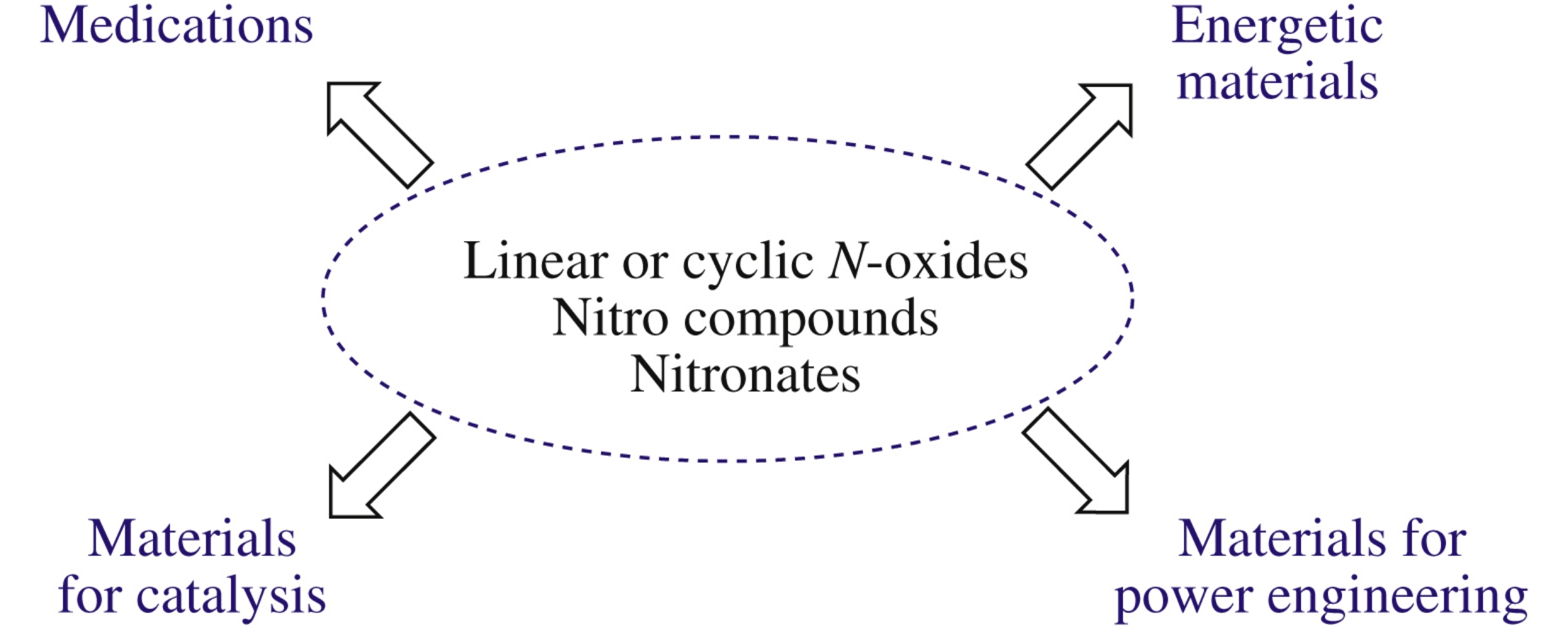
Abstract
Current trends and characteristic examples of recent advances in syntheses of practically important compounds bearing semi-polar nitrogen–oxygen bonds (NO-systems) are considered. Their applications for the preparation of pharmaceutically relevant molecules, energetic materials and some other useful products are briefly discussed with a focus on original reports published in the period 2015–2017.
References
1.

Nicolaou K.C., Hale C.R., Nilewski C., Ioannidou H.A.
Chemical Society Reviews,
2012
2.

Dorel R., Echavarren A.M.
Chemical Reviews,
2015
3.

Ananikov V., Liu X., Schneider U.
Chemistry - An Asian Journal,
2016
4.

Shirakawa S., Liu S., Kaneko S.
Chemistry - An Asian Journal,
2015
5.
Ananikov V.P., Khokhlova E.A., Egorov M.P., Sakharov A.M., Zlotin S.G., Kucherov A.V., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2015
6.
Ananikov V.P., Galkin K.I., Egorov M.P., Sakharov A.M., Zlotin S.G., Redina E.A., Isaeva V.I., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2016
7.
10.1016/j.mencom.2017.11.001_sbref0010a
Wang
Nitric Oxide Donors: For Pharmaceutical and Biological Applications,
2005
8.

