Abstract
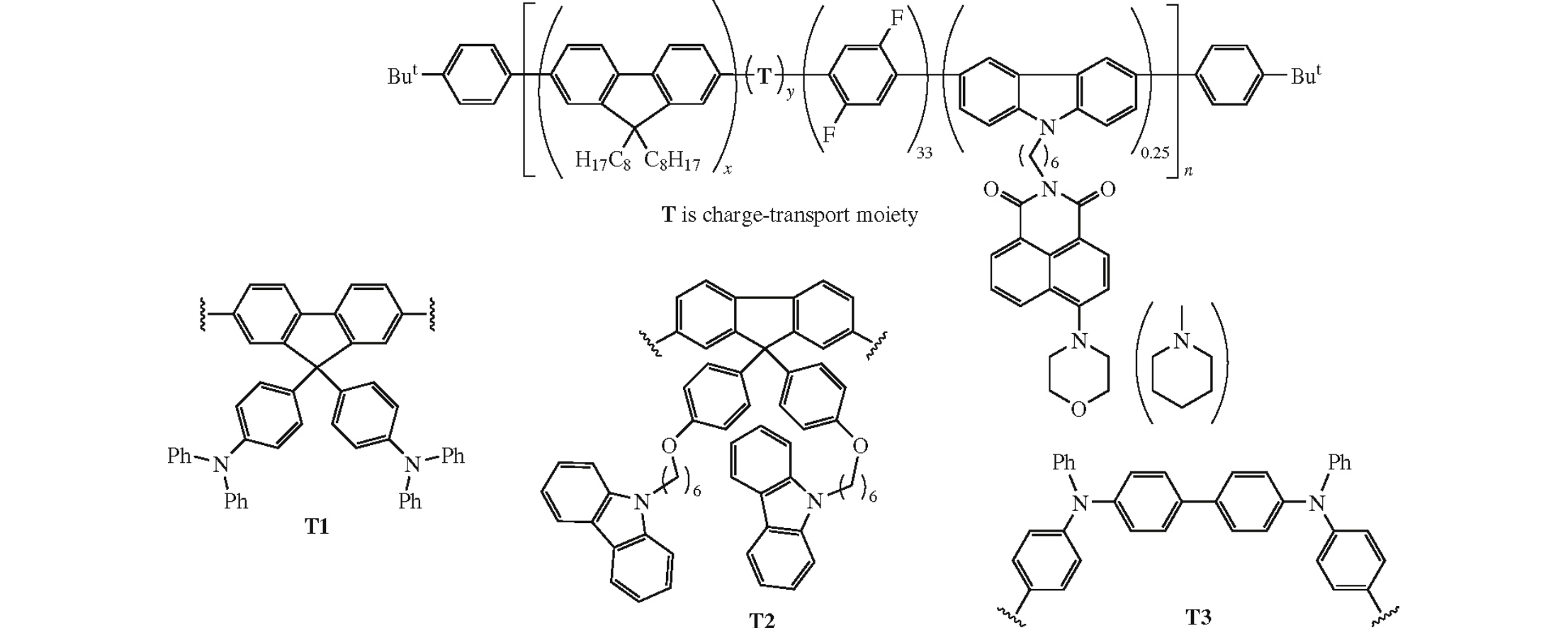
Polyfluorene copolymers containing 33 mol% of 2,5-difluoro-1,4-phenylene units and other comonomers comprising chargetransporting triphenylamine, carbazole, and 1,8-naphthalimide groups were synthesized by the Suzuki polycondensation under microwave irradiation. On heating the obtained copolymers to 180°C, their photoluminescence spectra remain stable. The best sample herein obtained provided the OLED brightness of 2830 cd m–2, current efficiency of 0.5 cd A–1 and CIE coordinates of x = 0.168 and y = 0.228.
References
1.
10.1016/j.mencom.2017.07.012_bib0005
Polyfluorenes,
2008
2.
10.1016/j.mencom.2017.07.012_bib0010
Kovalev
Russ. Chem. Rev.,
2014
3.

Kurdyukova I.V., Ishchenko A.A.
Russian Chemical Reviews,
2012
4.

Lee J., Hwang D.
Chemical Communications,
2003
5.

Wang P., Ho M., Yang S., Chen K., Hsu C.
Journal of Polymer Science, Part A: Polymer Chemistry,
2010
6.

Zhao Q., Liu S., Huang W.
Macromolecular Chemistry and Physics,
2009
7.

Poly(9,9-dialkyl-3,6-dibenzosilole)—a high energy gap host for phosphorescent light emitting devices
Chan K.L., Watkins S.E., Mak C.S., McKiernan M.J., Towns C.R., Pascu S.I., Holmes A.B.
Chemical Communications,
2005
8.

Wang E., Li C., Peng J., Cao Y.
Journal of Polymer Science, Part A: Polymer Chemistry,
2007
9.

Klärner G., Davey M.H., Chen W., Scott J.C., Miller R.D.
Advanced Materials,
1998
10.

Deep blue light-emitting polymers with fluorinated backbone for enhanced color purity and efficiency
Zhang T., Wang R., Ren H., Chen Z., Li J.
Polymer,
2012
11.

Giovanella U., Pasini M., Destri S., Porzio W., Botta C.
Synthetic Metals,
2008
12.

Shu C., Dodda R., Wu F., Liu M.S., Jen A.K.
Macromolecules,
2003
13.
Mróz W., Babushkina M.A., Kushakova N.S., Kovalev A.I., Antonova O.Y., Kaplin V.S., Pasini M., Galeotti F., Destri S., Giovanella U., Khotina I.A.
Mendeleev Communications,
2016
14.

Yuan M., Shih P., Chien C., Shu C.
Journal of Polymer Science, Part A: Polymer Chemistry,
2007
15.

Horii T., Shinnai T., Tsuchiya K., Mori T., Kijima M.
Journal of Polymer Science, Part A: Polymer Chemistry,
2012
16.

Chen X., Liao J., Liang Y., Ahmed M.O., Tseng H., Chen S.
Journal of the American Chemical Society,
2002
17.
Akkuratov A.V., Susarova D.K., Mukhacheva O.A., Troshin P.A.
Mendeleev Communications,
2016
18.

Ishii J., Sunaga T., Deguchi S., Tsukioka M.
High Performance Polymers,
2008
19.

Lin Y., Chen Z., Ye T., Dai Y., Ma D., Ma Z., Liu Q., Chen Y.
Polymer,
2010
20.

Lin Y., Ye T., Chen Y., Ma D., Chen Z., Dai Y., Li Y.
Journal of Polymer Science, Part A: Polymer Chemistry,
2010
21.

Liu M.S., Jiang X., Herguth P., Jen A.K.
Chemistry of Materials,
2001
22.

Ying L., Zou J., Yang W., Zhang A., Wu Z., Zhao W., Cao Y.
Dyes and Pigments,
2009
23.

Liu J., Cao J., Shao S., Xie Z., Cheng Y., Geng Y., Wang L., Jing X., Wang F.
Journal of Materials Chemistry A,
2008
24.

Zhang W., Lu P., Wang Z., Ma Y.
Journal of Polymer Science, Part A: Polymer Chemistry,
2013
25.

Nosova G.I., Lypenko D.A., Smyslov R.Y., Berezin I.A., Zhukova E.V., Mal’tsev E.I., Dmitriev A.V., Litvinova L.S., Solovskaya N.A., Dobrokhotov O.V., Abramov I.G., Yakimanskii A.V.
Polymer Science - Series B,
2014