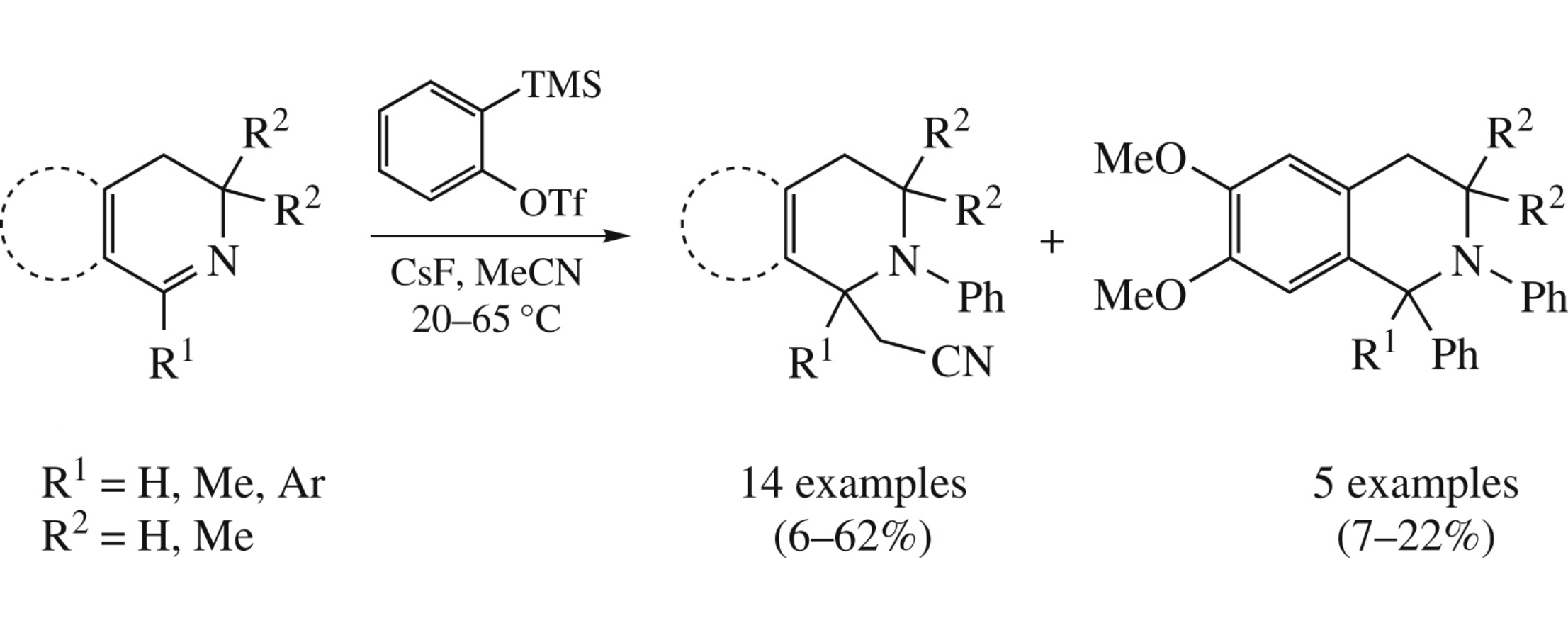
Abstract
Cyanomethyl-substituted tetrahydroisoquinolines and tetrahydrothieno[3,2-c]pyridines were synthesized by multicomponent reaction of the dihydro analogues of aforesaid systems with benzyne and acetonitrile. The products obtained relate to alkaloids of isoquinoline family of 1,2,3,4-tetrahydro level.
References
1.

Scott J.D., Williams R.M.
Chemical Reviews,
2002
2.

Antkiewicz-Michaluk L., Wąsik A., Michaluk J.
Neurotoxicity Research,
2013
3.
10.1016/j.mencom.2017.09.026_bib0015
Heterocycles in Natural Product Synthesis,
2011
4.

