
2008
35.

Krohn K., Beckmann K., Flörke U., Aust H., Draeger S., Schulz B., Busemann S., Bringmann G.
Tetrahedron,
1997
36.

Kresse G., Furthmüller J.
Computational Materials Science,
1996
37.

Perdew J.P., Burke K., Ernzerhof M.
Physical Review Letters,
1996
38.

Kresse G., Joubert D.
Physical Review B,
1999