Abstract
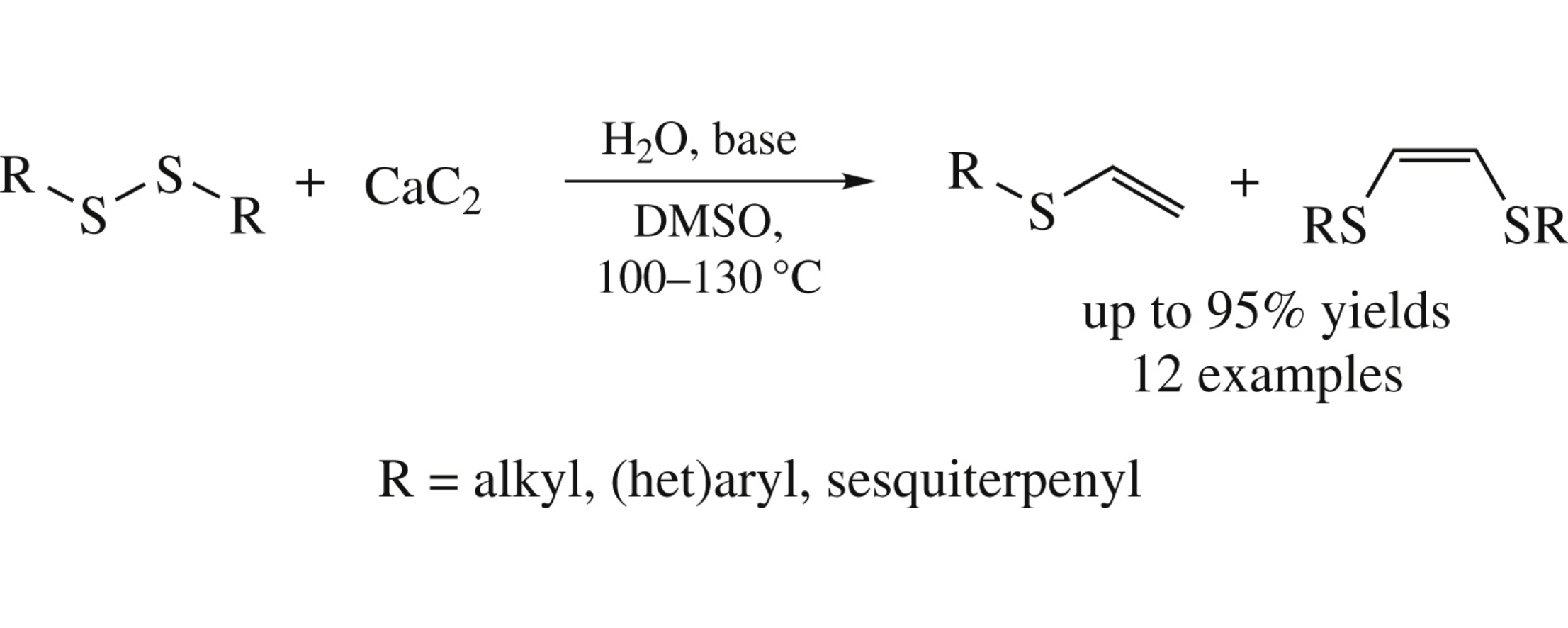
Bis-thioethenes and vinyl thioethers were obtained from the reaction of disulfides and calcium carbide in good to high yields using a simple synthetic procedure and common laboratory setup. The tolerance of the reaction was investigated by the examples of aliphatic, aromatic, heteroaromatic and sesquiterpenic substrates.
References
1.

Perin G., Lenardão E.J., Jacob R.G., Panatieri R.B.
Chemical Reviews,
2009
2.

Settambolo R., Mariani M., Caiazzo A.
Journal of Organic Chemistry,
1998
3.

Shimomura O., Sato T., Tomita I., Suzuki M., Endo T.
Journal of Polymer Science, Part A: Polymer Chemistry,
1997
4.

Orlov N.V., Chistyakov I.V., Starikova Z.A., Ananikov V.P., Beletskaya I.P.
Russian Chemical Bulletin,
2013
5.

Ananikov V.P., Orlov N.V., Zalesskiy S.S., Beletskaya I.P., Khrustalev V.N., Morokuma K., Musaev D.G.
Journal of the American Chemical Society,
2012
6.

Beghdadi S., Miladi I.A., Addis D., Romdhane H.B., Bernard J., Drockenmuller E.
Polymer Chemistry,
2012
7.
10.1016/j.mencom.2017.09.015_sbref0005g
Ananikov
Top. Organomet. Chem.,
2013
8.

Galkin K.I., Ananikov V.P.
Russian Chemical Reviews,
2016
9.

Irrgeher M., Schmidt H., Bretterbauer K., Gabriel H., Schwarzinger C.
Monatshefte fur Chemie,
2011
10.

Nakamura M., Endo K., Nakamura E.
Organic Letters,
2005
11.

Rodygin K.S., Werner G., Kucherov F.A., Ananikov V.P.
Chemistry - An Asian Journal,
2016
12.

Rodygin K.S., Ananikov V.P.
Green Chemistry,
2016
13.
Rodygin K.S., Kostin A.A., Ananikov V.P.
Mendeleev Communications,
2015
14.

