Abstract
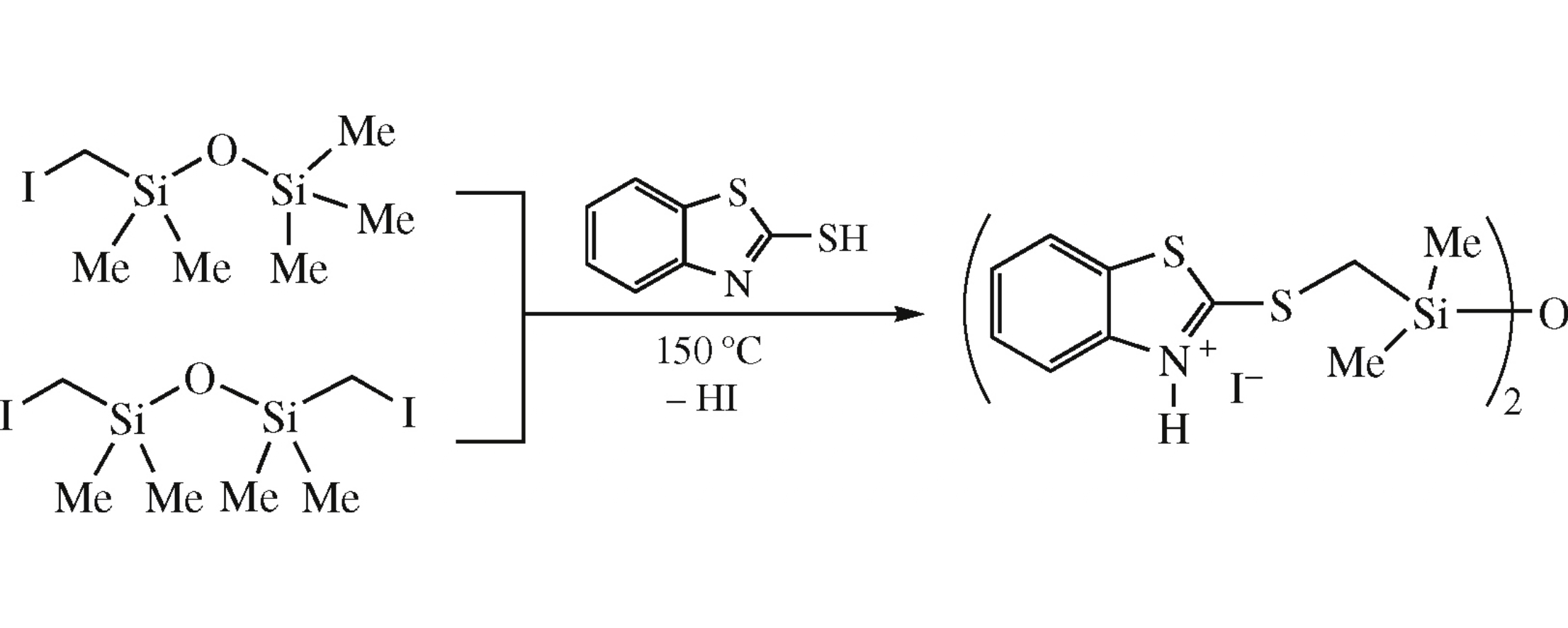
First organosilicon captax derivatives were obtained from 2-mercaptobenzothiazole and 1-(iodomethyl)-1,1,3,3,3-pentamethyl-or 1,3-bis(iodomethyl)-1,1,3,3-tetramethyldisiloxanes in the absence or in the presence of bases.
References
1.

Herrera Cano N., Ballari M.S., López A.G., Santiago A.N.
Journal of Agricultural and Food Chemistry,
2015
2.

Azam M.A.
Scientia Pharmaceutica,
2012
3.

Wang F., Cai S., Wang Z., Xi C.
Organic Letters,
2011
4.

Chen W., Huang Y., Gundala S.R., Yang H., Li M., Tai P.C., Wang B.
Bioorganic and Medicinal Chemistry,
2010
5.

Jiang L., Tan Y., Zhu X., Wang Z., Zuo Y., Chen Q., Xi Z., Yang G.
Journal of Agricultural and Food Chemistry,
2009
6.

Synthesis, antioxidant properties and radioprotective effects of new benzothiazoles and thiadiazoles
Cressier D., Prouillac C., Hernandez P., Amourette C., Diserbo M., Lion C., Rima G.
Bioorganic and Medicinal Chemistry,
2009
7.

Franchini C., Muraglia M., Corbo F., Florio M.A., Di Mola A., Rosato A., Matucci R., Nesi M., van Bambeke F., Vitali C.
Archiv der Pharmazie,
2009
8.

Huang W., Yang G.
Bioorganic and Medicinal Chemistry,
2006
9.

Ramadas K., Janarthanan N.
Synthetic Communications,
1999
10.

Katkova M.A., Borisov A.V., Fukin G.K., Baranov E.V., Averyushkin A.S., Vitukhnovsky A.G., Bochkarev M.N.
Inorganica Chimica Acta,
2006
11.

Mishra L.
Journal of Inorganic Biochemistry,
2001
12.

Zdrahala R.J., Zdrahala I.J.
Journal of Biomaterials Applications,
1999
13.

Bernacca G.M., Straub I., Wheatley D.J.
Journal of Biomedical Materials Research,
2002
14.
10.1016/j.mencom.2017.07.010_sbref0020c
Hyde
J. Heart Valve Dis.,
1999
15.
10.1016/j.mencom.2017.07.010_sbref0020d
Voronkov
Kremnii i zhizn (Silicon and Life),
1978
16.
10.1016/j.mencom.2017.07.010_sbref0020e
Hufnagel
Med. Instrum.,
1977
17.

Hu Y., Chen Z., Le Z., Zheng Q.
Synthetic Communications,
2004
18.

Deligeorgiev T., Kaloyanova S., Lesev N., Vaquero J.J.
Ultrasonics Sonochemistry,
2010
19.

Duarte A., Cunico W., Pereira C.M., Flores A.F., Freitag R.A., Siqueira G.M.
Ultrasonics Sonochemistry,
2010
20.

Narkhede H.P., More U.B., Dalal D.S., Pawar N.S., More D.H., Mahulikar P.P.
Synthetic Communications,
2007
21.

Nishio T., Shiwa K.
Heterocycles,
2004
22.

Wu F., M. Hussein W., P. Ross B., P. McGeary R.
Current Organic Chemistry,
2012
23.

Zhilitskaya L.V., Yarosh N.O., Shagun L.G., Dorofeev I.A., Larina L.I.
Chemistry of Heterocyclic Compounds,
2015
24.
Shagun L.G., Dorofeev I.A., Zhilitskaya L.V., Larina L.I., Yarosh N.O., Klyba L.V., Sanzheeva E.R.
Mendeleev Communications,
2015
25.
Yarosh N.O., Zhilitskaya L.V., Shagun L.G., Dorofeev I.A., Larina L.I., Klyba L.V.
Mendeleev Communications,
2016
26.
10.1016/j.mencom.2017.07.010_bib0060
Voronkov
Siloksanovaya svyaz (Siloxane Bond),
1976