Krause P., Wätzig E., Acil H., König S., Unthan-Fechner K., Tsikas D., Probst I.
Nitric Oxide - Biology and Chemistry,
2010
9.
10.1016/j.mencom.2017.11.001_bib0015
Agrawal
Organic Chemistry of Explosives,
2007
10.
10.1016/j.mencom.2017.11.001_bib0020
Ono
The Nitro Group in Organic Synthesis,
2001
11.
Zlotin S.G., Churakov A.M., Luk’yanov O.A., Makhova N.N., Sukhorukov A.Y., Tartakovsky V.A.
Mendeleev Communications,
2015
12.
10.1016/j.mencom.2017.11.001_sbref0030a
Fujii
2011
13.
10.1016/j.mencom.2017.11.001_sbref0030b
Contreras
2008
14.

Guglielmo S., Cortese D., Vottero F., Rolando B., Kommer V.P., Williams D.L., Fruttero R., Gasco A.
European Journal of Medicinal Chemistry,
2014
15.

Nortcliffe A., Ekstrom A.G., Black J.R., Ross J.A., Habib F.K., Botting N.P., O’Hagan D.
Bioorganic and Medicinal Chemistry,
2014
16.

Zhao N., Tian K., Cheng K., Han T., Hu X., Li D., Li Z., Hua H.
Bioorganic and Medicinal Chemistry,
2016
17.

Fang Y., Wang R., He M., Huang H., Wang Q., Yang Z., Li Y., Yang S., Jin Y.
Bioorganic and Medicinal Chemistry Letters,
2017
18.

Fershtat L.L., Larin A.A., Epishina M.A., Ovchinnikov I.V., Kulikov A.S., Ananyev I.V., Makhova N.N.
RSC Advances,
2016
19.
Kulikov A.S., Larin A.A., Fershtat L.L., Anikina L.V., Pukhov S.A., Klochkov S.G., Struchkova M.I., Romanova A.A., Ananyev I.V., Makhova N.N.
Arkivoc,
2017
20.
Fershtat L.L., Radzhabov M.R., Romanova A.A., Ananyev I.V., Makhova N.N.
Arkivoc,
2017
21.
![Versatile approach to heteroarylfuroxan derivatives from oximinofuroxans via a one-pot, nitration/thermolysis/[3+2]-cycloaddition cascade](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Larin A.A., Fershtat L.L., Ananyev I.V., Makhova N.N.
Tetrahedron Letters,
2017
22.
![Effective synthesis of 6-substituted 7H-tetrazolo[5,1-b][1,3,4]thiadiazines via a one-pot condensation/nitrosation/azide-tetrazole tautomerism reaction sequence](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Kulikov A.S., Epishina M.A., Fershtat L.L., Romanova A.A., Makhova N.N.
Tetrahedron Letters,
2017
23.

Ogurtsov V.A., Shastin A.V., Zlotin S.G., Rakitin O.A.
Tetrahedron Letters,
2016
24.
Ustyuzhanina N.E., Fershtat L.L., Gening M.L., Nifantiev N.E., Makhova N.N.
Mendeleev Communications,
2016
25.

Fernandes G.F., de Souza P.C., Marino L.B., Chegaev K., Guglielmo S., Lazzarato L., Fruttero R., Chung M.C., Pavan F.R., dos Santos J.L.
European Journal of Medicinal Chemistry,
2016
26.

Blangetti M., Rolando B., Chegaev K., Guglielmo S., Lazzarato L., Durante M., Masini E., Almirante N., Bastia E., Impagnatiello F., Fruttero R., Gasco A.
Bioorganic and Medicinal Chemistry Letters,
2017
27.

Bao N., Ou J., Xu M., Guan F., Shi W., Sun J., Chen L.
European Journal of Medicinal Chemistry,
2017
28.

Chen J., Wang T., Xu S., Zhang P., Lin A., Wu L., Yao H., Xie W., Zhu Z., Xu J.
European Journal of Medicinal Chemistry,
2017
29.

Fershtat L.L., Makhova N.N.
Russian Chemical Reviews,
2016
30.

