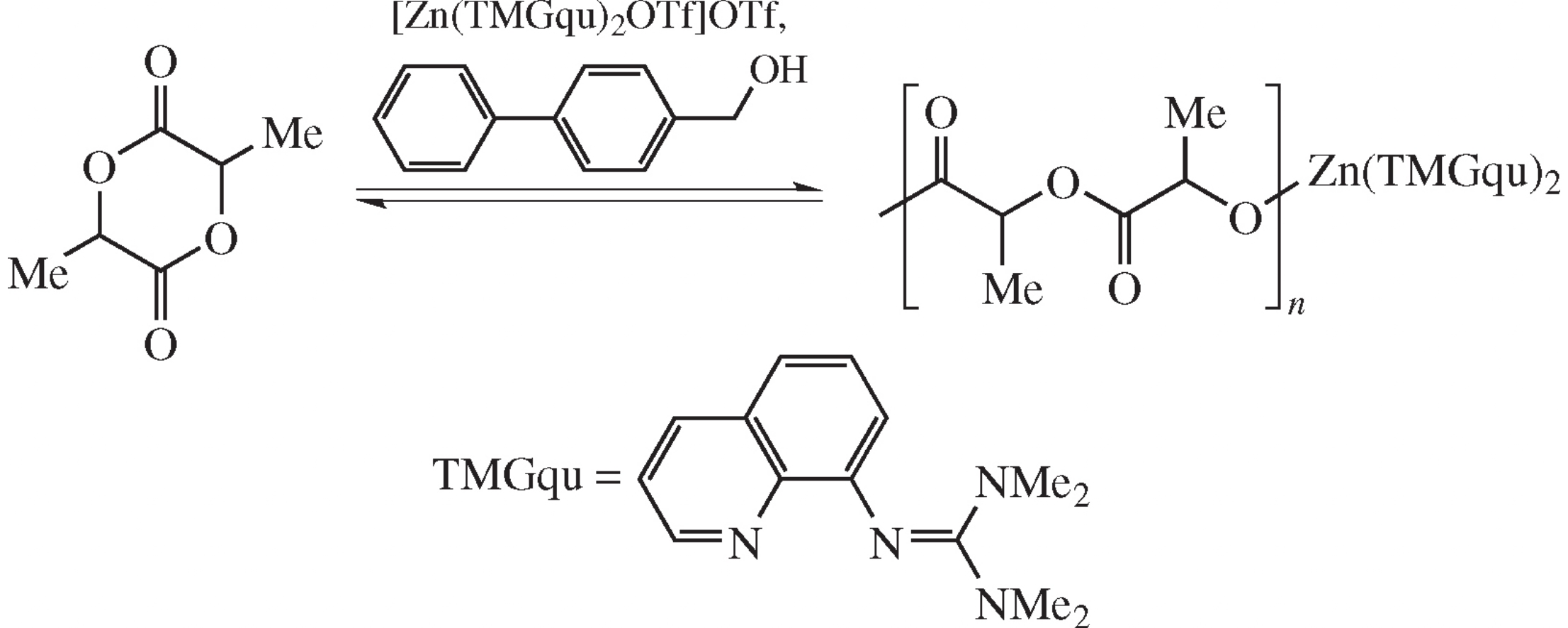
Abstract
A combination of Zn–guanidine complex catalyst and biphenyl-4-methanol activator was used as a biofriendly system for the synthesis of poly(d,l-lactide). Kinetic curves of the polymerization were measured by differential scanning calorimetry at various temperatures and concentrations of the activator. Due to an inclusion of guanidine ligands into the polymer chains, the synthesized polylactide is capable of fluorescing.
References
1.

Middleton J.C., Tipton A.J.
Biomaterials,
2000
2.

Farah S., Anderson D.G., Langer R.
Advanced Drug Delivery Reviews,
2016
3.

Peer D., Karp J.M., Hong S., Farokhzad O.C., Margalit R., Langer R.
Nature Nanotechnology,
2007
4.
Rogovina S.Z., Aleksanyan K.V., Grachev A.V., Berlin A.A., Prut E.V.
Mendeleev Communications,
2015
5.

Börner J., Flörke U., Huber K., Döring A., Kuckling D., Herres-Pawlis S.
Chemistry - A European Journal,
2009
6.
10.1016/j.mencom.2017.05.021_bib0030
Kreiser-Saunders
Macromol. Chem. Phys.,
1998
7.

Synthesis of polylactides in the presence of co-initiators with different numbers of hydroxyl groups
Korhonen H., Helminen A., Seppälä J.V.
Polymer,
2001
8.

Campos J.M., Ribeiro M.R., Ribeiro M.F., Deffieux A., Peruch F.
Macromolecular Chemistry and Physics,
2012
9.

Kinetics and thermodynamics of l-lactide polymerization studied by differential scanning calorimetry
Sedush N.G., Chvalun S.N.
European Polymer Journal,
2015
10.

Sedush N.G., Strelkov Y.Y., Chvalun S.N.
Polymer Science - Series B,
2014
11.

Kulagina T.G., Lebedev B.V., Kiparisova Y.G., Lyudvig Y.B., Barskaya I.G.
Polymer Science U S S R,
1982
12.

Börner J., dos Santos Vieira I., Pawlis A., Döring A., Kuckling D., Herres‐Pawlis S.
Chemistry - A European Journal,
2011