Abstract
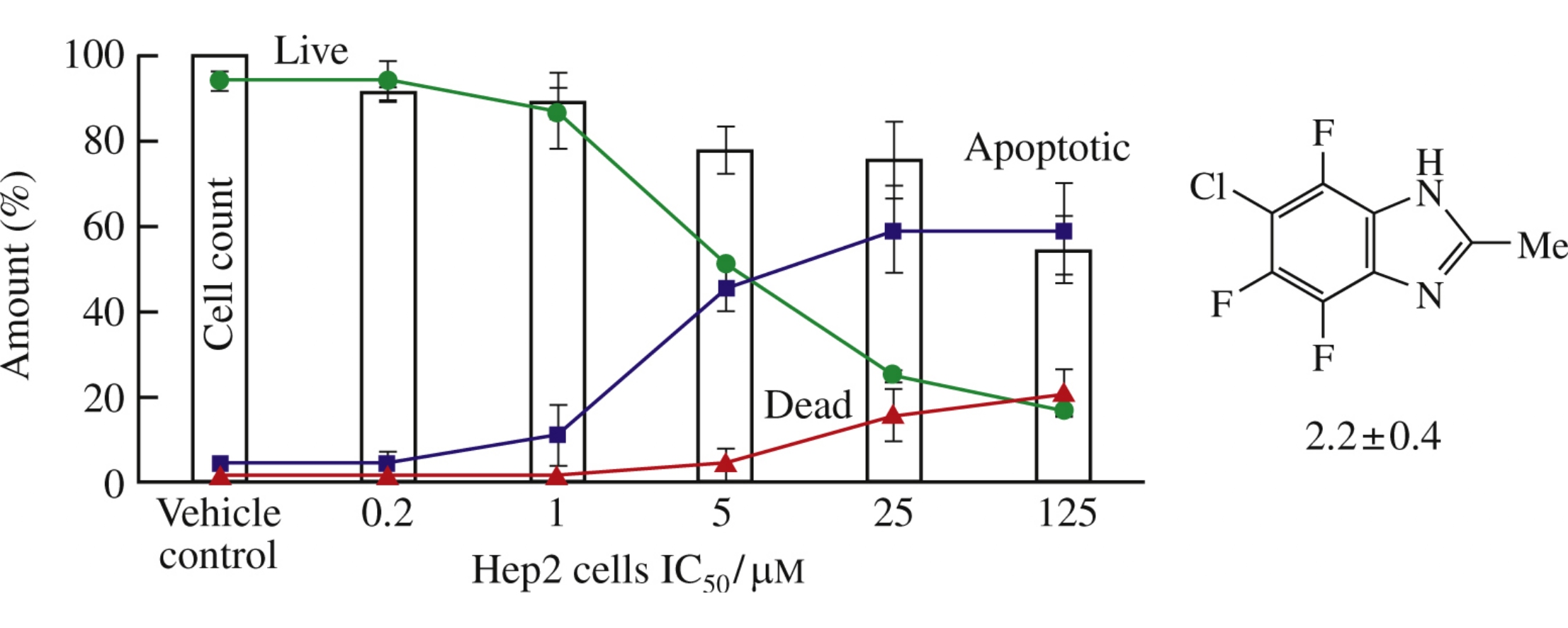
The title compounds synthesized from halogenated arene-1,2-diamines revealed, together with the latter, cytotoxicity towards the Hep2 (laryngeal epidermoid carcinoma) cells. The cytotoxicity was high and was accompanied by pronounced apoptotic activity at low concentrations for fluorinated diazoles, triazoles and selenadiazoles.
References
1.

O’Hagan D., Deng H.
Chemical Reviews,
2014
2.

O’Hagan D., B. Harper D.
Journal of Fluorine Chemistry,
1999
3.

Shchegol'kov E.V., Shchur I.V., Burgart Y.V., Saloutin V.I., Trefilova A.N., Ljushina G.A., Solodnikov S.Y., Markova L.N., Maslova V.V., Krasnykh O.P., Borisevich S.S., Khursan S.L.
Bioorganic and Medicinal Chemistry,
2017
4.

Zhou Y., Wang J., Gu Z., Wang S., Zhu W., Aceña J.L., Soloshonok V.A., Izawa K., Liu H.
Chemical Reviews,
2016
5.
Eletskaya B.Z., Konstantinova I.D., Paramonov A.S., Esipov R.S., Gruzdev D.A., Vigorov A.Y., Levit G.L., Miroshnikov A.I., Krasnov V.P., Charushin V.N.
Mendeleev Communications,
2016
6.
Kopchuk D.S., Nikonov I.L., Zyryanov G.V., Nosova E.V., Kovalev I.S., Slepukhin P.A., Rusinov V.L., Chupakhin O.N.
Mendeleev Communications,
2015
7.