Makhova N.N., Rakitin O.A.
Chemistry of Heterocyclic Compounds,
2017
31.

Serafim R.A., Pernichelle F.G., Ferreira E.I.
Expert Opinion on Drug Discovery,
2017
32.

Fershtat L.L., Makhova N.N.
ChemMedChem,
2017
33.

Hrabie J.A., Keefer L.K.
Chemical Reviews,
2002
34.

Smirnov G.A., Nikitin S.V., Gordeev P.B., Pokhvisneva G.V., Ternikova T.V., Luk’yanov O.A.
Russian Chemical Bulletin,
2015
35.
![Synthesis of 1,1’-[methylenebis(oxy)]-bis[3-alkyl-3-(2-hydroxyethyl)triaz-1-ene 2-oxides]](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Pokhvisneva G.V., Ternikova T.V., Smirnov G.A., Gordeev P.B., Nikitin S.V., Luk’yanov O.A.
Russian Chemical Bulletin,
2016
36.
![Synthesis of 3,3-bis(2-hydroxyethyl) and 3,3,3’-tris(2-hydroxyethyl) derivatives of 1,1’-[methylenebis(oxy)]bis(triaz-1-ene 2-oxides)](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Ternikova T.V., Pokhvisneva G.V., Smirnov G.A., Nikitin S.V., Gordeev P.B., Luk’yanov O.A.
Russian Chemical Bulletin,
2016
37.

Smirnov G.A., Gordeev P.B., Nikitin S.V., Pokhvisneva G.V., Ternikova T.V., Luk’yanov O.A.
Russian Chemical Bulletin,
2016
38.
Reaction of 3,3-disubstituted 1-chloromethoxy-1-triazene 2-oxides with tetramethylammonium hydroxide
Smirnov G.A., Gordeev P.B., Nikitin S.V., Nelyubina Y.V., Pokhvisneva G.V., Ternikova T.V., Luk’yanov O.A.
Mendeleev Communications,
2017
39.
10.1016/j.mencom.2017.11.001_bib0130
Pokhvisneva
Izv. Akad. Nauk, Ser. Khim.,
2017
40.
10.1016/j.mencom.2017.11.001_bib0135
Ternikova
Izv. Akad. Nauk, Ser. Khim.,
2017
41.
P.B., Gordeev, G.A., Smirnov, S.V., Nikitin, G.V., Pokhvisneva, T.V., Ternikova, O.A., Luk’yanov, P. B. Gordeev, G.A. Smirnov, S.V. Nikitin, G.V. Pokhvisneva, T.V. Ternikova and O.A. Luk’yanov, Izv. Akad. Nauk, Ser. Khim., 2017, in press.
42.

Smirnov G.A., Gordeev P.B., Nikitin S.V., Pokhvisneva G.V., Strelenko Y.A., Ternikova T.V., Luk´yanov O.A.
Russian Chemical Bulletin,
2015
43.

Sukhorukov A.Y., Sukhanova A.A., Zlotin S.G.
Tetrahedron,
2016
44.

Ballini R., Petrini M.
Advanced Synthesis and Catalysis,
2015
45.

Ballini R., Gabrielli S., Palmieri A., Petrini M.
Current Organic Chemistry,
2011
46.
10.1016/j.mencom.2017.11.001_sbref0155a
Baiazitov
Methods and Applications of Cycloaddition Reactions in Organic Syntheses,
2014
47.

Tabolin A., Sukhorukov A., Ioffe S., Dilman A.
Synthesis,
2017
48.

Perez V., Rabasso N., Fadel A.
European Journal of Organic Chemistry,
2015
49.

Rouf A., Şahin E., Tanyeli C.
Tetrahedron,
2017
50.
10.1016/j.mencom.2017.11.001_sbref0160c
Chemagin
Synthesis,
2010
51.

Kano T., Yamamoto A., Song S., Maruoka K.
Chemical Communications,
2011
52.
![Catalytic Asymmetric Formal [4 + 1] Annulation Leading to Optically Active cis-Isoxazoline N-Oxides](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Shi Z., Tan B., Leong W.W., Zeng X., Lu M., Zhong G.
Organic Letters,
2010
53.
![Enantioselective Construction of Oxa- and Aza-Angular Triquinanes through Tandem [4 + 1]/[3 + 2] Cycloaddition of Sulfur Ylides and Nitroolefins](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
An J., Lu L., Yang Q., Wang T., Xiao W.
Organic Letters,
2013
54.

