Abstract
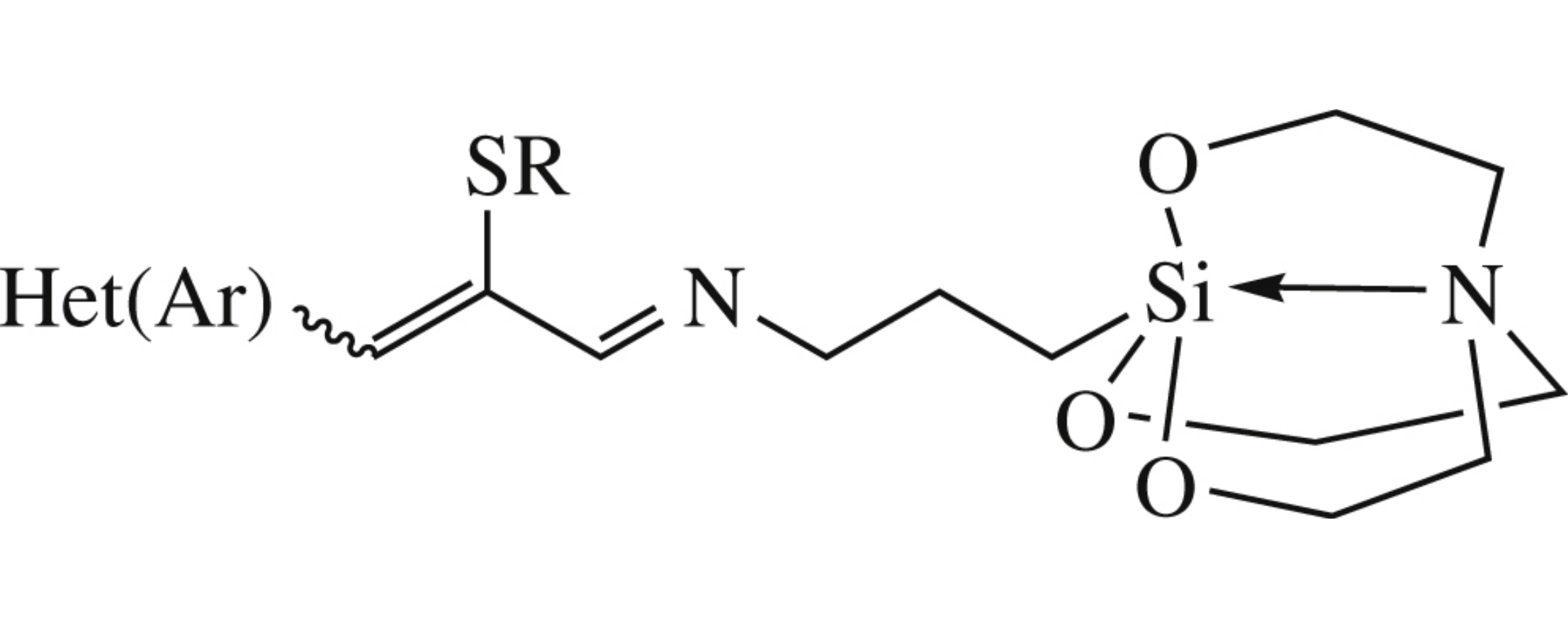
Novel highly functionalized and pharmacologically promising active silatranes equipped with an α,β-unsaturated imine group, Het(Ar)CH=C(SR)C(H)=N–, in the axial chain have been synthesized by the reaction of 3-aminopropylsilatrane and (Z)-2-alkylthio-3-(het)arylpropenals. The structures of these compounds were characterized by IR and 1H, 13C, 15N, 29Si, 1D and 2D NMR spectroscopy.
References
1.

Puri J.K., Singh R., Chahal V.K.
Chemical Society Reviews,
2011
2.
10.1016/j.mencom.2017.09.003_bib0010
Voronkov
Silatrany v meditsine i sel'skom khozyaistve (Silatranes in Medicine and Agriculture),
2005
3.

Jain N., Tripathi S.K., Nasim M.
International Journal of Polymeric Materials and Polymeric Biomaterials,
2013
4.

Materna K.L., Brennan B.J., Brudvig G.W.
Dalton Transactions,
2015
5.

Paul B., Vadivel S., Dhar S.S.
Chinese Chemical Letters,
2016
6.
Belyaeva V.V., Belogolova E.F., Bolgova Y.I., Trofimova O.M.
Mendeleev Communications,
2015
7.

Amide-tethered organosilatranes: Syntheses, structural characterization and photophysical properties
Singh G., Rani S., Saroa A., Promila, Arora A., Choquesillo-Lazarte D.
Inorganica Chimica Acta,
2015
8.

