Abstract
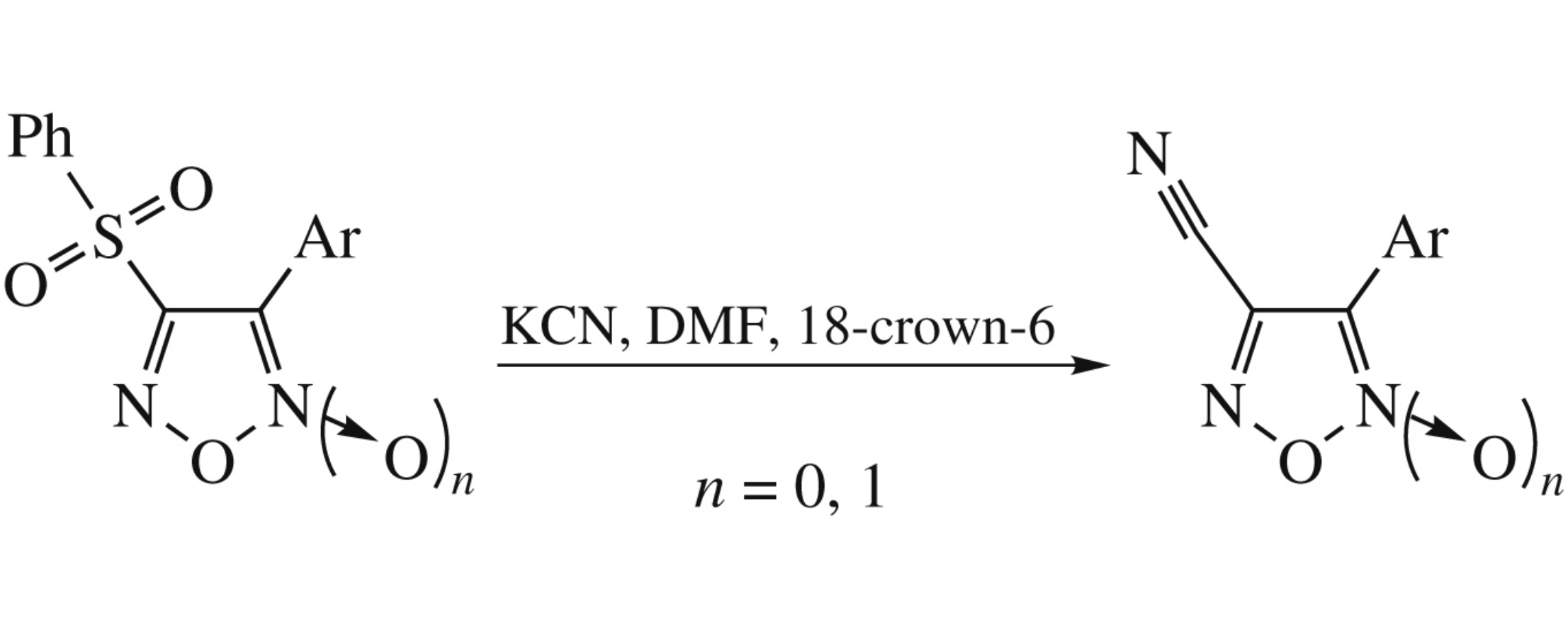
Potassium cyanide reacts with 3-aryl-4-phenylsulfonylfuroxan/ furazan under mild conditions in DMF at room temperature in the presence of 18-crown-6 to afford 3-aryl-4-cyanofuroxan/ furazan in 45–50% yield.
References
1.

Miller M.R., Okubo K., Roseberry M.J., Webb D.J., Megson I.L.
Journal of Cardiovascular Pharmacology,
2004
2.

Miller M.R., Megson I.L.
British Journal of Pharmacology,
2007
3.

Serafim R.A., Primi M.C., Trossini G.H., Ferreira E.I.
Current Medicinal Chemistry,
2012
4.

Granik V.G., Grigor"ev N.B.
Russian Chemical Bulletin,
2002
5.
10.1016/j.mencom.2017.11.008_bib0025
Gasco
Nitric Oxide Donors,
2005
6.

Feelisch M., Schönafingeri K., Noack H.
Biochemical Pharmacology,
1992
7.

Parakhin V.V., Luk’yanov O.A.
Russian Chemical Bulletin,
2016
8.
Starosotnikov A.M., Bastrakov M.A., Pavlov A.A., Fedyanin I.V., Dalinger I.L., Shevelev S.A.
Mendeleev Communications,
2016
9.

Medana C., Ermondi G., Fruttero R., Di Stilo A., Ferretti C., Gasco A.
Journal of Medicinal Chemistry,
1994
10.

Ghigo D., Calvino R., Heller R., Calvino R., Alessio P., Fruttero R., Gasco A., Bosia A., Pescarmona G.
Biochemical Pharmacology,
1992
11.

Calvino R., Fruttero R., Ghigo D., Bosia A., Pescarmona G.P., Gasco A.
Journal of Medicinal Chemistry,
1992
12.

Ferioli R., Folco G.C., Ferretti C., Gasco A.M., Medana C., Fruttero R., Civelli M., Gasco A.
British Journal of Pharmacology,
1995
13.
Ustyuzhanina N.E., Fershtat L.L., Gening M.L., Nifantiev N.E., Makhova N.N.
Mendeleev Communications,
2016
14.

Zhao J., Zhou M., Zuo J., Xu X., Zhang X., Yuan W.
Tetrahedron,
2015
15.

Fruttero R., Ferrarotti B., Serafino A., Gasco A.
European Journal of Organic Chemistry,
1990
16.
![An efficient access to (1H-tetrazol-5-yl)furoxan ammonium salts via a two-step dehydration/[3+2]-cycloaddition strategy](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Fershtat L.L., Epishina M.A., Kulikov A.S., Ovchinnikov I.V., Ananyev I.V., Makhova N.N.
Tetrahedron,
2015
17.

Gasco A.M., Fruttero R., Sorba G., Gasco A.
European Journal of Organic Chemistry,
1991
18.

Borodkin G.I., Shubin V.G.
Russian Chemical Reviews,
2017
19.
Fershtat L.L., Epishina M.A., Kulikov A.S., Makhova N.N.
Mendeleev Communications,
2015
20.

Del Grosso E., Boschi D., Lazzarato L., Cena C., Di Stilo A., Fruttero R., Moro S., Gasco A.
Chemistry and Biodiversity,
2005
21.

Ando A., Matsubara R., Takazawa S., Shimada T., Hayashi M.
Asian Journal of Organic Chemistry,
2016
22.
Fershtat L.L., Ashirbaev S.S., Kulikov A.S., Kachala V.V., Makhova N.N.
Mendeleev Communications,
2015
23.
10.1016/j.mencom.2017.11.008_bib0115
Ponzio
Gazz. Chim. Ital.,
1931