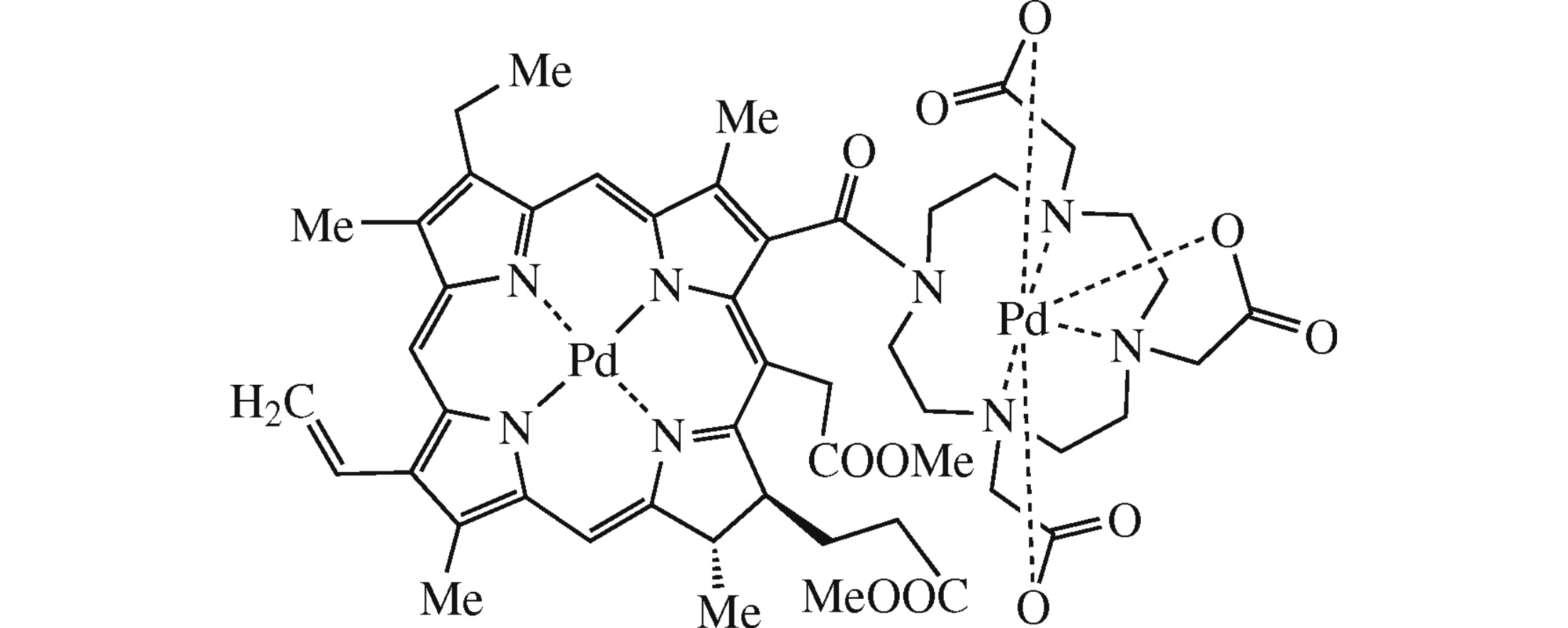
Abstract
Novel herein synthesized conjugate of chlorin e6 methyl ester with cyclen, as well as its mono- and dipalladium complexes, are regarded in view of their use in therapy and diagnostics.
References
1.
Yakushev A.A., Averin A.D., Maloshitskaya O.A., Syrbu S.A., Koifman O.I., Beletskaya I.P.
Mendeleev Communications,
2016
2.

Dobrev H.
Photodermatology Photoimmunology and Photomedicine,
2010
3.

Mironov A.F.
Russian Chemical Reviews,
2013
4.
10.1016/j.mencom.2017.07.005_bib0020
Rumyantseva
Vestnik MITKhT,
2014
5.

