Abstract
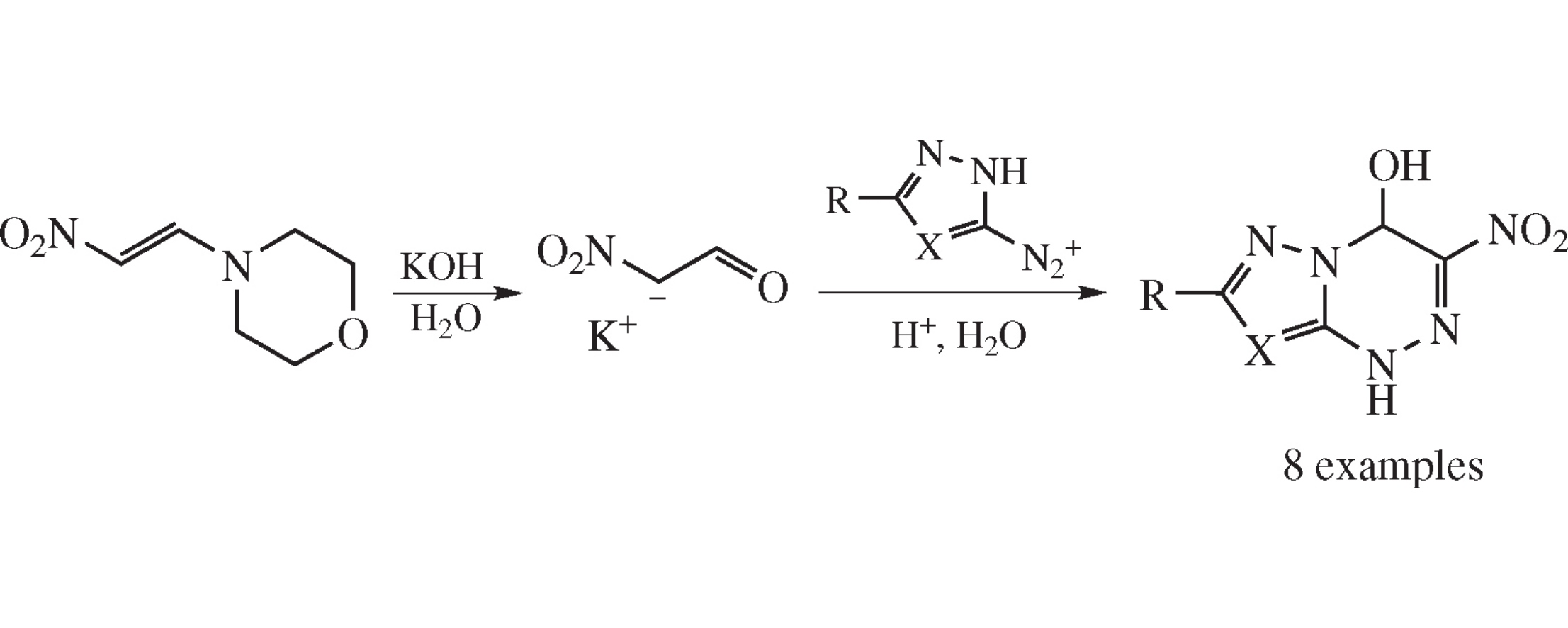
3-Nitro-4-hydroxy-1,4-dihydroazolo[5,1-c][1,2,4]triazines were obtained using nitroacetaldehyde potassium salt generated in situ from 1-morpholino-2-nitroethene.
References
1.

Rusinonv V.L., Petrov A.Y., Aleksandrov G.G., Chupakhin O.N.
Chemistry of Heterocyclic Compounds,
1994
2.

Ma X., Qiu N., Yang B., He Q., Hu Y.
MedChemComm,
2016
3.
D. Qian, A.Q. Han, M. Hamilton and E. Wang, Patent WO 2009/155527, 2009. (Chem. Abstr., 2009, 152, 97479).
4.

Denmark S.E., Thorarensen A., Middleton D.S.
Journal of Organic Chemistry,
1995
5.
![Tandem [4 + 2]/[3 + 2] Cycloadditions of Nitroalkenes. 9. Synthesis of (−)-Rosmarinecine](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Denmark S.E., Thorarensen A., Middleton D.S.
Journal of the American Chemical Society,
1996
6.
![Tandem [4 + 2]/[3 + 2] Cycloadditions of Nitroalkenes. 13. The Synthesis of (−)-Detoxinine](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Denmark S.E., Hurd A.R., Sacha H.J.
Journal of Organic Chemistry,
1997
7.

Denmark S.E., Martinborough E.A.
Journal of the American Chemical Society,
1999
8.

Denmark S.E., Cottell J.J.
Journal of Organic Chemistry,
2001
9.
![Tandem Inter [4+2]/Intra [3+2] Cycloadditions of Nitroalkenes. Application to the Synthesis of Aminocarbasugars](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Denmark S., Juhl M.
Helvetica Chimica Acta,
2002
10.
Zlotin S.G., Churakov A.M., Luk’yanov O.A., Makhova N.N., Sukhorukov A.Y., Tartakovsky V.A.
Mendeleev Communications,
2015
11.

Severin T., Böhme H.
Chemische Berichte,
1968
12.
Meerwein H., Florian W., Schön N., Stopp G.
1961
13.

Babievskii K.K., Belikov V.M., Tikhonova N.A.
Russian Chemical Bulletin,
1970
14.

HURD C.D., SHERWOOD, Jr L.T.
Journal of Organic Chemistry,
1948
15.

Faulques M., Rene L., Royer R.
Synthesis,
1982
16.
10.1016/j.mencom.2017.05.023_bib0080
Ono
Sci. Synth.,
2007
17.

Gate E.N., Threadgill M.D., Stevens M.F., Chubb D., Vickers L.M., Langdon S.P., Hickman J.A., Gescher A.
Journal of Medicinal Chemistry,
1986
18.

Dilman A., Kondratyev N., Zemtsov A., Levin V., Struchkova M.
Synthesis,
2012
19.
![Azolo[5,1-c]-1,2,4-triazines as a new class of antiviral compounds](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Rusinov V.L., Ulomskii E.N., Chupakhin O.N., Charushin V.N.
Russian Chemical Bulletin,
2008
20.
Voinkov E.K., Ulomskiy E.N., Rusinov V.L., Savateev K.V., Fedotov V.V., Gorbunov E.B., Isenov M.L., Eltsov O.S.
Mendeleev Communications,
2016
21.

Rusinov V.L., Pilicheva T.L., Chupakhin O.N., Klyuev N.A., Allakhverdieva D.T.
Chemistry of Heterocyclic Compounds,
1986