Huang W., Ni C., Zhao Y., Zhang W., Dilman A.D., Hu J.
Tetrahedron,
2012
5.

Statkova-Abeghe S., Angelov P.A., Ivanov I., Nikolova S., Kochovska E.
Tetrahedron Letters,
2007
6.

Barham J.P., John M.P., Murphy J.A.
Beilstein Journal of Organic Chemistry,
2014
7.

Taylor A.M., Schreiber S.L.
Organic Letters,
2005
8.

Singh K., Singh P., Kaur A., Singh P.
Synlett,
2012
9.

Boess E., Schmitz C., Klussmann M.
Journal of the American Chemical Society,
2012
10.

Nobuta T., Tada N., Fujiya A., Kariya A., Miura T., Itoh A.
Organic Letters,
2013
11.

Hu J., Wang J., Nguyen T.H., Zheng N.
Beilstein Journal of Organic Chemistry,
2013
12.

Nawaz F., Mohanan K., Charles L., Rajzmann M., Bonne D., Chuzel O., Rodriguez J., Coquerel Y.
Chemistry - A European Journal,
2013
13.

Bunescu A., Piemontesi C., Wang Q., Zhu J.
Chemical Communications,
2013
14.

Hendrick C.E., Wang Q.
Journal of Organic Chemistry,
2014
15.

Shi J., Qiu D., Wang J., Xu H., Li Y.
Journal of the American Chemical Society,
2015
16.
![Synthesis of Indazoles by the [3+2] Cycloaddition of Diazo Compounds with Arynes and Subsequent Acyl Migration](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Liu Z., Shi F., Martinez P.D., Raminelli C., Larock R.C.
Journal of Organic Chemistry,
2007
17.
Kopchuk D.S., Nikonov I.L., Zyryanov G.V., Nosova E.V., Kovalev I.S., Slepukhin P.A., Rusinov V.L., Chupakhin O.N.
Mendeleev Communications,
2015
18.

Jeganmohan M., Cheng C.
Chemical Communications,
2006
19.

Bhojgude S.S., Bhunia A., Biju A.T.
Accounts of Chemical Research,
2016
20.

Liu K., Liu L., Gu C., Dai B., He L.
RSC Advances,
2016
21.

Suh S., Chenoweth D.M.
Organic Letters,
2016
22.

Stephens D., Zhang Y., Cormier M., Chavez G., Arman H., Larionov O.V.
Chemical Communications,
2013
23.

Bhunia A., Roy T., Pachfule P., Rajamohanan P.R., Biju A.T.
Angewandte Chemie - International Edition,
2013
24.

Bhunia A., Porwal D., Gonnade R.G., Biju A.T.
Organic Letters,
2013
25.

Sha F., Huang X.
Angewandte Chemie - International Edition,
2009
26.

Bhunia A., Roy T., Gonnade R.G., Biju A.T.
Organic Letters,
2014
27.

Glushkov V.A., Shurov S.N., Maiorova O.A., Postanogova G.A., Feshina E.V., Shklyaev Y.V.
Chemistry of Heterocyclic Compounds,
2001
28.

Alajarin M., Lopez-Leonardo C., Raja R., Orenes R.
Organic Letters,
2011
29.

Lopez-Leonardo C., Raja R., López-Ortiz F., Ángel del Águila-Sánchez M., Alajarin M.
European Journal of Organic Chemistry,
2014
30.
![Enamide-Benzyne-[2 + 2] Cycloaddition: Stereoselective Tandem [2 + 2]−Pericyclic Ring-Opening−IntramolecularN-Tethered [4 + 2] Cycloadditions](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Feltenberger J.B., Hayashi R., Tang Y., Babiash E.S., Hsung R.P.
Organic Letters,
2009
31.

Varlamov A.V., Guranova N.I., Novikov R.A., Ilyushenkova V.V., Khrustalev V.N., Baleeva N.S., Borisova T.N., Voskressensky L.G.
RSC Advances,
2016