Hsieh S., Chao W., Hsieh C.
Journal of Nanoscience and Nanotechnology,
2009
9.

Huang K., Hsieh C., Kan H., Hsieh M., Hsieh S., Chau L., Cheng T., Lin W.
Sensors and Actuators, B: Chemical,
2012
10.

Lyubchenko Y.L., Shlyakhtenko L.S.
Methods,
2009
11.
Alentiev D.A., Chapala P.P., Filatova M.P., Finkelshtein E.S., Bermeshev M.V.
Mendeleev Communications,
2016
12.

Singh G., Arora A., Mangat S.S., Rani S., Kaur H., Goyal K., Sehgal R., Maurya I.K., Tewari R., Choquesillo-Lazarte D., Sahoo S., Kaur N.
European Journal of Medicinal Chemistry,
2016
13.

Li Z., Tian D.
Heterocyclic Communications,
2003
14.

Li Z., Song X., Su H., Chen J.
Heterocyclic Communications,
2005
15.

Singh G., Saroa A., Girdhar S., Rani S., Sahoo S., Choquesillo-Lazarte D.
Inorganica Chimica Acta,
2015
16.

Han A., Li L., Qing K., Qi X., Hou L., Luo X., Shi S., Ye F.
Bioorganic and Medicinal Chemistry Letters,
2013
17.

Ye F., Song X., Liu J., Xu X., Wang Y., Hu L., Wang Y., Liang G., Guo P., Xie Z.
Chemical Biology and Drug Design,
2015
18.

Adamovich S.N., Mirskov R.G., Mirskova A.N., Voronkov M.G.
Russian Chemical Bulletin,
2012
19.

Mirskova A.N., Adamovich S.N., Mirskov R.G., Voronkov M.G.
Russian Chemical Bulletin,
2014
20.
Adamovich S.N., Ushakov I.A., Mirskova A.N., Mirskov R.G., Voronov V.K.
Mendeleev Communications,
2014
21.
10.1016/j.mencom.2017.09.003_sbref0065a
Nie
Chin. J. Struct. Chem.,
2014
22.

Singh G., Girdhar S., Khullar S., Mandal S.K.
Journal of Coordination Chemistry,
2015
23.

Zuo J., Bi C., Fan Y., Buac D., Nardon C., Daniel K.G., Dou Q.P.
Journal of Inorganic Biochemistry,
2013
24.

Duff B., Reddy Thangella V., Creaven B.S., Walsh M., Egan D.A.
European Journal of Pharmacology,
2012
25.

Singh R., Mutneja R., Kaur V., Wagler J., Kroke E.
Journal of Organometallic Chemistry,
2013
26.

Dumitriu A., Cazacu M., Shova S., Turta C., Simionescu B.C.
Polyhedron,
2012
27.

Mutneja R., Singh R., Kaur V., Wagler J., Fels S., Kroke E.
New Journal of Chemistry,
2016
28.

Lin Y., Song B., Han A., Hu S., Ye F., Xie Z.
Phosphorus, Sulfur and Silicon and the Related Elements,
2011
29.
K. Shozo, Jpn. Kokai Tokkyo Koho, JP 62,212,392 (Chem. Abstr., 1988, 108, 221892t).
30.
K. Shozo, Jpn. Kokai Tokkyo Koho, JP 62,221,692 (Chem. Abstr., 1988, 108, 186983z).
31.
10.1016/j.mencom.2017.09.003_sbref0085c
Ding
Chem. J. Chin. Univ.,
1989
32.
Keiko N.A., Vchislo N.V., Verochkina E.A., Chuvashev Y.A., Larina L.I.
Mendeleev Communications,
2016
33.

Keiko N.A., Vchislo N.V.
Asian Journal of Organic Chemistry,
2016
34.

Keiko N.A., Vchislo N.V.
Asian Journal of Organic Chemistry,
2016
35.

Keiko N.A., Vchislo N.V., Verochkina E.A., Larina L.I.
Tetrahedron,
2014
36.

Calow A.D., Carbó J.J., Cid J., Fernández E., Whiting A.
Journal of Organic Chemistry,
2014
37.
Maltsev O.V., Kucherenko A.S., Zlotin S.G.
Mendeleev Communications,
2011
38.
Guryev A.A., Anokhin M.V., Averin A.D., Beletskaya I.P.
Mendeleev Communications,
2016
39.

Afonin A.V., Ushakov I.A., Vashchenko A.V., Simonenko D.E., Ivanov A.V., Vasil'tsov A.M., Mikhaleva A.I., Trofimov B.A.
Magnetic Resonance in Chemistry,
2009
40.

Afonin A.V., Toryashinova D.D., Schmidt E.Y.
Journal of Molecular Structure THEOCHEM,
2004