Abstract
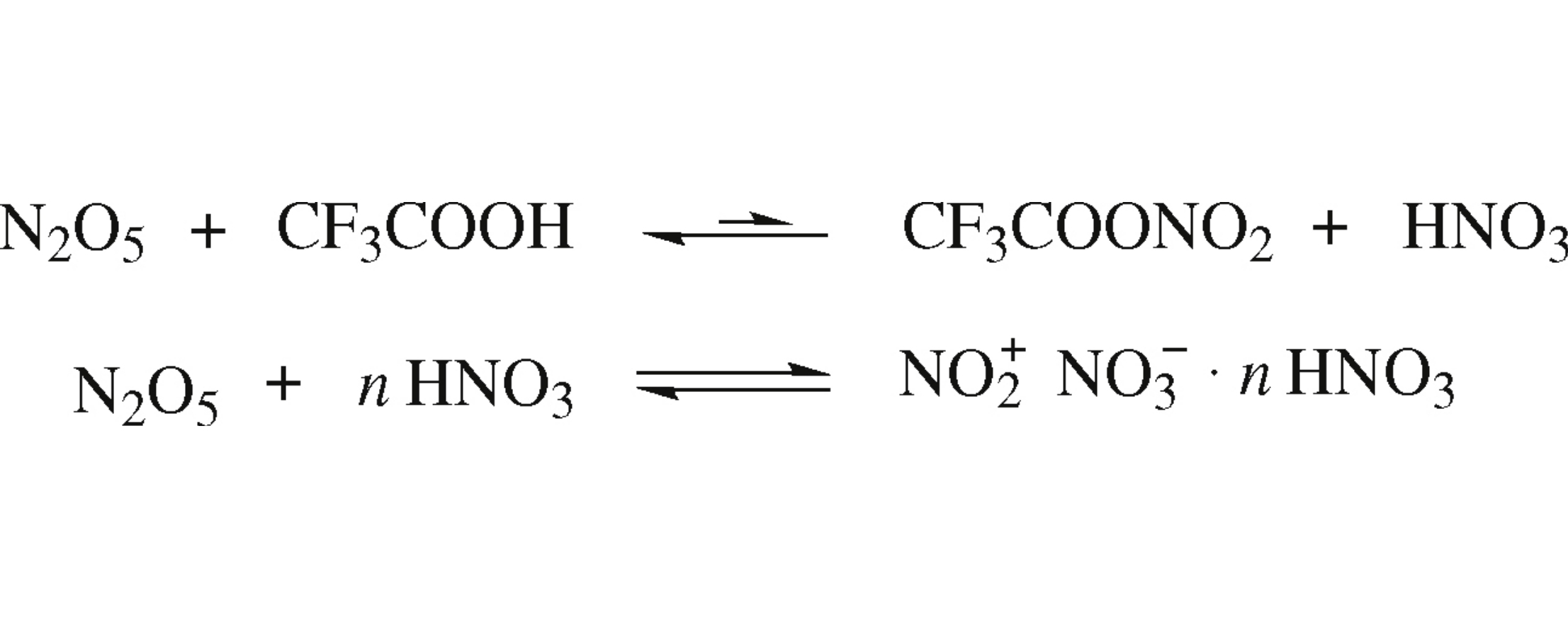
Dinitrogen pentoxide solution in trifluoroacetic acid simultaneously has features of both known types of its interaction with acids, viz. with weak and strong acids. It partially reacts with CF3COOH to give the covalent trifluoroacetyl nitrate and nitric acid, however the major part of N2O5 exists in the covalent form in solution to concurrently produce nitronium ions due to the presence of HNO3. Quantum chemical calculations confirm the equilibrium nature of CF3COOHN2O5 interaction.
References
1.
10.1016/j.mencom.2017.07.011_bib0005
Chemistry of Energetic Materials,
1991
2.
10.1016/j.mencom.2017.07.011_bib0010
Agrawal
Organic Chemistry of Explosives,
2010
3.

TALAWAR M., SIVABALAN R., POLKE B., NAIR U., GORE G., ASTHANA S.
Journal of Hazardous Materials,
2005
4.
10.1016/j.mencom.2017.07.011_bib0020
Hammerl
Encyclopedia of Inorganic and Bioinorganic Chemistry,
2011
5.

Addison C.C., Logan N.
Advances in Inorganic Chemistry and Radiochemistry,
1964
6.

Field B.O., Hardy C.J.
Quarterly Reviews Chemical Society,
1964
7.

Goddard D.R., Hughes E.D., Ingold C.K.
Journal of the Chemical Society (Resumed),
1950
8.

Wilson W.W., Christe K.O.
Inorganic Chemistry,
1987
9.

Hisatsune I.C., Devlin J.P., Wada Y.
Spectrochimica Acta,
1962
10.
10.1016/j.mencom.2017.07.011_bib0050
Hoggett
Nitration and Aromatic Reactivity,
1971
11.
Zelenov V.P., Bukalov S.S., Leites L.A., Aysin R.R., Subbotin A.N., Struchkova M.I., Fedyanin I.V.
Mendeleev Communications,
2017
12.

Olah G.A., Kuhn S.J., Flood S.H., Evans J.C.
Journal of the American Chemical Society,
1962
13.

Millen D.J.
Journal of the Chemical Society (Resumed),
1950
14.

Harrar J.E., Rigdon L.P., Rice S.F.
Journal of Raman Spectroscopy,
1997
15.
10.1016/j.mencom.2017.07.011_bib0075
Odokienko
Zh. Prikl. Khim.,
1978
16.

Olah G.A., Gupta B.G., Narang S.C.
Journal of the American Chemical Society,
1979
17.

Mason J., Christe K.O.
Inorganic Chemistry,
1983
18.

Klapötke T.M., McIntyre G., Schulz A.
Journal of the Chemical Society Dalton Transactions,
1996
19.

Ingold C.K., Millen D.J., Poole H.G.
Journal of the Chemical Society (Resumed),
1950
20.

Edwards H.G., Turner J.M., Fawcett V.
Journal of the Chemical Society Faraday Transactions,
1995
21.

Harrar J.E., Pearson R.K.
Journal of the Electrochemical Society,
1983
22.

Ingold C.K., Millen D.J.
Journal of the Chemical Society (Resumed),
1950
23.
10.1016/j.mencom.2017.07.011_bib0115
Lagodzinskaya
Khim. Fiz.,
1989
24.

Kuhn S.J., Olah G.A.
Journal of the American Chemical Society,
1961