Eggenspiller A., Michelin C., Desbois N., Richard P., Barbe J., Denat F., Licona C., Gaiddon C., Sayeh A., Choquet P., Gros C.P.
European Journal of Organic Chemistry,
2013
6.

Luo J., Chen L., Hu P., Chen Z.
Inorganic Chemistry,
2014
7.

Caravan P., Ellison J.J., McMurry T.J., Lauffer R.B.
Chemical Reviews,
1999
8.

Livramento J.B., Tóth É., Sour A., Borel A., Merbach A.E., Ruloff R.
Angewandte Chemie - International Edition,
2004
9.

CHAN K., WONG W.
Coordination Chemistry Reviews,
2007
10.

Werner E., Datta A., Jocher C., Raymond K.
Angewandte Chemie - International Edition,
2008
11.

Song Y., Kohlmeir E.K., Meade T.J.
Journal of the American Chemical Society,
2008
12.

Que E.L., Chang C.J.
Chemical Society Reviews,
2010
13.

Hindré F., Plouzennec M.L., de Certaines J.D., Foultier M.T., Patrice T., Simonneaux G.
Journal of Magnetic Resonance Imaging,
1993
14.

Song Y., Zong H., Trivedi E.R., Vesper B.J., Waters E.A., Barrett A.G., Radosevich J.A., Hoffman B.M., Meade T.J.
Bioconjugate Chemistry,
2010
15.

Goswami L.N., White W.H., Spernyak J.A., Ethirajan M., Chen Y., Missert J.R., Morgan J., Mazurchuk R., Pandey R.K.
Bioconjugate Chemistry,
2010
16.
Lebedeva V.S., Karmova F.M., Toukach F.V., Mironov A.F.
Mendeleev Communications,
2015
17.
Lebedeva V.S., Karmova F.M., Mironov A.F.
Mendeleev Communications,
2015
18.

Dudkin S.V., Makarova E.A., Lukyanets E.A.
Russian Chemical Reviews,
2016
19.

Rumyantseva V.D., Ivanovskaya N.P., Konovalenko L.I., Tsukanov S.V., Mironov A.F., Osin N.S.
Russian Journal of Bioorganic Chemistry,
2008
20.
A. F. Mironov, Ross. Khim. Zh. (Zh. Ross. Khim. Ob-va im. D. I. Mendeleeva), 2006, no. 4, 61.(in Russian).
21.

Brandis A.S., Kozyrev A.N., Mironov A.F.
Tetrahedron,
1992
22.

Ramalingam S., Karabacak M., Periandy S., Puviarasan N., Tanuja D.
Spectrochimica Acta - Part A: Molecular and Biomolecular Spectroscopy,
2012
23.
A. F. Mironov, R.I. Reshetnikov, M.A. Grin, R.E. Yakubovskaya, E.A. Plotnikova, N.B. Morozova, A.A. Tsygankov, A.V. Feofanov, D.E. Ermakova and A.V. Efremenko, Patent RU 2548675C2, 2015.
24.

Kim J., Kim D., Kang M., Kim H., Kim J., Yu E., Jeong J.
Archives of Pharmacal Research,
2006
25.

Obata M., Hirohara S., Tanaka R., Kinoshita I., Ohkubo K., Fukuzumi S., Tanihara M., Yano S.
Journal of Medicinal Chemistry,
2009
26.
A. Sherts, I. Salomon, A. Brendis and Kh. Sheer, Patent RU 2234508, 2004.
27.

Borisov S.M., Papkovsky D.B., Ponomarev G.V., DeToma A.S., Saf R., Klimant I.
Journal of Photochemistry and Photobiology A: Chemistry,
2009
28.
10.1016/j.mencom.2017.07.005_bib0100
Zamilatskov
Makrogeterotsikly /Macro-heterocycles,
2012
29.
10.1016/j.mencom.2017.07.005_bib0105
Li
J. Org. Chem.,
2003
30.

Cakić N., Gündüz S., Rengarasu R., Angelovski G.
Tetrahedron Letters,
2015