Abstract
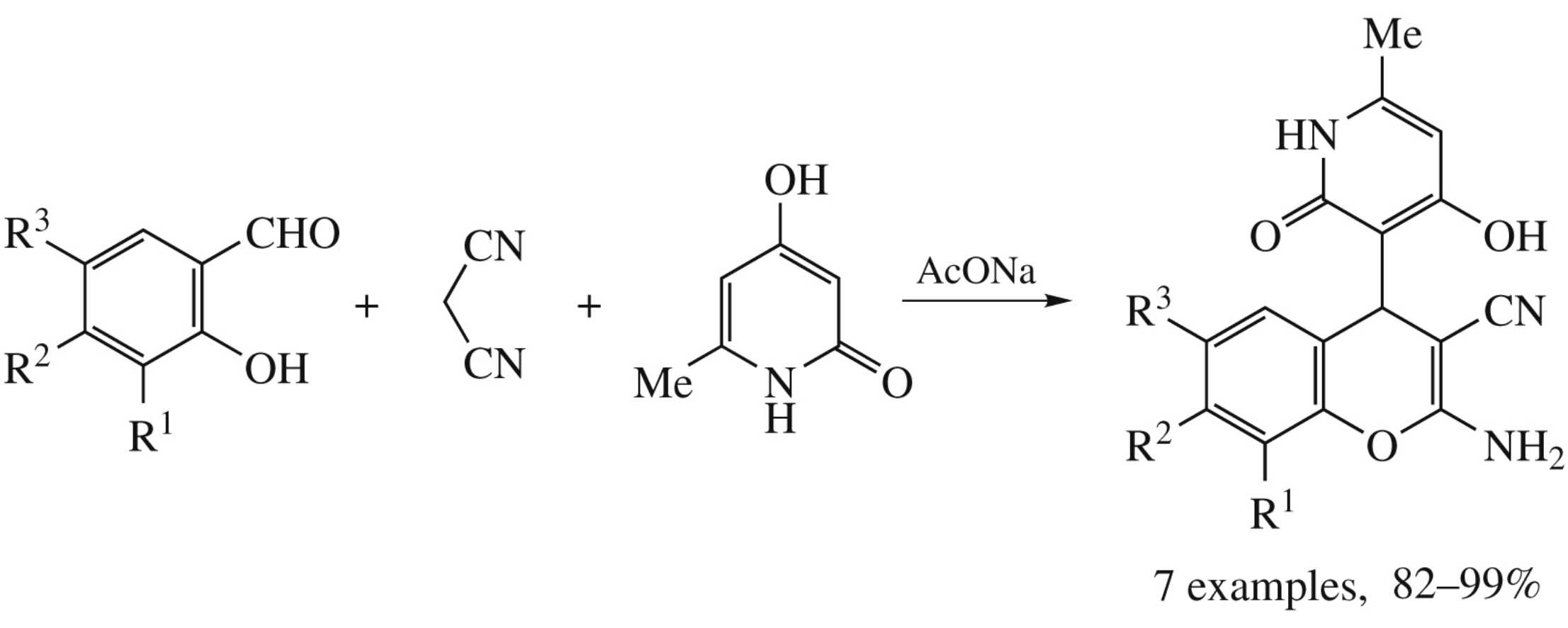
Sodium acetate catalyzed assembling of salicylaldehydes, malononitrile and 4-hydroxy-6-methylpyridin-2(1H)-one in small amount of ethanol gives rapidly (5 min) substituted 4-pyridinyl-2-amino-4H-chromenes in 82–99% yields. This novel domino reaction opens the fast, facile, and flexible way to the new type of functionalized 2-amino-4H-chromene scaffold containing uracil-like moiety, which are relevant compounds for diverse biomedical applications.
References
1.
10.1016/j.mencom.2017.11.006_bib0005
Domino Reactions: Concepts for Efficient Organic Synthesis,
2014
2.

Wender P.A.
Natural Product Reports,
2014
3.