Wang J., Sánchez-Roselló M., Aceña J.L., del Pozo C., Sorochinsky A.E., Fustero S., Soloshonok V.A., Liu H.
Chemical Reviews,
2013
8.

O’Hagan D.
Journal of Fluorine Chemistry,
2010
9.

Kirk K.L.
Journal of Fluorine Chemistry,
2006
10.

Isanbor C., O’Hagan D.
Journal of Fluorine Chemistry,
2006
11.
10.1016/j.mencom.2017.09.002_sbref0015a
Prakash Reddy
Organofluorine Compounds in Biology and Medicine,
2015
12.
10.1016/j.mencom.2017.09.002_sbref0015b
Gillis
J. Med. Chem.,
2015
13.

Murphy C.D., Sandford G.
Expert Opinion on Drug Metabolism and Toxicology,
2015
14.

Böhm H., Banner D., Bendels S., Kansy M., Kuhn B., Müller K., Obst-Sander U., Stahl M.
ChemBioChem,
2004
15.

Park B.K., Kitteringham N.R., O'Neill P.M.
Annual Review of Pharmacology and Toxicology,
2001
16.

Ford M.C., Ho P.S.
Journal of Medicinal Chemistry,
2015
17.

Pereira J.A., Pessoa A.M., Cordeiro M.N., Fernandes R., Prudêncio C., Noronha J.P., Vieira M.
European Journal of Medicinal Chemistry,
2015
18.

Ajani O.O.
European Journal of Medicinal Chemistry,
2014
19.
10.1016/j.mencom.2017.09.002_sbref0025c
Ren
Med. Chem.,
2014
20.

Bansal Y., Silakari O.
Bioorganic and Medicinal Chemistry,
2012
21.

Mikhailovskaya T.F., Makarov A.G., Selikhova N.Y., Makarov A.Y., Pritchina E.A., Bagryanskaya I.Y., Vorontsova E.V., Ivanov I.D., Tikhova V.D., Gritsan N.P., Slizhov Y.G., Zibarev A.V.
Journal of Fluorine Chemistry,
2016
22.

Makarov A.G., Selikhova N.Y., Makarov A.Y., Malkov V.S., Bagryanskaya I.Y., Gatilov Y.V., Knyazev A.S., Slizhov Y.G., Zibarev A.V.
Journal of Fluorine Chemistry,
2014
23.

Lee J., Jeong Y., Lee S., Kim D., Oh S., Lim H., Oh H., Kim S., Kim W., Jung J.
Cancer Letters,
2010
24.

Elmore S.A., Dixon D., Hailey J.R., Harada T., Herbert R.A., Maronpot R.R., Nolte T., Rehg J.E., Rittinghausen S., Rosol T.J., Satoh H., Vidal J.D., Willard-Mack C.L., Creasy D.M.
Toxicologic Pathology,
2016
25.

Elmore S.
Toxicologic Pathology,
2007
26.

27.

Allen F.H., Kennard O., Watson D.G., Brammer L., Orpen A.G., Taylor R.
Journal of the Chemical Society Perkin Transactions 2,
1987
28.

Solovieva A.O., Vorotnikov Y.A., Trifonova K.E., Efremova O.A., Krasilnikova A.A., Brylev K.A., Vorontsova E.V., Avrorov P.A., Shestopalova L.V., Poveshchenko A.F., Mironov Y.V., Shestopalov M.A.
Journal of Materials Chemistry B,
2016
29.

Heaton A., Hill M., Drakesmith F.
Journal of Fluorine Chemistry,
1997
30.

Brooke G.M., Burdon J., Tatlow J.C.
Journal of the Chemical Society (Resumed),
1961
31.
![Cyclic aryleneazachalcogenenes. Part III [1] synthesis of polyfluorinated 2,1,3-benzothia-(selena) diazoles](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Zibarev A.V., Miller A.O.
Journal of Fluorine Chemistry,
1990
32.

Burton D.E., Lambie A.J., Lane D.W., Newbold G.T., Percival A.
Journal of the Chemical Society C Organic,
1968
33.

Latosińska J.N., Latosińska M., Maurin J.K., Orzeszko A., Kazimierczuk Z.
Journal of Physical Chemistry A,
2014
34.
GraphPad Software, La Jolla, CA, USA (www.graphpad.com).
35.

Sheldrick G.M.
Acta crystallographica. Section C, Structural chemistry,
2015
36.
SADABS, Version 2008-1, Bruker AXS, Madison, WI, USA, 2008.