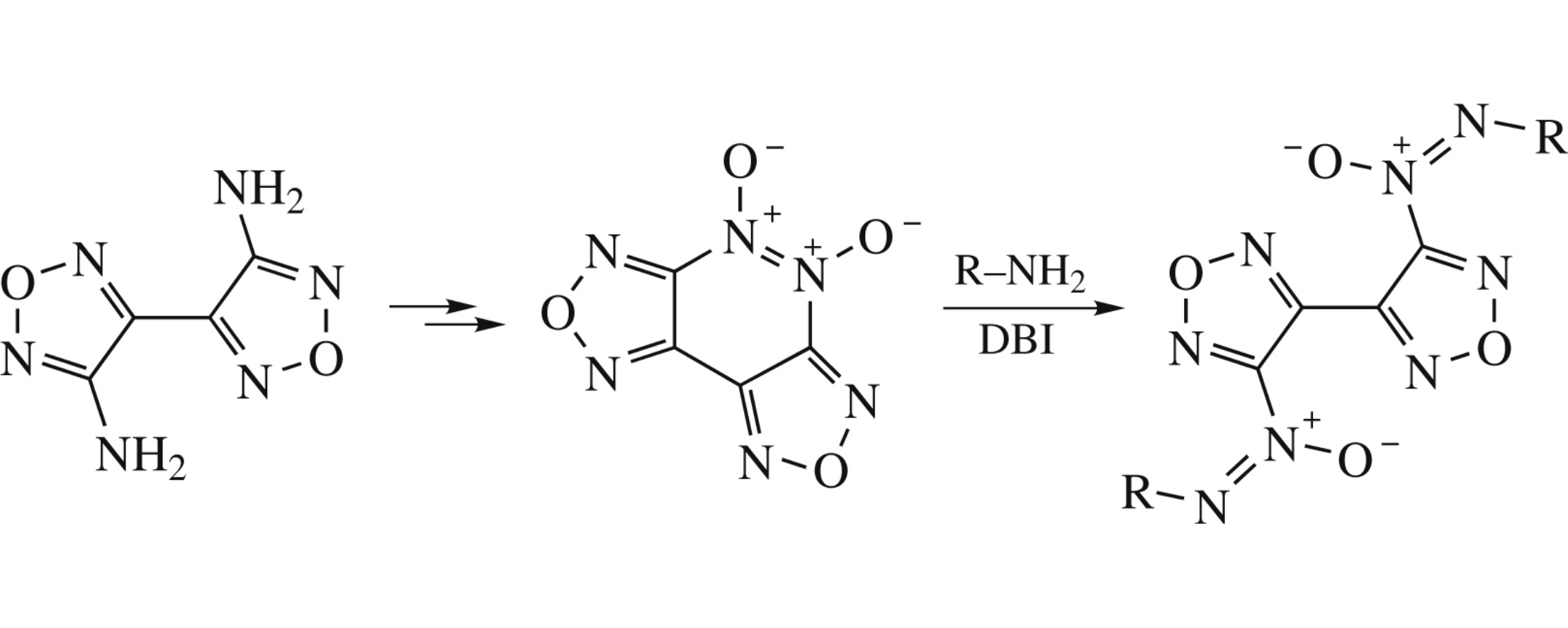
Abstract
The novel heterocyclic system, bis[1,2,5]oxadiazolo[3,4-c:3’,4’-e]pyridazine 4,5-dioxide, was obtained along with other products in the course of oxidation of 3,3’-bi-1,2,5-oxadiazole-4,4’-diamine. This compound acts as a synthetic equivalent of 4,4’-dinitroso-3,3’-bifurazan affording bis-diazene oxide derivative in the Kovacic reaction.
References
1.

Iachini Bellisarii F., Radico F., Muscente F., Horowitz J., De Caterina R.
Cardiovascular Drugs and Therapy,
2011
2.

Miller M.R., Megson I.L.
British Journal of Pharmacology,
2007
3.

4.

Kerwin J.F., Heller M.
Medicinal Research Reviews,
1994
5.
Ananikov V.P., Khokhlova E.A., Egorov M.P., Sakharov A.M., Zlotin S.G., Kucherov A.V., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2015
6.
Zlotin S.G., Churakov A.M., Luk’yanov O.A., Makhova N.N., Sukhorukov A.Y., Tartakovsky V.A.
Mendeleev Communications,
2015
7.
Bastrakov M.A., Starosotnikov A.M., Fedyanin I.V., Kachala V.V., Shevelev S.A.
Mendeleev Communications,
2014
8.
Fershtat L.L., Epishina M.A., Kulikov A.S., Makhova N.N.
Mendeleev Communications,
2015
9.
![An efficient access to (1H-tetrazol-5-yl)furoxan ammonium salts via a two-step dehydration/[3+2]-cycloaddition strategy](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Fershtat L.L., Epishina M.A., Kulikov A.S., Ovchinnikov I.V., Ananyev I.V., Makhova N.N.
Tetrahedron,
2015
10.
Konstantinova L.S., Amelichev S.A., Zlotin S.G., Struchkova M.I., Godovikova T.I., Rakitin O.A.
Mendeleev Communications,
2015
11.