Clarke P.A., Santos S., Martin W.H.
Green Chemistry,
2007
4.
Vereshchagin A.N., Elinson M.N., Anisina Y.E., Ryzhkov F.V., Goloveshkin A.S., Bushmarinov I.S., Zlotin S.G., Egorov M.P.
Mendeleev Communications,
2015
5.
10.1016/j.mencom.2017.11.006_bib0025
Anastas
Green Chemistry: Theory and Practice,
1998
6.
Trofimov B.A., Schmidt E.Y., Bidusenko I.A., Cherimichkina N.A.
Mendeleev Communications,
2015
7.
![One-Pot ‘On-solvent’ Multicomponent Protocol for the Synthesis of Medicinally Relevant 4H-Pyrano[3,2-c]quinoline Scaffold](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Vereshchagin A.N., Elinson M.N., Nasybullin R.F., Ryzhkov F.V., Bobrovsky S.I., Bushmarinov I.S., Egorov M.P.
Helvetica Chimica Acta,
2015
8.
Elinson M.N., Ryzhkov F.V., Nasybullin R.F., Zaimovskaya T.A., Egorov M.P.
Mendeleev Communications,
2014
9.

Narayan S., Muldoon J., Finn M.G., Fokin V.V., Kolb H.C., Sharpless K.B.
Angewandte Chemie - International Edition,
2005
10.
Demchuk D.V., Elinson M.N., Nikishin G.I.
Mendeleev Communications,
2011
11.

Elinson M.N., Nasybullin R.F., Ryzhkov F.V., Egorov M.P.
Comptes Rendus Chimie,
2013
12.
Ananikov V.P., Khokhlova E.A., Egorov M.P., Sakharov A.M., Zlotin S.G., Kucherov A.V., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2015
13.
Ananikov V.P., Galkin K.I., Egorov M.P., Sakharov A.M., Zlotin S.G., Redina E.A., Isaeva V.I., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2016
14.

Evans B.E., Rittle K.E., Bock M.G., DiPardo R.M., Freidinger R.M., Whitter W.L., Lundell G.F., Veber D.F., Anderson P.S., Chang R.S., Lotti V.J., Cerino D.J., Chen T.B., Kling P.J., Kunkel K.A., et. al.
Journal of Medicinal Chemistry,
1988
15.

Hiranrat A., Chitbankluoi W., Mahabusarakam W., Limsuwan S., Voravuthikunchai S.P.
Natural Product Research,
2012
16.

Shimokawa S., Kumamoto T., Ishikawa T., Takashi M., Higuchi Y., Chaichantipyuth C., Chansakaow S.
Natural Product Research,
2013
17.
L. Johansen, C. Owens, C. Mawhinney, T.W. Chappell, A.T. Brown, M.G. Frank and R. Altmeyer, Patent WO 2008/33466 A1, 2008.
18.
W.-Q. Zhao and D. L. Alkon, Patent US 2013/0273545 A1, 2013.
19.
J. Collard, O. Khorkova-Sherman and J. H. Hsiao, Patent WO 2013/36403 A1, 2013.
20.
A. Tomiyama, H. Tomiyama, H. Miyamoto, K. Hayashi and S. Mochizuki, Patent EP 1767534 A1, 2007.
21.

Anderson D.R., Hegde S., Reinhard E., Gomez L., Vernier W.F., Lee L., Liu S., Sambandam A., Snider P.A., Masih L.
Bioorganic and Medicinal Chemistry Letters,
2005
22.
W. Hunter, P. Toleikis, D. Gravett, D. Grau, A. Borisy, C. Keith, B. Auspitz, M. Nichols, E. Jost-Price and G. N. Serbedzija, Patent US 2007/208134 A1, 2007.
23.

Kasibhatla S., Gourdeau H., Meerovitch K., Drewe J., Reddy S., Qiu L., Zhang H., Bergeron F., Bouffard D., Yang Q., Herich J., Lamothe S., Cai S.X., Tseng B.
Molecular Cancer Therapeutics,
2004
24.