Zhong C., Gautam L., Petersen J., Akhmedov N., Shi X.
Chemistry - A European Journal,
2010
55.
![Phosphane-Catalyzed [4+1] Annulation between Nitroalkenes and Morita-Baylis-Hillman Carbonates: Facile Synthesis of IsoxazolineN-Oxides by Phosphorus Ylides](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Zhou R., Duan C., Yang C., He Z.
Chemistry - An Asian Journal,
2014
56.

Sahoo G., Rahaman H., Madarász Á., Pápai I., Melarto M., Valkonen A., Pihko P.M.
Angewandte Chemie - International Edition,
2012
57.

Seebach D., Sun X., Sparr C., Ebert M., Schweizer W.B., Beck A.K.
Helvetica Chimica Acta,
2012
58.

Gurubrahamam R., Chen Y.M., Huang W., Chan Y., Chang H., Tsai M., Chen K.
Organic Letters,
2016
59.

Fedorov O.V., Levin V.V., Volodin A.D., Struchkova M.I., Korlyukov A.A., Dilman A.D.
Tetrahedron Letters,
2016
60.

Mikhaylov A.A., Dilman A.D., Novikov R.A., Khoroshutina Y.A., Struchkova M.I., Arkhipov D.E., Nelyubina Y.V., Tabolin A.A., Ioffe S.L.
Tetrahedron Letters,
2016
61.
![Novel Formal [3+3] Cycloaddition of Silyl Nitronates with Activated Cyclopropanes and Its Application in the Synthesis of Pyrroline-N-oxides](/storage/images/resized/xqixcltwJYe6H8Uco2JbAFfIOzt7UNKH0OcPOPzO_small_thumb.webp)
Ioffe S., Mikhaylov A., Novikov R., Khomutova Y., Arkhipov D., Korlyukov A., Tabolin A., Tomilov Y.
Synlett,
2014
62.

Tabolin A.A., Gorbacheva E.O., Novikov R.A., Khoroshutina Y.A., Nelyubina Y.V., Ioffe S.L.
Russian Chemical Bulletin,
2016
63.

Shved A.S., Tabolin A.A., Novikov R.A., Nelyubina Y.V., Timofeev V.P., Ioffe S.L.
European Journal of Organic Chemistry,
2016
64.

Sukhorukov A.Y., Kapatsyna M.A., Yi T.L., Park H.R., Naumovich Y.A., Zhmurov P.A., Khomutova Y.A., Ioffe S.L., Tartakovsky V.A.
European Journal of Organic Chemistry,
2014
65.

Naumovich Y.A., Buckland V.E., Sen'ko D.A., Nelyubina Y.V., Khoroshutina Y.A., Sukhorukov A.Y., Ioffe S.L.
Organic and Biomolecular Chemistry,
2016
66.

Zhmurov P.A., Khoroshutina Y.A., Novikov R.A., Golovanov I.S., Sukhorukov A.Y., Ioffe S.L.
Chemistry - A European Journal,
2017
67.

Berner O., Tedeschi L., Enders D.
European Journal of Organic Chemistry,
2002
68.

Halimehjani A.Z., Namboothiri I.N., Hooshmand S.E.
RSC Advances,
2014
69.

Halimehjani A.Z., Namboothiri I.N., Hooshmand S.E.
RSC Advances,
2014
70.

Halimehjani A.Z., Namboothiri I.N., Hooshmand S.E.
RSC Advances,
2014
71.

Motornov V.A., Muzalevskiy V.M., Tabolin A.A., Novikov R.A., Nelyubina Y.V., Nenajdenko V.G., Ioffe S.L.
Journal of Organic Chemistry,
2017
72.

Scheffler U., Mahrwald R.
Chemistry - A European Journal,
2013
73.

Nayak S., Panda P., Bhakta S., Mishra S.K., Mohapatra S.
RSC Advances,
2016
74.

Almaşi D., Alonso D.A., Nájera C.
Tetrahedron Asymmetry,
2007
75.

Marqués-López E., Herrera R.P., Christmann M.
Natural Product Reports,
2010
76.

Matsev O.V., Beletskaya I.P., Zlotin S.G.
Russian Chemical Reviews,
2011
77.