Fershtat L.L., Ananyev I.V., Makhova N.N.
RSC Advances,
2015
12.
Fershtat L.L., Ashirbaev S.S., Kulikov A.S., Kachala V.V., Makhova N.N.
Mendeleev Communications,
2015
13.

Fershtat L.L., Larin A.A., Epishina M.A., Ovchinnikov I.V., Kulikov A.S., Ananyev I.V., Makhova N.N.
RSC Advances,
2016
14.

Ogurtsov V.A., Shastin A.V., Zlotin S.G., Rakitin O.A.
Tetrahedron Letters,
2016
15.

Fershtat L.L., Larin A.A., Epishina M.A., Kulikov A.S., Ovchinnikov I.V., Ananyev I.V., Makhova N.N.
Tetrahedron Letters,
2016
16.

Fershtat L.L., Makhova N.N.
Russian Chemical Reviews,
2016
17.
M. D. Dutov, Yu. V. Khropov, A. Ya. Kots, N.N. Belushkina, O.G. Busygina, I.S. Severina and S. A. Shevelev, Patent RU 2122582, 1997.
18.

Rehse K., Müller U.
Archiv der Pharmazie,
1995
19.

Kirilyuk I.A., Utepbergenov D.I., Mazhukin D.G., Fechner K., Mertsch K., Khramtsov V.V., Blasig I.E., Haseloff R.F.
Journal of Medicinal Chemistry,
1998
20.
![Vasorelaxant and antiplatelet activity of 4,7-dimethyl-1,2,5-oxadiazolo[3,4-d]pyridazine 1,5,6-trioxide: Role of soluble guanylate cyclase, nitric oxide and thiols](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Kots A.Y., Grafov M.A., Khropov Y.V., Betin V.L., Belushkina N.N., Busygina O.G., Yazykova M.Y., Ovchinnikov I.V., Kulikov A.S., Makhova N.N., Medvedeva N.A., Bulargina T.V., Severina I.S.
British Journal of Pharmacology,
2000
21.

Kots A.Y., Bian K., Murad F.
Current Medicinal Chemistry,
2011
22.
![Sodium arenetellurolate-catalyzed selective conversion of nitro aromatics to aromatic azoxy or azo compounds and its application for facile preparation of 3,3'- and 4,4'-bis[.beta.-(aryltelluro)vinyl]azobenzenes from (3- and 4-nitrophenyl)acetylenes](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Ohe K., Uemura S., Sugita N., Masuda H., Taga T.
Journal of Organic Chemistry,
1989
23.

Whittleton S.N., Dunitz J.D.
Acta Crystallographica Section B,
1982
24.
![The synthesis of benzo[c] cinnoline dioxides](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Kempter F.E., Castle R.N.
Journal of Heterocyclic Chemistry,
2009
25.

Kanoktanaporn S., MacBride J.A.
Journal of the Chemical Society Perkin Transactions 1,
1978
26.
10.1016/j.mencom.2017.09.005_bib0130
Nonciaux
Bull. Soc. Chim. Fr.,
1973
27.

Sheremetev A.B., Novikova T.S., Mel'nikova T.M., Khmel'nitskii L.I.
Russian Chemical Bulletin,
1990
28.

Khodot E.N., Svirskaya L.G., Chlenov I.E.
Russian Chemical Bulletin,
1994
29.
Mel’nikova T.M., Novikova T.S., Khmel’nitskii L.I., Sheremetev A.B.
Mendeleev Communications,
2001
30.

Batog L.V., Konstantinova L.S., Rozhkov V.Y.
Russian Chemical Bulletin,
2005
31.

Vlasova O.G., Rakitin O.A., Khmel'nitskii L.I.
Russian Chemical Bulletin,
1992
32.

Hurst D.
Australian Journal of Chemistry,
1983
33.

Rakitin O.A., Vlasova O.G., Chertanova L.F., Khmel'nitskii L.I.
Russian Chemical Bulletin,
1990
34.

Coburn M.D., Hiskey M.A., Lee K.-., Ott D.G., Stinecipher M.M.
Journal of Heterocyclic Chemistry,
1993
35.

Luk'yanov O.A., Smirnov G.A., Vasil'ev A.M.
Russian Chemical Bulletin,
1990
36.

Zlotin S.G., Luk'yanov O.A.
Russian Chemical Reviews,
1993
37.

Sheremetev A.B., Kulagina V.O., Kryazhevskikh I.A., Melnikova T.M., Aleksandrova N.S.
Russian Chemical Bulletin,
2002