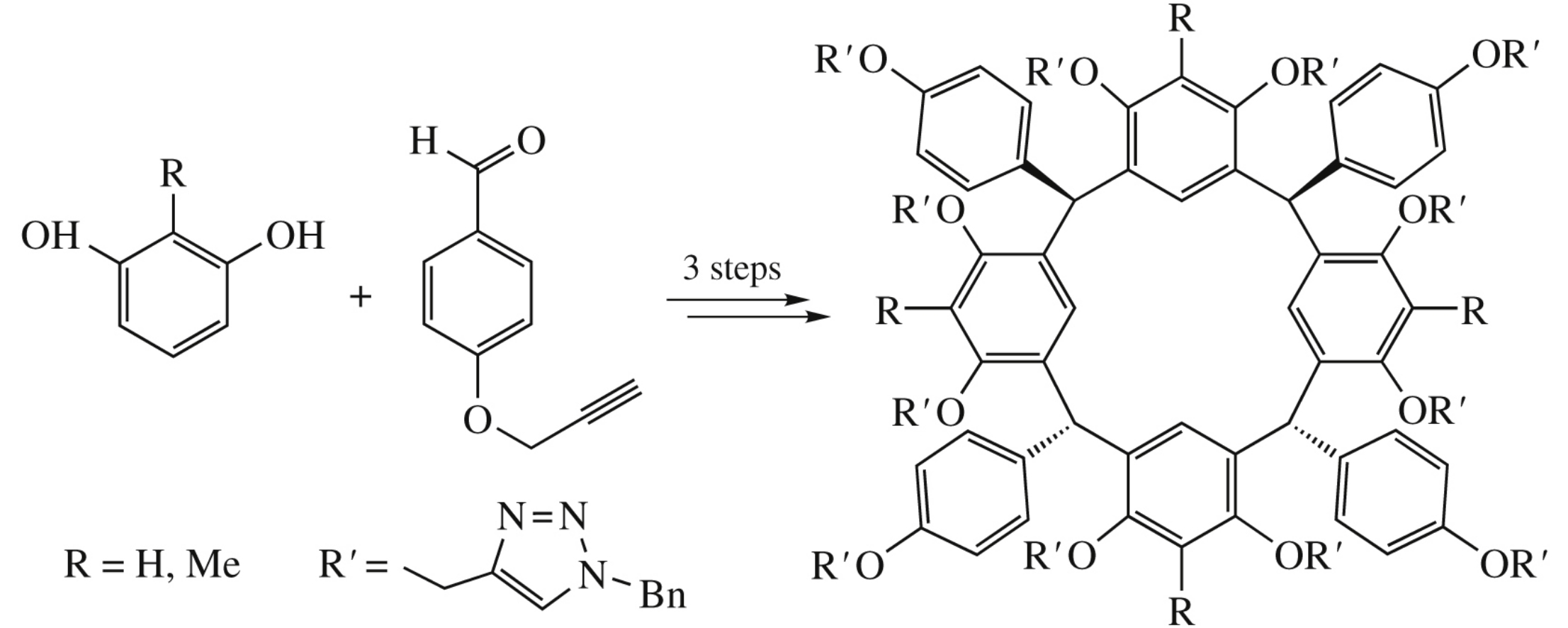
Abstract
A single-step acid-catalyzed condensation of resorcinol or 2-methylresorcinol with 4-(prop-2-yn-1-yloxy)benzaldehyde stereoselectively gives exclusively rctt-isomers of new calix[4]- resorcinols in chair conformation bearing four terminal alkyne groups at aromatic substituents. Further alkylation of free hydroxy groups with propargyl bromide affords new calix[4]- resorcinols containing 12 terminal alkyne groups. Subsequent click reaction of these compounds with benzyl azide results in highly functionalized calix[4]resorcinols with 12 triazole- linked branches.
References
1.

Bryant J.J., Bunz U.H.
Chemistry - An Asian Journal,
2013
2.

Tăbăcaru A., Furdui B., Ghinea I.O., Cârâc G., Dinică R.M.
Inorganica Chimica Acta,
2017
3.

Wang C., Ikhlef D., Kahlal S., Saillard J., Astruc D.
Coordination Chemistry Reviews,
2016
4.
Burilov V.A., Nugmanov R.I., Ibragimova R.R., Solovieva S.E., Antipin I.S.
Mendeleev Communications,
2015
5.

Ryu E., Zhao Y.
Organic Letters,
2005
6.

Morales-Sanfrutos J., Ortega-Muñoz M., Lopez-Jaramillo J., Hernandez-Mateo F., Santoyo-Gonzalez F.
Journal of Organic Chemistry,
2008
7.

