Abstract
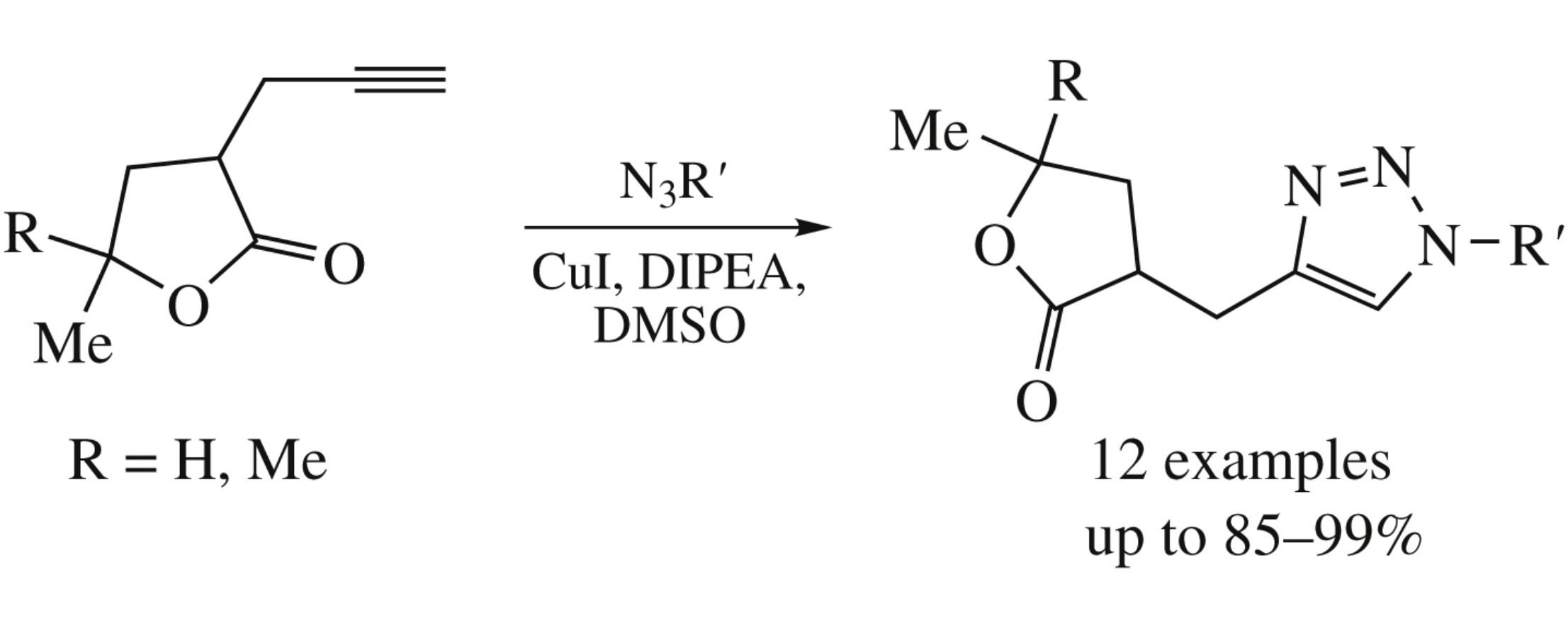
Copper catalyzed [2 + 3] cycloaddition of 3-propargyl- γ-butyrolactones with azides affords the corresponding 1,2,3-triazoles connected with lactone moiety in high yields. In the presence of air and copper(i) halides, the starting propargylated lactones are prone to dimerize to form bis-lactone diynes.
References
1.

Ito Y., Kamo S., Sadhu S.K., Ohtsuki T., Ishibashi M., Kano Y.
Chemical and Pharmaceutical Bulletin,
2009
2.

Trisuwan K., Rukachaisirikul V., Sukpondma Y., Phongpaichit S., Preedanon S., Sakayaroj J.
Chemical and Pharmaceutical Bulletin,
2009
3.

Shin H.J., Lee S.Y., Kim J.S., Lee S., Choi R.J., Chung H.S., Kim Y.S., Kang S.S.
Chemical and Pharmaceutical Bulletin,
2012
4.

Phan N.H., Thuan N.T., Ngoc N.T., Thao N.P., Kim S., Koh Y.S., Thanh N.V., Cuong N.X., Nam N.H., Kiem P.V., Kim Y.H., Minh C.V.
Chemical and Pharmaceutical Bulletin,
2015
5.
![Cooperative N-heterocyclic carbene (NHC)–Lewis acid-mediated regioselective umpolung formal [3 + 2] annulations of alkynyl aldehydes with isatins](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Zhang Y., Lu Y., Tang W., Lu T., Du D.
Organic and Biomolecular Chemistry,
2014
6.

Cailleau T., Cooke J.W., Davies S.G., Ling K.B., Naylor A., Nicholson R.L., Price P.D., Roberts P.M., Russell A.J., Smith A.D., Thomson J.E.
Organic and Biomolecular Chemistry,
2007
7.

Brimble M.A., Bryant C.J.
Organic and Biomolecular Chemistry,
2007
8.

Habel A., Boland W.
Organic and Biomolecular Chemistry,
2008
9.
Valiullina Z.R., Akhmet’yanova V.A., Vostrikov N.S., Miftakhov M.S.
Mendeleev Communications,
2016
10.
Sharipov B.T., Pershin A.A., Valeev F.A.
Mendeleev Communications,
2017
11.

Jian Y., Wu Y.
Organic and Biomolecular Chemistry,
2010
12.

Kochikyan T.V., Arutyunyan E.V., Arutyunyan V.S., Avetisyan A.A.
Russian Journal of Organic Chemistry,
2008
13.

Kochikyan T.V., Arutyunyan E.V., Samvelyan M.A., Arutyunyan V.S., Avetisyan A.A., Paronikyan R.V., Stepanyan G.M.
Pharmaceutical Chemistry Journal,
2009
14.

Kochikyan T.V., Samvelyan M.A., Arutyunyan V.S.
Russian Journal of Organic Chemistry,
2011
15.
10.1016/j.mencom.2017.11.007_bib0065
Arutyunyan
Arm. Khim. Zh.,
1985
16.

Rostovtsev V.V., Green L.G., Fokin V.V., Sharpless K.B.
Angewandte Chemie - International Edition,
2002
17.
![Peptidotriazoles on Solid Phase: [1,2,3]-Triazoles by Regiospecific Copper(I)-Catalyzed 1,3-Dipolar Cycloadditions of Terminal Alkynes to Azides](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Tornøe C.W., Christensen C., Meldal M.
Journal of Organic Chemistry,
2002
18.

Sokolova N.V., Nenajdenko V.G.
RSC Advances,
2013
19.
Ghochikyan T.V., Muzalevskiy V.M., Samvelyan M.A., Galstyan A.S., Nenajdenko V.G.
Mendeleev Communications,
2016
20.

Nenajdenko V.G., Gulevich A.V., Sokolova N.V., Mironov A.V., Balenkova E.S.
European Journal of Organic Chemistry,
2010
21.

Sokolova N.V., Latyshev G.V., Lukashev N.V., Nenajdenko V.G.
Organic and Biomolecular Chemistry,
2011
22.

Jian Y., Wu Y.
Organic and Biomolecular Chemistry,
2010