Abstract
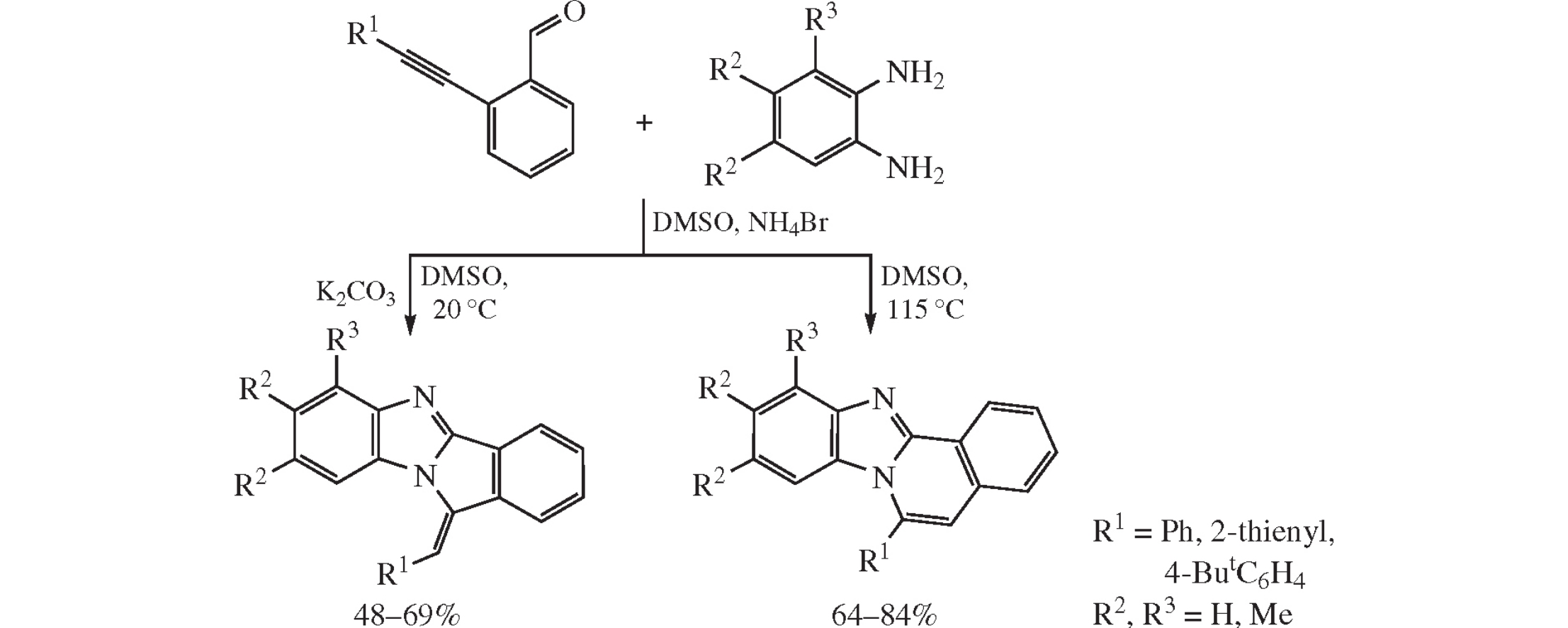
Cyclization of o-alkynylbenzaldehydes with o-diaminobenzenes in DMSO under the sequential action of NH4Br and K2CO3 affords 11-arylmethylidene-11H-isoindolo[2,1-a]-benzimidazoles as a result of 5-exo-dig ring closure; whereas replacement of treating with K2CO3 by heating at 110–115°C results in 6-endo-dig cyclization with formation of 6-arylbenzimidazo[2,1-a]isoquinolines.
References
1.

Takao K., Munakata R., Tadano K.
Chemical Reviews,
2005
2.

Mieczkowski A., Roy V., Agrofoglio L.A.
Chemical Reviews,
2010
3.

Rida S.M., El-Hawash S.A., Fahmy H.T., Hazzaa A.A., El-Meligy M.M.
Archives of Pharmacal Research,
2006
4.
10.1016/j.mencom.2017.05.004_sbref0010b
Deady
Anti-Cancer Drug Des.,
2000
5.
10.1016/j.mencom.2017.05.004_sbref0010c
Pandey
Indian J. Chem., Sect. B,
1999
6.

Zhe Xiong L., Nurul Huda Q., Leslie W. D., Gideon M. P.
Biological Chemistry Hoppe-Seyler,
1996
7.
![Antineoplastic activity of benzimidazo[1,2-b]-isoquinolines, indolo[2,3-b]quinolines, and pyridocarbazoles](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Weinkauf R.L., Chen A.Y., Yu C., Liu L., Barrows L., LaVoie E.J.
Bioorganic and Medicinal Chemistry,
1994
8.

Dyker G., Stirner W., Henkel G.
European Journal of Organic Chemistry,
2000
9.
![One-pot concise syntheses of benzimidazo[2,1-a]isoquinolines by a microwave-accelerated tandem process](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Okamoto N., Sakurai K., Ishikura M., Takeda K., Yanada R.
Tetrahedron Letters,
2009
10.

Ouyang H., Tang R., Zhong P., Zhang X., Li J.
Journal of Organic Chemistry,
2010
11.

Rustagi V., Aggarwal T., Verma A.K.
Green Chemistry,
2011
12.
![Copper-Catalyzed Cascade Synthesis of Alkyl 6-Aminobenzimidazo[2,1-a]isoquinoline-5-carboxylates](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Lu J., Fu H.
Journal of Organic Chemistry,
2011
13.

Lu G., Cai C.
Advanced Synthesis and Catalysis,
2013
14.

Zhao X.Z., Maddali K., Marchand C., Pommier Y., Burke T.R.
Bioorganic and Medicinal Chemistry,
2009
15.

Pandey S., Shyam Srivastava R.
Letters in Drug Design and Discovery,
2010
16.

Dydio P., Lichosyt D., Jurczak J.
Chemical Society Reviews,
2011
17.

Arulmurugan S., P. Kavitha H., Sathishkumar S., Arulmozhi R.
Mini-Reviews in Organic Chemistry,
2015
18.
Shavrin K.N., Gvozdev V.D., Nefedov O.M.
Mendeleev Communications,
2013
19.

Gvozdev V.D., Shavrin K.N., Nefedov O.M.
Russian Chemical Bulletin,
2013
20.
Gvozdev V.D., Shavrin K.N., Egorov M.P., Nefedov O.M.
Mendeleev Communications,
2015
21.

Gvozdev V.D., Shavrin K.N., Nefedov O.M.
Russian Chemical Bulletin,
2014
22.
Gvozdev V.D., Shavrin K.N., Egorov M.P., Nefedov O.M.
Mendeleev Communications,
2016
23.
![Cyclization of alk-4-ynals with o-diaminoarenes as a selective one-pot synthesis of arylmethylidene-substituted 2,3-dihydro-1H-pyrrolo[1,2-a]benzimidazoles and 7,8-dihydro-6H-pyrrolo[1’,2’:1,2]imidazo[4,5-b]pyridines](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Gvozdev V.D., Shavrin K.N., Baskir E.G., Egorov M.P., Nefedov O.M.
Russian Chemical Bulletin,
2016
24.
![Nucleophilic addition of benzimidazoles to alkynyl bromides/palladium-catalyzed intramolecular C-H vinylation: synthesis of benzo[4,5]imidazo[2,1-a]isoquinolines.](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Peng J., Shang G., Chen C., Miao Z., Li B.
Journal of Organic Chemistry,
2013