Pearson W.H., Lee I.Y., Mi Y., Stoy P.
Journal of Organic Chemistry,
2004
15.

16.

Shaikh A.K., Cobb A.J., Varvounis G.
Organic Letters,
2012
17.
![Tandem photocyclization-intramolecular addition reactions of aryl vinyl sulfides. Observation of a novel [2 + 2] cycloaddition-allylic sulfide rearrangement](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Dittami J.P., Nie X.Y., Nie H., Ramanathan H., Buntel C., Rigatti S., Bordner J., Decosta D.L., Williard P.
Journal of Organic Chemistry,
1992
18.
![A novel entry into the bicyclo[5.4.0]undecane ring system](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Rigby J.H., Sage J.M., Raggon J.
Journal of Organic Chemistry,
1982
19.
![Short synthesis of bicyclo[3.2.2]nona-3,6,8-trien-2-one](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Rigby J.H., Sage J.M.
Journal of Organic Chemistry,
1983
20.
![[2 + 2] cycloadditions of 2,2-bis(trifluoromethyl)ethylene-1,1-dicarbonitrile with vinyl sulfides and ketene S,S-acetals](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Brückner R., Huisgen R.
Tetrahedron Letters,
1990
21.

Trost B.M., Tanigawa Y.
Journal of the American Chemical Society,
1979
22.

Kopping B., Chatgilialoglu C., Zehnder M., Giese B.
Journal of Organic Chemistry,
1992
23.

Moriarty R.M., Vaid R.K., Duncan M.P.
Synthetic Communications,
1987
24.

Davis F.A., Reddy R.T., Han W., Carroll P.J.
Journal of the American Chemical Society,
1992
25.

Glahsl G., Herrmann R.
Journal of the Chemical Society Perkin Transactions 1,
1988
26.

O'Mahony G.E., Ford A., Maguire A.R.
Journal of Sulfur Chemistry,
2012
27.

Nakabayashi K., Abiko Y., Mori H.
Macromolecules,
2013
28.

Liu J., Ueda M.
Journal of Materials Chemistry A,
2009
29.

Abiko Y., Matsumura A., Nakabayashi K., Mori H.
Polymer,
2014
30.

Kausar A., Zulfiqar S., Sarwar M.I.
Polymer Reviews,
2014
31.

Perez-Pineiro R., Dai S., Alvarez-Puebla R., Wigginton J., Al-Hourani B.J., Fenniri H.
Tetrahedron Letters,
2009
32.

Huang H., Chang C., Liu I., Tsai H., Lai M., Tsiang R.C.
Journal of Polymer Science, Part A: Polymer Chemistry,
2005
33.

Beletskaya I., Ananikov V., Kabeshov M.
Synlett,
2005
34.

Gusarova N.K., Chernysheva N.A., Yas’ko S.V., Trofimov B.A.
Russian Chemical Bulletin,
2013
35.
10.1016/j.mencom.2017.09.015_sbref0075b
Trofimov
Zh. Org. Khim.,
1973
36.

Fernandes A.C., Romão C.C.
Tetrahedron,
2006
37.

Yoo B.W., Song M.S., Park M.C.
Synthetic Communications,
2007
38.

Abele E., Dzenitis O., Rubina K., Lukevics E.
Chemistry of Heterocyclic Compounds,
2002
39.

Lo Conte M., Pacifico S., Chambery A., Marra A., Dondoni A.
Journal of Organic Chemistry,
2010
40.

Kondoh A., Takami K., Yorimitsu H., Oshima K.
Journal of Organic Chemistry,
2005
41.

Beletskaya I.P., Ananikov V.P.
Chemical Reviews,
2011
42.

Ananikov V.P., Orlov N.V., Beletskaya I.P.
Russian Chemical Bulletin,
2005
43.

Okimoto Y., Sakaguchi S., Ishii Y.
Journal of the American Chemical Society,
2002
44.

Petukhova N.P., Prilezhaeva E.N., Voropaev V.N.
Russian Chemical Bulletin,
1972
45.

Ananikov V.P., Orlov N.V., Kabeshov M.A., Beletskaya I.P., Starikova Z.A.
Organometallics,
2008
46.

Werner G., Rodygin K.S., Kostin A.A., Gordeev E.G., Kashin A.S., Ananikov V.P.
Green Chemistry,
2017
47.

Gyrdymova Y.V., Sudarikov D.V., Rubtsova S.A., Kuchin A.V.
Chemistry of Natural Compounds,
2017
48.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009
49.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007
50.
10.1016/j.mencom.2017.09.015_bib0105
Sheldrick
Acta Crystallogr.,,
2015
51.

Chavan M.J., Wakte P.S., Shinde D.B.
Phytomedicine,
2010
52.

Zhang W., Yao Z., Zhang Y., Zhang X., Takaishi Y., Duan H.
Planta Medica,
2010
53.

Venturi C.R., Danielli L.J., Klein F., Apel M.A., Montanha J.A., Bordignon S.A., Roehe P.M., Fuentefria A.M., Henriques A.T.
Pharmaceutical Biology,
2014
54.

Astani A., Reichling J., Schnitzler P.
Evidence-based Complementary and Alternative Medicine,
2011
55.

Wu Q., Wang W., Dai X., Wang Z., Shen Z., Ying H., Yu C.
Journal of Ethnopharmacology,
2012