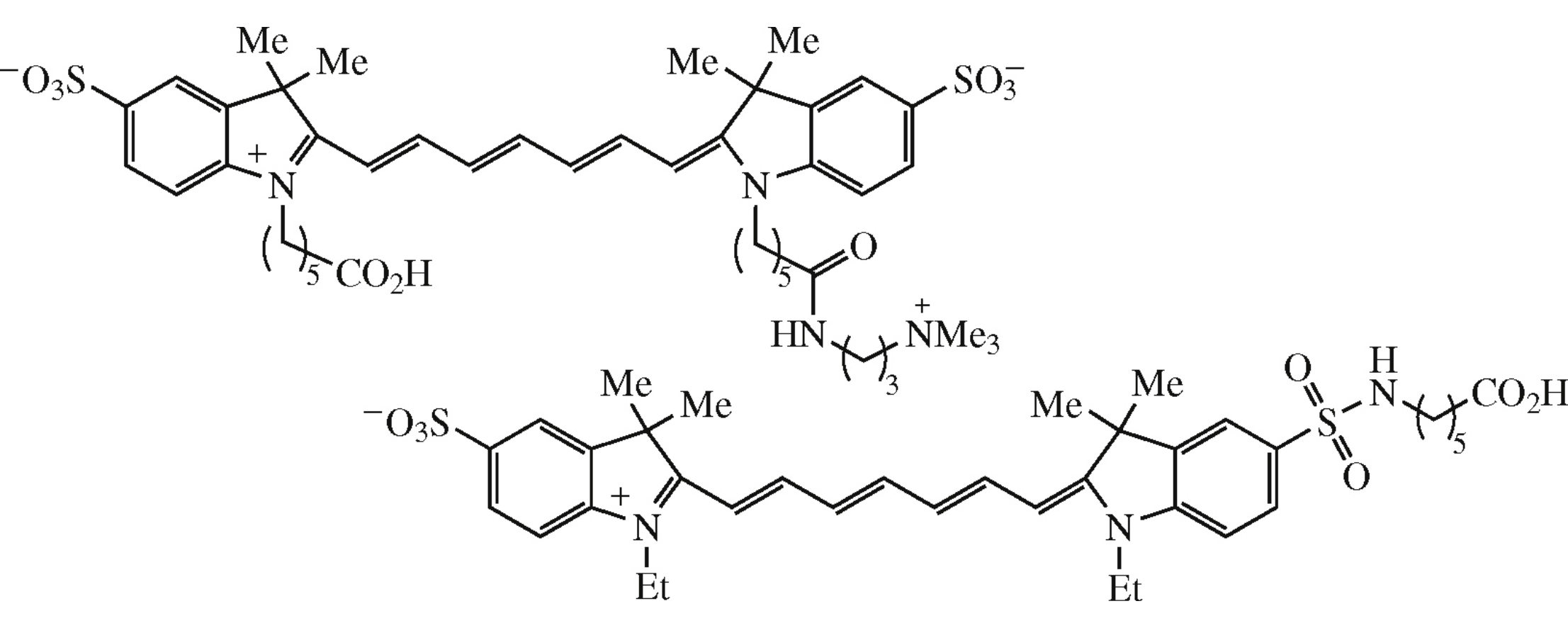
Abstract
Two novel electroneutral indotricarbocyanine dyes with two sulfo groups have been synthesized. They possess higher photo and thermal stability as compared to known analogue and were conjugated with aminoallyl-dUTP for enzymatic labeling of DNA by polymerase chain reaction.
References
1.

Nanjunda R., Owens E.A., Mickelson L., Alyabyev S., Kilpatrick N., Wang S., Henary M., Wilson W.D.
Bioorganic and Medicinal Chemistry,
2012
2.
10.1016/j.mencom.2017.07.013_bib0010
Mojzych
2008
3.

Camerin M., Jori G., Ciana L.D., Fabbroni S., Bonacchi S., Montalti M., Prodi L.
Photochemical and Photobiological Sciences,
2009
4.
Lim C., Shin J., Lee Y., Kim J., Oh K.S., Yuk S.H., Jeong S.Y., Kwon I.C., Kim S.
Theranostics,
2012
5.

