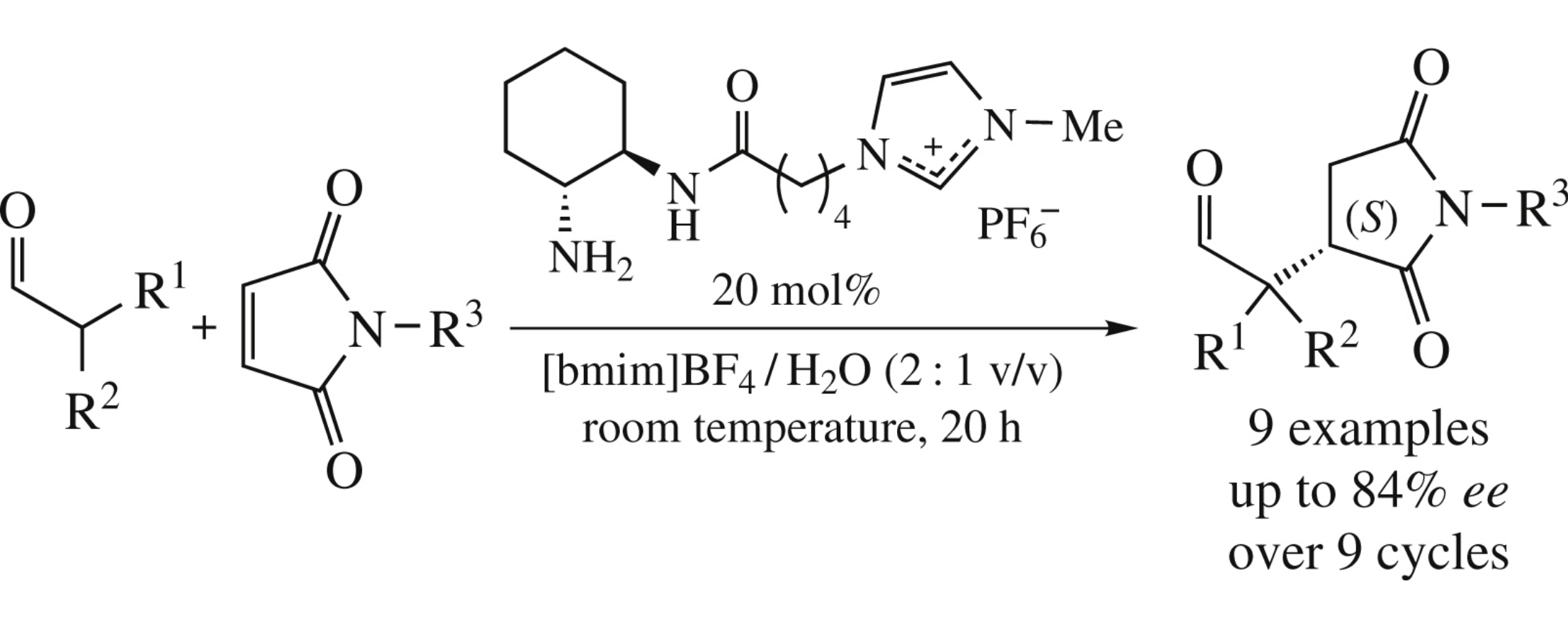
Abstract
A primary amine-based aqueous ionic liquid-supported recyclable catalytic system for asymmetric Michael addition of aldehydes to maleimides has been developed. The best enantioselectivity (up to 84% ee) was attained at the [bmim]BF4/water 2 : 1 (v/v) ratio.
References
1.
Science of Synthesis: Asymmetric Organocatalysis, vol. 1, eds. B. List and K. Maruoka, vol. 2, ed. B. List, Thieme, Stuttgart, 2012.
2.
10.1016/j.mencom.2017.09.014_sbref0005b
Comprehensive Enantioselective Organocatalysis: Catalysts, Reactions, and Applications,
2013
3.
Zlotin S.G., Makhova N.N.
Mendeleev Communications,
2010
4.

Raj M., Singh V.K.
Chemical Communications,
2009
5.

Reddy P.N., Padmaja P., Subba Reddy B.V., Rambabu G.
RSC Advances,
2015
6.

Kucherenko A.S., Syutkin D.E., Zlotin S.G.
Russian Chemical Bulletin,
2008
7.

Matsev O.V., Beletskaya I.P., Zlotin S.G.
Russian Chemical Reviews,
2011
8.

Chauhan P., Kaur J., Chimni S.S.
Chemistry - An Asian Journal,
2012
9.

Fredenhagen A., Tamura S.Y., Kenny P.T., Komura H., Naya Y., Nakanishi K., Nishiyama K., Sugiura M., Kita H.
Journal of the American Chemical Society,
1987
10.
10.1016/j.mencom.2017.09.014_sbref0040b
Malochet-Grivois
Anti-Cancer Drug Des.,
1992
11.
10.1016/j.mencom.2017.09.014_sbref0040c
Ando
Clin. Cancer Res.,
2002
12.

