Abstract
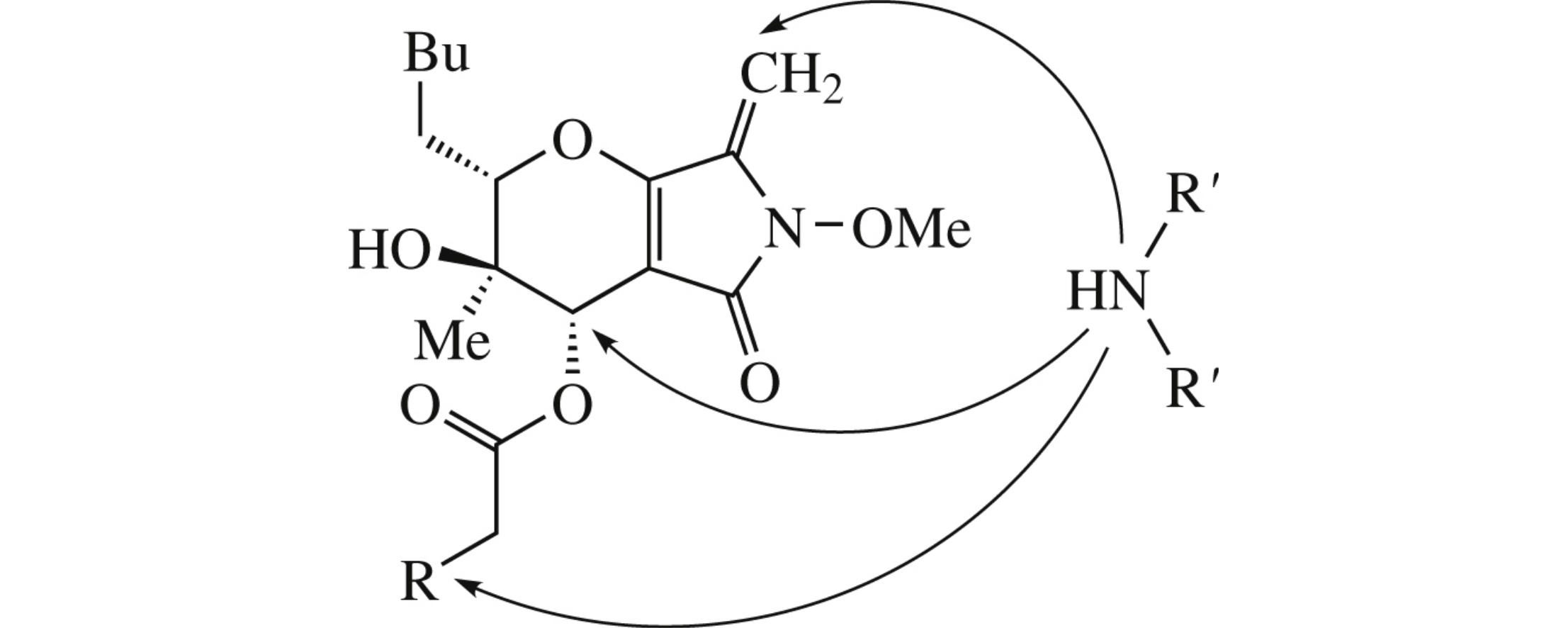
6-O-Acylated phaeosphaeride A and phaeosphaeride B in reaction with secondary cyclic amines undergo stereospecific replacement of acyloxy group by pharmacophoric amino moiety. In case of chloroacetoxy derivative, chlorine displacement by such amines proceeds in the presence of triethylamine.
References
1.

Maloney K.N., Hao W., Xu J., Gibbons J., Hucul J., Roll D., Brady S.F., Schroeder F.C., Clardy J.
Organic Letters,
2006
2.

Kobayashi K., Kobayashi Y., Nakamura M., Tamura O., Kogen H.
Journal of Organic Chemistry,
2015
3.
10.1016/j.mencom.2017.09.020_bib0015
Abzianidze
Acta Crystallogr.,
2015
4.

Abzianidze V.V., Prokofieva D.S., Chisty L.A., Bolshakova K.P., Berestetskiy A.O., Panikorovskii T.L., Bogachenkov A.S., Holder A.A.
Bioorganic and Medicinal Chemistry Letters,
2015
5.
Abzianidze V.V., Bolshakova K.P., Prokofieva D.S., Berestetskiy A.O., Kuznetsov V.A., Trishin Y.G.
Mendeleev Communications,
2017
6.

Kawatsura M., Uchida K., Terasaki S., Tsuji H., Minakawa M., Itoh T.
Organic Letters,
2014
7.

Hirakawa T., Ikeda K., Ikeda D., Tanaka T., Ogasa H., Kawatsura M., Itoh T.
Tetrahedron,
2011
8.

Takeuchi R., Ue N., Tanabe K., Yamashita K., Shiga N.
Journal of the American Chemical Society,
2001
9.

Roth H.J., Langer G.
Archiv der Pharmazie,
1968
10.
10.1016/j.mencom.2017.09.020_bib0050
Xun
Zhongguo Yaowu Huaxue Zazhi (Chin. J. Med. Chem.),
2005
11.

IWASA K., SUGIURA M., TAKAO N.
Chemical and Pharmaceutical Bulletin,
2011
12.

Kobayashi K., Kunimura R., Tanaka K., Tamura O., Kogen H.
Tetrahedron,
2017