Gourdeau H., Leblond L., Hamelin B., Desputeau C., Dong K., Kianicka I., Custeau D., Boudreau C., Geerts L., Cai S., Drewe J., Labrecque D., Kasibhatla S., Tseng B.
Molecular Cancer Therapeutics,
2004
25.

Skommer J., Wlodkowic D., Mättö M., Eray M., Pelkonen J.
Leukemia Research,
2006
26.
S. X. Cai, J.A. Drewe, S. Kasibhatla, W.E. Kemnitzer, B.Y. Tseng, C. Blais, H. Goudreau and D. Labrecque, Patent WO 2008/5572 A1, 2008.
27.

Connolly G.P., Duley J.A.
Trends in Pharmacological Sciences,
1999
28.

CARDINI C.E., PALADINI A.C., CAPUTTO R., LELOIR L.F.
Nature,
1950
29.

Persson B., Sjöman M., Niklasson F., Ronquist G.
European Urology,
1991
30.

Peters G.J., van Groeningen C.J., Laurensse E.J., Lankelma J., Leyva A., Pinedo H.M.
Cancer Chemotherapy and Pharmacology,
1987
31.
10.1016/j.mencom.2017.11.006_bib0155
Connolly
J. Pharmacol.,
1993
32.

Choi J.W., Shin C.Y., Choi M.S., Yoon S.Y., Ryu J.H., Lee J., Kim W., El Kouni M.H., Ko K.H.
Journal of Neurotrauma,
2008
33.
![Pot, atom and step-economic (PASE) synthesis of medicinally relevant spiro[oxindole-3,4′-pyrano[4,3-b]pyran] scaffold](/storage/images/resized/3SpVxcYL33bOvPq4sHxJLH2NeKNeDloahSUpNiO4_small_thumb.webp)
Elinson M.N., Ryzhkov F.V., Korolev V.A., Egorov M.P.
Heterocyclic Communications,
2016
34.
![Solvent-free and ‘on-water’ multicomponent assembling of aldehydes, 3-methyl-2-pyrazoline-5-one, and malononitrile: fast and efficient approach to medicinally relevant pyrano[2,3-c]pyrazole scaffold](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Elinson M.N., Nasybullin R.F., Ryzhkov F.V., Zaimovskaya T.A., Nikishin G.I.
Monatshefte fur Chemie,
2015
35.
![Green Approach to the Design of Functionalized Medicinally Privileged 4-Aryl-1,4-dihydropyrano[2,3-c]-pyrazole-5-carbonitrile Scaffold](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Ilovaisky A.I., Medvedev M.G., Merkulova V.M., Elinson M.N., Nikishin G.I.
Journal of Heterocyclic Chemistry,
2013
36.

Elinson M.N., Ryzhkov F.V., Vereshchagin A.N., Gorbunov S.V., Egorov M.P.
Comptes Rendus Chimie,
2015
37.
![Fast Efficient and General PASE Approach to Medicinally Relevant 4H,5H-Pyrano-[4,3-b]pyran-5-one and 4,6-Dihydro-5H-pyrano-[3,2-c]pyridine-5-one Scaffolds](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Elinson M.N., Ryzhkov F.V., Nasybullin R.F., Vereshchagin A.N., Egorov M.P.
Helvetica Chimica Acta,
2016
38.

Vereshchagin A.N., Elinson M.N., Ryzhkov F.V., Nasybullin R.F., Bobrovsky S.I., Goloveshkin A.S., Egorov M.P.
Comptes Rendus Chimie,
2015
39.

Elinson M.N., Feducovich S.K., Stepanov N.O., Vereshchagin A.N., Nikishin G.I.
Tetrahedron,
2008
40.
Elinson M.N., Medvedev M.G., Ilovaisky A.I., Merkulova V.M., Zaimovskaya T.A., Nikishin G.I.
Mendeleev Communications,
2013
41.

Elinson M.N., Nasybullin R.F., Ryzhkov F.V., Zaimovskaya T.A., Egorov M.P.
Monatshefte fur Chemie,
2014
42.

Patai S., Israeli Y.
Journal of the Chemical Society (Resumed),
1960