Freiberg C., Brunner N.A., Schiffer G., Lampe T., Pohlmann J., Brands M., Raabe M., Häbich D., Ziegelbauer K.
Journal of Biological Chemistry,
2004
13.

Isaka M., Rugseree N., Maithip P., Kongsaeree P., Prabpai S., Thebtaranonth Y.
Tetrahedron,
2005
14.

Uddin J., Ueda K., Siwu E.R., Kita M., Uemura D.
Bioorganic and Medicinal Chemistry,
2006
15.

Nöth J., Frankowski K.J., Neuenswander B., Aubé J., Reiser O.
Journal of Combinatorial Chemistry,
2008
16.

Fenster E., Hill D., Reiser O., Aubé J.
Beilstein Journal of Organic Chemistry,
2012
17.

3,3-Dialkyl- and 3-Alkyl-3-Benzyl-Substituted 2-Pyrrolidinones: A New Class of Anticonvulsant Agents
Reddy P.A., Hsiang B.C., Latifi T.N., Hill M.W., Woodward K.E., Rothman S.M., Ferrendelli J.A., Covey D.F.
Journal of Medicinal Chemistry,
1996
18.

Das Sarma K., Zhang J., Huang Y., Davidson J.G.
European Journal of Organic Chemistry,
2006
19.

Spaltenstein A., Almond M.R., Bock W.J., Cleary D.G., Furfine E.S., Hazen R.J., Kazmierski W.M., Salituro F.G., Tung R.D., Wright L.L.
Bioorganic and Medicinal Chemistry Letters,
2000
20.

Kazmierski W.M., Andrews W., Furfine E., Spaltenstein A., Wright L.
Bioorganic and Medicinal Chemistry Letters,
2004
21.

Barnes D.M., Ji J., Fickes M.G., Fitzgerald M.A., King S.A., Morton H.E., Plagge F.A., Preskill M., Wagaw S.H., Wittenberger S.J., Zhang J.
Journal of the American Chemical Society,
2002
22.

Tang K., Zhang J.
Neurological Research,
2002
23.

Bartoli G., Bosco M., Carlone A., Cavalli A., Locatelli M., Mazzanti A., Ricci P., Sambri L., Melchiorre P.
Angewandte Chemie - International Edition,
2006
24.

Ye W., Jiang Z., Zhao Y., Goh S.L., Leow D., Soh Y., Tan C.
Advanced Synthesis and Catalysis,
2007
25.

Jiang Z., Ye W., Yang Y., Tan C.
Advanced Synthesis and Catalysis,
2008
26.

Jiang Z., Pan Y., Zhao Y., Ma T., Lee R., Yang Y., Huang K., Wong M., Tan C.
Angewandte Chemie - International Edition,
2009
27.

Zu L., Xie H., Li H., Wang J., Jiang W., Wang W.
Advanced Synthesis and Catalysis,
2007
28.

Alba A., Valero G., Calbet T., Font-Bardía M., Moyano A., Rios R.
Chemistry - A European Journal,
2010
29.

Tanaka F., Thayumanavan R., Barbas C.F.
Journal of the American Chemical Society,
2003
30.

Tanaka F., Thayumanavan R., Mase N., Barbas C.F.
Tetrahedron Letters,
2004
31.

Nakashima K., Kawada M., Hirashima S., Kosugi A., Kato M., Yoshida A., Koseki Y., Miura T.
Tetrahedron Asymmetry,
2016
32.

Zhao G., Xu Y., Sundén H., Eriksson L., Sayah M., Córdova A.
Chemical Communications,
2007
33.

Flores-Ferrándiz J., Fiser B., Gómez-Bengoa E., Chinchilla R.
European Journal of Organic Chemistry,
2015
34.

Yu F., Jin Z., Huang H., Ye T., Liang X., Ye J.
Organic and Biomolecular Chemistry,
2010
35.

Yu F., Sun X., Jin Z., Wen S., Liang X., Ye J.
Chemical Communications,
2010
36.

Avila A., Chinchilla R., Nájera C.
Tetrahedron Asymmetry,
2012
37.

Avila A., Chinchilla R., Gómez-Bengoa E., Nájera C.
Tetrahedron Asymmetry,
2013
38.

Flores-Ferrándiz J., Chinchilla R.
Tetrahedron Asymmetry,
2014
39.

Gómez-Bengoa E., Nájera C., Vízcaíno-Milla P., Sansano J., Fiser B.
Synthesis,
2015
40.

Fernandes T.D., Vizcaíno-Milla P., Ravasco J.M., Ortega-Martínez A., Sansano J.M., Nájera C., Costa P.R., Fiser B., Gómez-Bengoa E.
Tetrahedron Asymmetry,
2016
41.

Flores-Ferrándiz J., Chinchilla R.
Tetrahedron Asymmetry,
2017
42.

Kucherenko A.S., Siyutkin D.E., Nigmatov A.G., Chizhov A.O., Zlotin S.G.
Advanced Synthesis and Catalysis,
2012
43.

44.

Kashin A.S., Galkin K.I., Khokhlova E.A., Ananikov V.P.
Angewandte Chemie - International Edition,
2016