Cecioni S., Matthews S.E., Blanchard H., Praly J., Imberty A., Vidal S.
Carbohydrate Research,
2012
8.
![Clicked and long spaced galactosyl- and lactosylcalix[4]arenes: new multivalent galectin-3 ligands](/storage/images/resized/ex6KJoZujZOZFZh7jGfeHauiftuB3CI7iwJVFRDg_small_thumb.webp)
Bernardi S., Fezzardi P., Rispoli G., Sestito S.E., Peri F., Sansone F., Casnati A.
Beilstein Journal of Organic Chemistry,
2014
9.
![p-tert-Butylcalix[4]arene core based ferrocenyl dendrimers: Novel sensor for toxic Hg2+ ion even in presence of Zn2+, Cu2+ and Ag+ ions](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Saravanan V., Kannan A., Rajakumar P.
Sensors and Actuators, B: Chemical,
2017
10.
![A fluorescent bis-NBD derivative of calix[4]arene: Switchable response to Ag+ and HCHO in solution phase](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Zhang S., Yang H., Ma Y., Fang Y.
Sensors and Actuators, B: Chemical,
2016
11.
![Calix[4]arene triazole-linked pyrene: click synthesis, assembly on graphene oxide, and highly sensitive carbaryl sensing in serum](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Sun Y., Mao X., Luo L., Tian D., Li H.
Organic and Biomolecular Chemistry,
2015
12.

Song M., Sun Z., Han C., Tian D., Li H., Kim J.S.
Chemistry - An Asian Journal,
2014
13.
![Click synthesis of triazole-linked calix[4]arene ionophores. Potentiometric and ESI-MS screening of ion-selectivity](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Czirok J.B., Jágerszki G., Tóth K., Révész Á., Drahos L., Bitter I.
Journal of Inclusion Phenomena and Macrocyclic Chemistry,
2013
14.
![A specific and ratiometric chemosensor for Hg2+ based on triazole coupled ortho-methoxyphenylazocalix[4]arene](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Wang N., Sun C., Chung W.
Tetrahedron,
2011
15.
![Triazole- and azo-coupled calix[4]arene as a highly sensitive chromogenic sensor for Ca2+ and Pb2+ ions](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Chang K., Su I., Lee G., Chung W.
Tetrahedron Letters,
2007
16.
![Novel analogs of antitumor agent calixarene 0118: Synthesis, cytotoxicity, click labeling with 2-[18F]fluoroethylazide, and in vivo evaluation](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Läppchen T., Dings R.P., Rossin R., Simon J.F., Visser T.J., Bakker M., Walhe P., van Mourik T., Donato K., van Beijnum J.R., Griffioen A.W., Lub J., Robillard M.S., Mayo K.H., Grüll H., et. al.
European Journal of Medicinal Chemistry,
2015
17.
![Design, synthesis, and drug solubilising properties of the first folate–calix[4]arene conjugate](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Consoli G.M., Granata G., Geraci C.
Organic and Biomolecular Chemistry,
2011
18.
![Novel effective dye sorbents: synthesis and properties of 1,2,3-triazole-modified thiacalix[4]arene polymers based on click chemistry](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Lai J., Yang F., Guo H., Jiao Z.
Iranian Polymer Journal (English Edition),
2014
19.

Tunstad L.M., Tucker J.A., Dalcanale E., Weiser J., Bryant J.A., Sherman J.C., Helgeson R.C., Knobler C.B., Cram D.J.
Journal of Organic Chemistry,
1989
20.

Thoden van Velzen E.U., Engbersen J.F., Reinhoudt D.N.
Journal of the American Chemical Society,
1994
21.

Hoegberg A.G.
Journal of Organic Chemistry,
1980
22.
![Rapid synthesis of dendrimers based on calix[4]resorcinarenes](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Yamakawa Y., Ueda M., Nagahata R., Takeuchi K., Asai M.
Journal of the Chemical Society Perkin Transactions 1,
1998
23.
![Synthesis of rctt, rccc, and rcct diastereomers of calix[4]methylresorcinarenes based on p-tolualdehyde. X-ray diffraction study of the rcct isomer. Formation of rctt and rccc cavitands in a cone conformation](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Prosvirkin A.V., Kazakova E.K., Gubaidullin A.T., Litvinov I.A., Gruner M., Habicher W.D., Konovalov A.I.
Russian Chemical Bulletin,
2005
24.
![One-step synthesis of rccc- and rctt-diastereomers of novel calix[4]resorcinols based on a para-thiophosphorylated derivative of benzaldehyde](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Knyazeva I.R., Sokolova V.I., Gruner M., Habicher W.D., Syakaev V.V., Khrizanforova V.V., Gabidullin B.M., Gubaidullin A.T., Budnikova Y.H., Burilov A.R., Pudovik M.A.
Tetrahedron Letters,
2013
25.
![Thiophosphorylated derivatives of meta- and ortho-hydroxybenzaldehydes in one-step syntheses of novel calix[4]resorcinols](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Knyazeva I.R., Matveeva V.I., Syakaev V.V., Gabidullin B.M., Gubaidullin A.T., Gruner M., Habicher W.D., Burilov A.R., Pudovik M.A.
Tetrahedron Letters,
2014
26.
![Phosphorus-containing calix[4]resorcinols: Synthesis and properties](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Knyazeva I.R., Burilov A.R., Pudovik M.A., Sinyashin O.G.
Phosphorus, Sulfur and Silicon and the Related Elements,
2016
27.

Pal M., Parasuraman K., Yeleswarapu K.R.
Organic Letters,
2003