Abstract
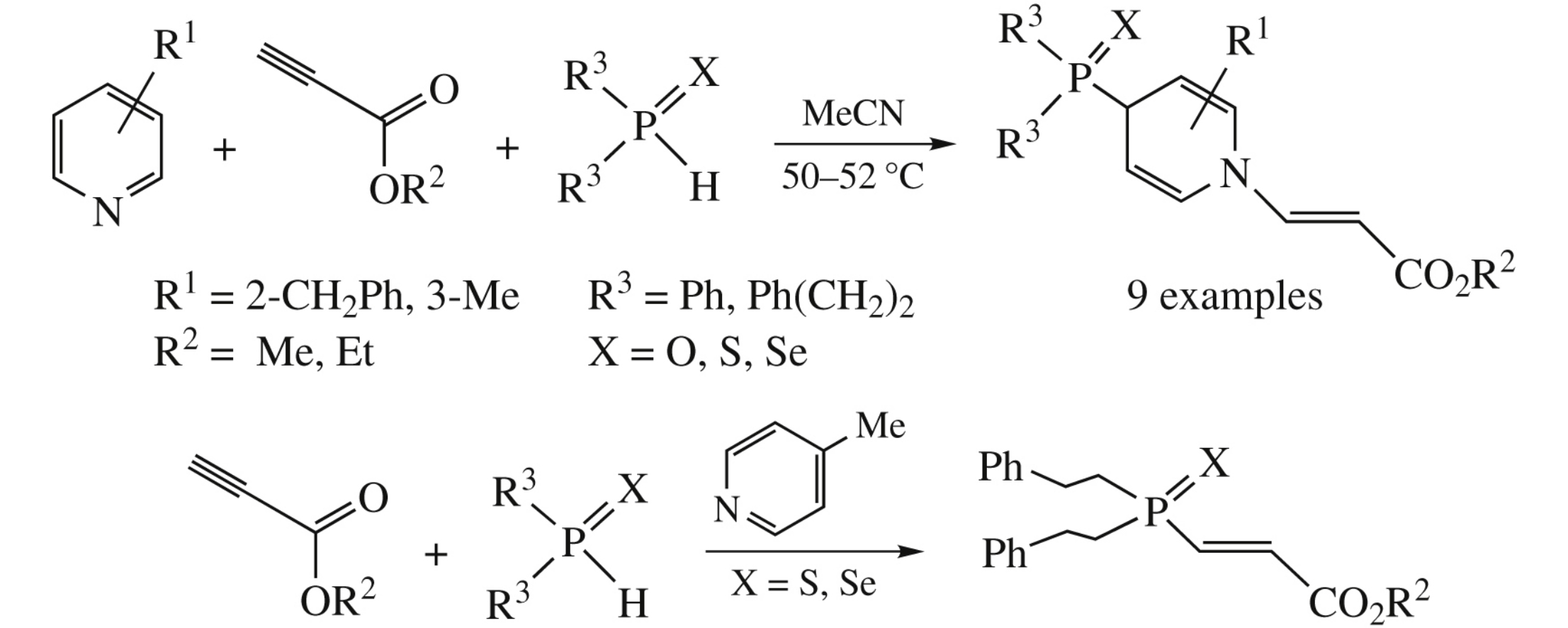
The non-catalyzed reaction of 2- and 3-substituted pyridines with alkyl propiolates and secondary phosphine chalcogenides (50–52°C, MeCN) produces stereo-, regio- and chemoselectively 1-[(E)-2-(alkoxycarbonyl)ethenyl]-4-chalcogenophosphoryl- 1,4-dihydropyridines in 57–90% yields, the adducts of pyridines with alkyl propiolates being the zwitterionic intermediates. 4-Methylpyridine mainly catalyzes nucleophilic addition of secondary phosphine chalcogenides to alkyl propiolates.
References
1.
Trofimov B.A., Nikitina L.P., Belyaeva K.V., Andriyankova L.V., Mal’kina A.G., Bagryanskaya I.Y., Afonin A.V., Ushakov I.A.
Mendeleev Communications,
2016
2.
Belyaeva K.V., Andriyankova L.V., Nikitina L.P., Afonin A.V., Ushakov I.A., Mal’kina A.G., Trofimov B.A.
Mendeleev Communications,
2015
3.

Allais C., Grassot J., Rodriguez J., Constantieux T.
Chemical Reviews,
2014
4.

Silva A., Silva E., Varandas P.
Synthesis,
2013
5.

Wan J., Liu Y.
RSC Advances,
2012
6.

Gusarova N.K., Volkov P.A., Ivanova N.I., Arbuzova S.N., Khrapova K.O., Albanov A.I., Smirnov V.I., Borodina T.N., Trofimov B.A.
Tetrahedron Letters,
2015
7.

Gusarova N.K., Volkov P.A., Ivanova N.I., Khrapova K.O., Albanov A.I., Afonin A.V., Borodina T.N., Trofimov B.A.
Tetrahedron Letters,
2016
8.
Karimi A.R., Bagherian F., Sourinia M.
Mendeleev Communications,
2013
9.
Stepnova E.A., Tikhonov V.E., Babushkina T.A., Klemenkova Z.S., Yamskov I.A.
Mendeleev Communications,
2006
10.
Korolev V.V., Vorobyev D.Y., Glebov E.M., Grivin V.P., Plyusnin V.F., Koshkin A.V., Fedorova O.A., Gromov S.P., Alfimov M.V., Shklyaev Y.V., Vshivkova T.S., Rozhkova Y.S., Tolstikov A.G., Lokshin V.A., Samat A., et. al.
Mendeleev Communications,
2006
11.

Prasanthi G., Prasad K.V., Bharathi K.
European Journal of Medicinal Chemistry,
2014
12.

Kumar R.S., Idhayadhulla A., Abdul Nasser A.J., Selvin J.
European Journal of Medicinal Chemistry,
2011
13.

Abbas H.S., El Sayed W.A., Fathy N.M.
European Journal of Medicinal Chemistry,
2010
14.

Fassihi A., Azadpour Z., Delbari N., Saghaie L., Memarian H.R., Sabet R., Alborzi A., Miri R., Pourabbas B., Mardaneh J.
European Journal of Medicinal Chemistry,
2009
15.
![Ring‐Opening of Pyridines with Acylacetylenes and Water: Straightforward Access to 5‐[(Z)‐Acylethenyl]amino‐2,4‐pentadienals](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Trofimov B.A., Andriyankova L.V., Belyaeva K.V., Nikitina L.P., Afonin A.V., Mal'kina A.G.
European Journal of Organic Chemistry,
2015
16.
10.1016/j.mencom.2017.11.004_sbref0030b
1979
17.
10.1016/j.mencom.2017.11.004_bib0035
Tenenbaum
1961
18.

Semenzin D., Etemad-Moghadam G., Albouy D., Diallo O., Koenig M.
Journal of Organic Chemistry,
1997
19.
![Addition reactions of heterocyclic compounds. Part XXXVIII. Cycl[3,2,2]azines and indolizines from pyridines and methyl propiolate](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Acheson R.M., Robinson D.A.
Journal of the Chemical Society C Organic,
1968
20.

Acheson R.M., Elmore N.F.
Advances in Heterocyclic Chemistry,
1979
21.

Clarke P.A., Santos S., Martin W.H.
Green Chemistry,
2007
22.
Vereshchagin A.N., Elinson M.N., Anisina Y.E., Ryzhkov F.V., Goloveshkin A.S., Bushmarinov I.S., Zlotin S.G., Egorov M.P.
Mendeleev Communications,
2015