Alemán J., Cabrera S.
Chemical Society Reviews,
2013
78.

Hong B., Raja A., Sheth V.
Synthesis,
2015
79.

Ishikawa H., Shiomi S.
Organic and Biomolecular Chemistry,
2016
80.

Vinogradov M.G., Turova O.V., Zlotin S.G.
Russian Chemical Reviews,
2017
81.
10.1016/j.mencom.2017.11.001_bib0230
List
Science of Synthesis: Asymmetric Organocatalysis,
2012
82.

Kucherenko A.S., Lisnyak V.G., Kostenko A.A., Kochetkov S.V., Zlotin S.G.
Organic and Biomolecular Chemistry,
2016
83.

Tukhvatshin R.S., Kucherenko A.S., Nelyubina Y.V., Zlotin S.G.
ACS Catalysis,
2017
84.

Volla C.M., Atodiresei I., Rueping M.
Chemical Reviews,
2013
85.

Chauhan P., Mahajan S., Kaya U., Hack D., Enders D.
Advanced Synthesis and Catalysis,
2015
86.

Gasperi T., Vetica F., de Figueiredo R., Orsini M., Tofani D.
Synthesis,
2015
87.
10.1016/j.mencom.2017.11.001_bib0250
Kerton
Alternative Solvents for Green Chemistry,
2015
88.

Filatova E.V., Turova O.V., Kuchurov I.V., Kostenko A.A., Nigmatov A.G., Zlotin S.G.
Journal of Supercritical Fluids,
2016
89.
Sukhanova A.A., Nelyubina Y.V., Zlotin S.G.
Mendeleev Communications,
2016
90.
91.

Terrier F., Dust J.M., Buncel E.
Tetrahedron,
2012
92.

Roche S.P., Porco J.A.
Angewandte Chemie - International Edition,
2011
93.

Joshi-Pangu A., Cohen R.D., Tudge M.T., Chen Y.
Journal of Organic Chemistry,
2016
94.

Shawali A.S.
Chemical Reviews,
1993
95.

Bertuzzi G., Sinisi A., Caruana L., Mazzanti A., Fochi M., Bernardi L.
ACS Catalysis,
2016
96.
![3-R-4-Nitro-6,7-furoxanobenzo[d]isoxazoles – a new type of condensed nitroarenes capable of Diels–Alder reaction](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Bastrakov M.A., Starosotnikov A.M., Kachala V.V., Dalinger I.L., Shevelev S.A.
Chemistry of Heterocyclic Compounds,
2015
97.
Starosotnikov A.M., Bastrakov M.A., Pavlov A.A., Fedyanin I.V., Dalinger I.L., Shevelev S.A.
Mendeleev Communications,
2016
98.
![Synthesis of novel polycyclic heterosystems from 5-nitro[1,2,5]selenadiazolo[3,4-e]benzofuroxans](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Bastrakov M.A., Starosotnikov A.M., Pavlov A.A., Dalinger I.L., Shevelev S.A.
Chemistry of Heterocyclic Compounds,
2016
99.
Bastrakov M.A., Kucherova A.Y., Fedorenko A.K., Starosotnikov A.M., Fedyanin I.V., Dalinger I.L., Shevelev S.A.
Arkivoc,
2017
100.

Chistyakov V.A., Semenyuk Y.P., Morozov P.G., Prazdnova E.V., Chmykhalo V.K., Kharchenko E.Y., Kletskii M.E., Borodkin G.S., Lisovin A.V., Burov O.N., Kurbatov S.V.
Russian Chemical Bulletin,
2015
101.

Altomare C., Carotti A., Casini G., Cellamare S., Ferappi M., Gavuzzo E., Mazza F., Pantaleoni G., Giorgi R.
Journal of Medicinal Chemistry,
1988
102.
10.1016/j.mencom.2017.11.001_sbref0295b
Altomare
Arzneimittelforschung,
1992
103.
10.1016/j.mencom.2017.11.001_sbref0300a
Politzer
Mol. Phys.,
2014
104.

Yin P., Zhang Q., Shreeve J.M.
Accounts of Chemical Research,
2015
105.

