Abstract
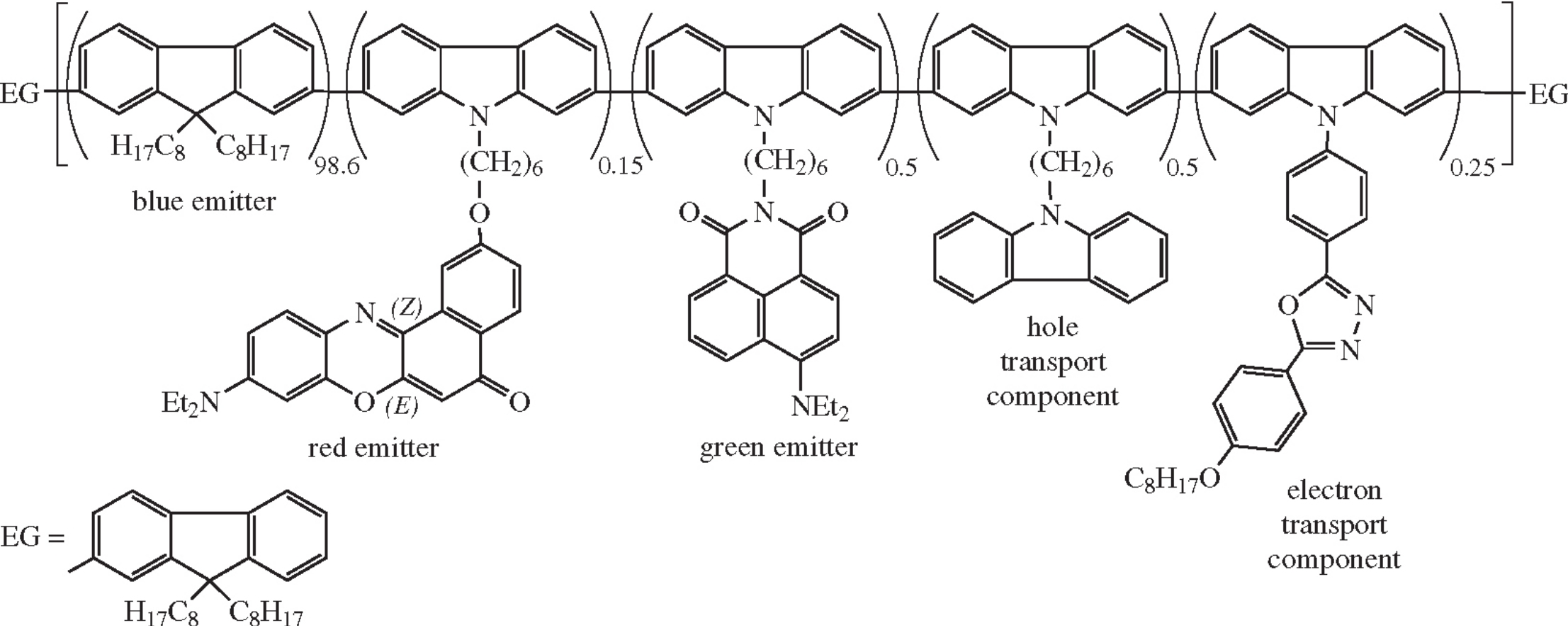
Seven polyfluorenes containing new carbazole derivatives of Nile Red dye and 1,8-naphthalimide in the chain (obtained by the Suzuki and Yamamoto coupling) are characterized by the Nile Red emission band being less intense in their electroluminescence spectra than in photoluminescence spectra. In the optimal composition, 0.15 mol% fraction of Nile Red and 0.5 mol% fraction of N-[6-(2,7-dibromocarbazol-9-yl)hexyl]-4-diethylamino-1,8-naphthalimide provide a nearly white light electroluminescence (CIE coordinates x = 0.28; y = 0.36) with the brightness of 3278 cd m–2 and the current efficiency of 1.86 cd A–1.
References
1.
10.1016/j.mencom.2017.05.016_bib0005
Polyfluorenes,
2008
2.

Günes S., Neugebauer H., Sariciftci N.S.
Chemical Reviews,
2007
3.

Kurdyukova I.V., Ishchenko A.A.
Russian Chemical Reviews,
2012
4.

Bao B., Yuwen L., Zheng X., Weng L., Zhu X., Zhan X., Wang L.
Journal of Materials Chemistry A,
2010
5.

Dong S., Ou D., Qin J., Li Z.
Journal of Polymer Science, Part A: Polymer Chemistry,
2011
6.
Kuznetsov I.E., Susarova D.K., Inasaridze L.N., Klyuev M.V., Troshin P.A.
Mendeleev Communications,
2015
7.
Dmitriev A.V., Yusupov A.R., Irgashev R.A., Kazin N.A., Mal’tsev E.I., Lypenko D.A., Rusinov G.L., Vannikov A.V., Charushin V.N.
Mendeleev Communications,
2016
8.
Akkuratov A.V., Susarova D.K., Mukhacheva O.A., Troshin P.A.
Mendeleev Communications,
2016
9.

Jose J., Burgess K.
Journal of Organic Chemistry,
2006
10.

Tu G.L., Mei C.Y., Zhou Q.G., Cheng Y.X., Geng Y.H., Wang L.X., Ma D.G., Jing X.B., Wang F.S.
Advanced Functional Materials,
2006
11.

Liu J., Shao S. ., Chen L., Xie Z. ., Cheng Y. ., Geng Y. ., Wang L. ., Jing X. ., Wang F. .
Advanced Materials,
2007
12.

Sreearunothai P., Estrada A., Asaoka S., Kowalczyk M., Jang S., Cook A.R., Preses J.M., Miller J.R.
Journal of Physical Chemistry C,
2011
13.

Nosova G.I., Lypenko D.A., Smyslov R.Y., Berezin I.A., Zhukova E.V., Mal’tsev E.I., Dmitriev A.V., Litvinova L.S., Solovskaya N.A., Dobrokhotov O.V., Abramov I.G., Yakimanskii A.V.
Polymer Science - Series B,
2014
14.
Arkhipova A.N., Panchenko P.A., Fedorov Y.V., Fedorova O.A.
Mendeleev Communications,
2017
15.
![Hole formation and transfer in poly[9,9-di(ethylhexyl)fluorene] and an amine end-capped derivative in solution](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Burrows H.D., Seixas de Melo J., Forster M., Güntner R., Scherf U., Monkman A.P., Navaratnam S.
Chemical Physics Letters,
2004
16.

Yevlampieva N.P., Khurchak A.P., Nosova G.I., Smyslov R.Y., Berezin I.A., Ilgach D.M., Kopylova T.N., Gadirov R.M., Yakimansky A.V.
Chemical Physics Letters,
2016