Zhang S., Metelev V., Tabatadze D., Zamecnik P.C., Bogdanov A.
Proceedings of the National Academy of Sciences of the United States of America,
2008
6.
10.1016/j.mencom.2017.07.013_bib0030
Mojzych
2008
7.

Flanagan, J.H., Khan S.H., Menchen S., Soper S.A., Hammer R.P.
Bioconjugate Chemistry,
1997
8.
Pakhomov A.A., Kononevich Y.N., Korlyukov A.A., Martynov V.I., Muzafarov A.M.
Mendeleev Communications,
2016
9.

Bouit P., Rauh D., Neugebauer S., Delgado J.L., Piazza E.D., Rigaut S., Maury O., Andraud C., Dyakonov V., Martin N.
Organic Letters,
2009
10.
![Synthesis of water-soluble near-infrared cyanine dyes functionalized with [(succinimido)oxy]carbonyl group](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Strekowski L., Mason C.J., Lee H., Gupta R., Sowell J., Patonay G.
Journal of Heterocyclic Chemistry,
2003
11.

Steffens D.L., Jang G.Y., Sutter S.L., Brumbaugh J.A., Middendorf L.R., Mühlegger K., Mardis E.R., Weinstock L.A., Wilson R.K.
Genome Research,
1995
12.

Bertolino C.A., Caputo G., Barolo C., Viscardi G., Coluccia S.
Journal of Fluorescence,
2006
13.

Shershov V.E., Kuznetsova V.E., Lysov Y.P., Guseinov T.O., Barsky V.E., Spitsyn M.A., Zasedateleva O.A., Vasiliskov V.A., Surzhikov S.A., Zasedatelev A.S., Chudinov A.V.
Biophysics (Russian Federation),
2015
14.

Shershov V.E., Spitsyn M.A., Kuznetsova V.E., Timofeev E.N., Ivashkina O.A., Abramov I.S., Nasedkina T.V., Zasedatelev A.S., Chudinov A.V.
Dyes and Pigments,
2013
15.
Lebedeva N.S., Gubarev Y.A., Koifman O.I.
Mendeleev Communications,
2015
16.

González A.
Synthetic Communications,
1988
17.

Choi H.S., Nasr K., Alyabyev S., Feith D., Lee J.H., Kim S.H., Ashitate Y., Hyun H., Patonay G., Strekowski L., Henary M., Frangioni J.V.
Angewandte Chemie - International Edition,
2011
18.

Njiojob C.N., Owens E.A., Narayana L., Hyun H., Choi H.S., Henary M.
Journal of Medicinal Chemistry,
2015
19.
G. Caputo and C. L. Della, Patent EP 1491591 A1, 2004.
20.

Lin Y., Weissleder R., Tung C.
Bioconjugate Chemistry,
2002
21.

Strekowski L., Lee H., Mason J.C., Say M., Patonay G.
Journal of Heterocyclic Chemistry,
2007
22.

Song F., Peng X., Lu E., Zhang R., Chen X., Song B.
Journal of Photochemistry and Photobiology A: Chemistry,
2004
23.

Wang L., Fan J., Qiao X., Peng X., Dai B., Wang B., Sun S., Zhang L., Zhang Y.
Journal of Photochemistry and Photobiology A: Chemistry,
2010
24.

Gra O.A., Glotov A.S., Nikitin E.A., Glotov O.S., Kuznetsova V.E., Chudinov A.V., Sudarikov A.B., Nasedkina T.V.
American Journal of Hematology,
2008
25.
Kuznetsova V.E., Spitsyn M.A., Shershov V.E., Guseinov T.O., Fesenko E.E., Lapa S.A., Ikonnikova A.Y., Avdonina M.A., Nasedkina T.V., Zasedatelev A.S., Chudinov A.V.
Mendeleev Communications,
2016
26.

Gryadunov D., Mikhailovich V., Lapa S., Roudinskii N., Donnikov M., Pan'kov S., Markova O., Kuz'min A., Chernousova L., Skotnikova O., Moroz A., Zasedatelev A., Mirzabekov A.
Clinical Microbiology and Infection,
2005