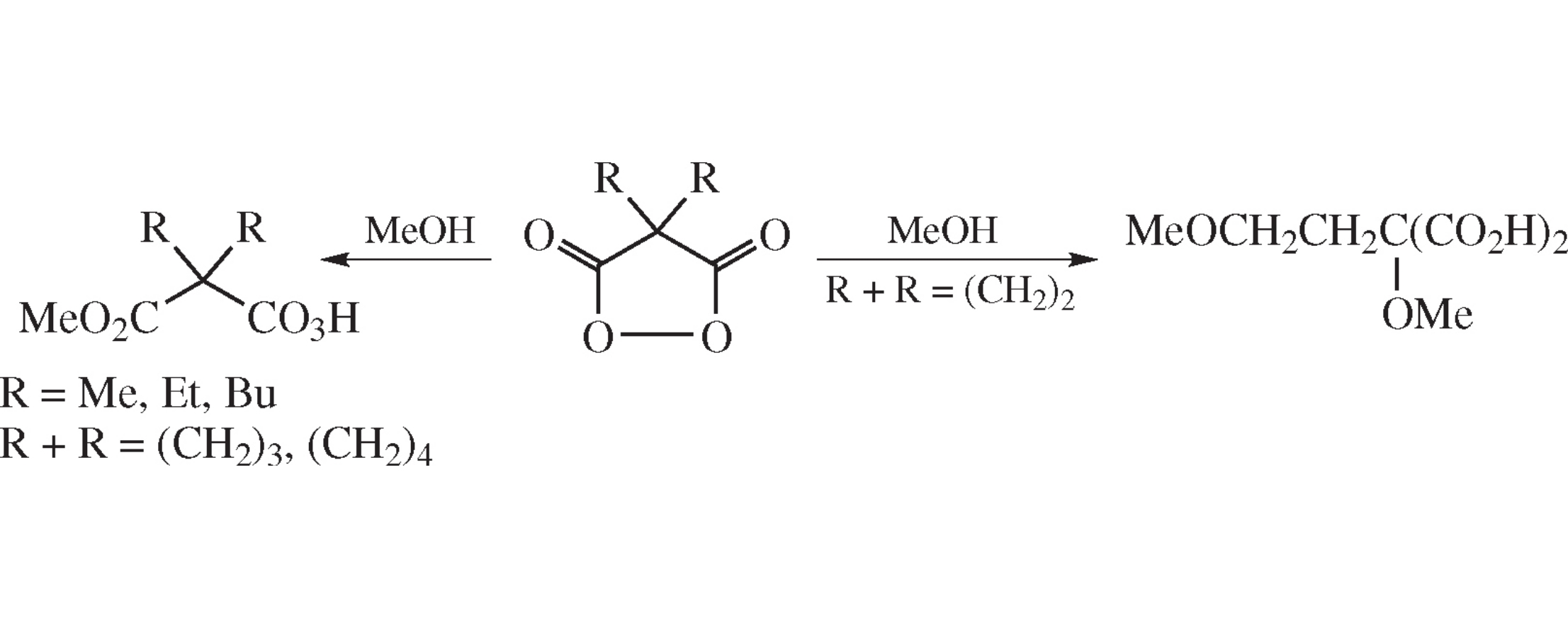
Abstract
The spontaneous reaction of disubstituted malonyl peroxides (MPOs) with methanol affording monopermalonic acid monomethyl esters is fast (minutes) for lower homologues but is sharply decelerated (days) for the higher ones. Spirocyclopropyl-MPO is an exception in which the nucleophilic opening of the spiroactivated cyclopropane ring leads to 2,4-dimethoxy-2-carboxybutanoic acid.
References
1.

Adam W., Rucktaeschel R.
Journal of the American Chemical Society,
1971
2.

Chapman O.L., Wojtkowski P.W., Adam W., Rodriguez O., Rucktaeschel R.
Journal of the American Chemical Society,
1972
3.

Adam W., Diehl J.W.
Journal of the Chemical Society Chemical Communications,
1972
4.

Adam W., Liu J., Rodriguez O.
Journal of Organic Chemistry,
1973
5.

Martin M.M., Hammer F.T., Zador E.
Journal of Organic Chemistry,
1973
6.

Photolysis and thermolysis of di-n-butylmalonyl peroxide. Evidence for .alpha.-lactone intermediates
Adam W., Rucktaeschel R.
Journal of Organic Chemistry,
1978
7.

Adam W., Cadiz C., Mazenod F.
Tetrahedron Letters,
1981
8.

Darmon M.J., Schuster G.B.
Journal of Organic Chemistry,
1982
9.

Porter J.E., Schuster G.B.
Journal of Organic Chemistry,
1983
10.

Porter J.E., Schuster G.B.
Journal of Organic Chemistry,
1985
11.

Griffith J.C., Jones K.M., Picon S., Rawling M.J., Kariuki B.M., Campbell M., Tomkinson N.C.
Journal of the American Chemical Society,
2010
12.

Schwarz M., Reiser O.
Angewandte Chemie - International Edition,
2011
13.

Picon S., Rawling M., Campbell M., Tomkinson N.C.
Organic Letters,
2012
14.

Jones K.M., Tomkinson N.C.
Journal of Organic Chemistry,
2012
15.

Rawling M.J., Tomkinson N.C.
Organic and Biomolecular Chemistry,
2013
16.

Rawling M.J., Rowley J.H., Campbell M., Kennedy A.R., Parkinson J.A., Tomkinson N.C.
Chemical Science,
2014
17.

Alamillo-Ferrer C., Davidson S.C., Rawling M.J., Theodoulou N.H., Campbell M., Humphreys P.G., Kennedy A.R., Tomkinson N.C.
Organic Letters,
2015
18.

Alamillo-Ferrer C., Karabourniotis-Sotti M., Kennedy A.R., Campbell M., Tomkinson N.C.
Organic Letters,
2016
19.

Dragan A., Kubczyk T.M., Rowley J.H., Sproules S., Tomkinson N.C.
Organic Letters,
2015
20.

Terent’ev A., Vil’ V., Nikishin G., Adam W.
Synlett,
2015
21.

Terent’ev A.O., Vil’ V.A., Gorlov E.S., Nikishin G.I., Pivnitsky K.K., Adam W.
Journal of Organic Chemistry,
2016
22.

Adam W., Rucktaeschel R.
Journal of Organic Chemistry,
1972
23.
Lapitskaya M.A., Vil’ V.A., Daeva E.D., Terent’ev A.O., Pivnitsky K.K.
Mendeleev Communications,
2016
24.
Terent’ev A.O., Vil’ V.A., Mulina O.M., Pivnitsky K.K., Nikishin G.I.
Mendeleev Communications,
2014
25.
Adam W., Epe B., Schiffmann D., Vargas F., Wild D.
1988
26.

Adam W., Hückmann S., Vargas F.
Tetrahedron Letters,
1989
27.
K. M. Jones, PhD Thesis, Cardiff University, 2010.
28.

Laszlo P.
Progress in Nuclear Magnetic Resonance Spectroscopy,
1967
29.
10.1016/j.mencom.2017.05.008_sbref0065b
Ronayne
1969
30.

Williams D.H., Wilson D.A.
Journal of the Chemical Society B Physical Organic,
1966
31.

Edgar Anderson J.
Tetrahedron Letters,
1965
32.

Danishefsky S., Singh R.K.
Journal of Organic Chemistry,
1975
33.

Danishefsky S., Singh R.K.
Journal of the American Chemical Society,
1975
34.

Danishefsky S.
Accounts of Chemical Research,
1979