Christe K.O., Dixon D.A., Vasiliu M., Wagner R.I., Haiges R., Boatz J.A., Ammon T.L.
Propellants, Explosives, Pyrotechnics,
2015
106.
10.1016/j.mencom.2017.11.001_sbref0310a
Jorgensen
J. Hazard. Mater.,
2011
107.
10.1016/j.mencom.2017.11.001_sbref0310b
Politzer
Cent. Eur. J. Energ. Mater.,
2013
108.
10.1016/j.mencom.2017.11.001_sbref0310c
Mendoza-Cortes
J. Comput. Chem.,
2016
109.

Ye C., An Q., Goddard III W.A., Cheng T., Liu W., Zybin S.V., Ju X.
Journal of Materials Chemistry A,
2015
110.

Klenov M.S., Guskov A.A., Anikin O.V., Churakov A.M., Strelenko Y.A., Fedyanin I.V., Lyssenko K.A., Tartakovsky V.A.
Angewandte Chemie - International Edition,
2016
111.

Klenov M.S., Anikin O.V., Churakov A.M., Strelenko Y.A., Fedyanin I.V., Ananyev I.V., Tartakovsky V.A.
European Journal of Organic Chemistry,
2015
112.

Churakov A.M., Tartakovsky V.A.
Chemical Reviews,
2004
113.

Guskov A.A., Klenov M.S., Churakov A.M., Tartakovsky V.A.
Russian Chemical Bulletin,
2016
114.

Tan B., Huang M., Huang H., Long X., Li J., Nie F., Huang J.
Propellants, Explosives, Pyrotechnics,
2013
115.

Voronin A.A., Zelenov V.P., Churakov A.M., Strelenko Y.A., Fedyanin I.V., Tartakovsky V.A.
Tetrahedron,
2014
116.

Klenov M.S., Anikin O.V., Guskov A.A., Churakov A.M., Strelenko Y.A., Ananyev I.V., Bushmarinov I.S., Dmitrienko A.O., Lyssenko K.A., Tartakovsky V.A.
European Journal of Organic Chemistry,
2016
117.

Voronin A.A., Churakov A.M., Klenov M.S., Strelenko Y.A., Fedyanin I.V., Tartakovsky V.A.
European Journal of Organic Chemistry,
2017
118.
10.1016/j.mencom.2017.11.001_sbref0355a
He
J. Mat. Chem. A.,
2016
119.
10.1016/j.mencom.2017.11.001_sbref0355b
Tang
Chem. Eur. J.,
2016
120.

Potassium 4,5-Bis(dinitromethyl)furoxanate: A Green Primary Explosive with a Positive Oxygen Balance
He C., Shreeve J.M.
Angewandte Chemie - International Edition,
2015
121.

Fershtat L.L., Larin A.A., Epishina M.A., Kulikov A.S., Ovchinnikov I.V., Ananyev I.V., Makhova N.N.
Tetrahedron Letters,
2016
122.

Yu Q., Yang H., Ju X., Lu C., Cheng G.
ChemistrySelect,
2017
123.

Liu N., Zeman S., Shu Y., Wu Z., Wang B., Yin S.
RSC Advances,
2016
124.

Peng Y., Jiang Y., Peng X., Liu J., Lai W.
ChemPhysChem,
2016
125.
10.1016/j.mencom.2017.11.001_sbref0380a
Li
Chem. Eng. Process,
2017
126.

Zhang Y., Zhang D., Dong K., Lv P., Pang S., Sun C.
Organic Process Research and Development,
2016
127.

Kuchurov I.V., Zharkov M.N., Fershtat L.L., Makhova N.N., Zlotin S.G.
ChemSusChem,
2017
128.
M. N. Zharkov, I.G. Kuchurov, I.V. Fomenkov and S. G. Zlotin, Patent RU 2610282 C1, 2017;
129.
10.1016/j.mencom.2017.11.001_sbref0390b
Zharkov
Synthesis,
2017
130.
M. N. Zharkov, I.G. Kuchurov, I.V. Fomenkov and S. G. Zlotin, Patent RU 2611009, 2017.
131.

Semakin A.N., Agababyan D.P., Kim S., Lee S., Sukhorukov A.Y., Fedina K.G., Oh J., Ioffe S.L.
Tetrahedron Letters,
2015
132.

Zavozin A.G., Simirskaya N.I., Nelyubina Y.V., Zlotin S.G.
Tetrahedron,
2016