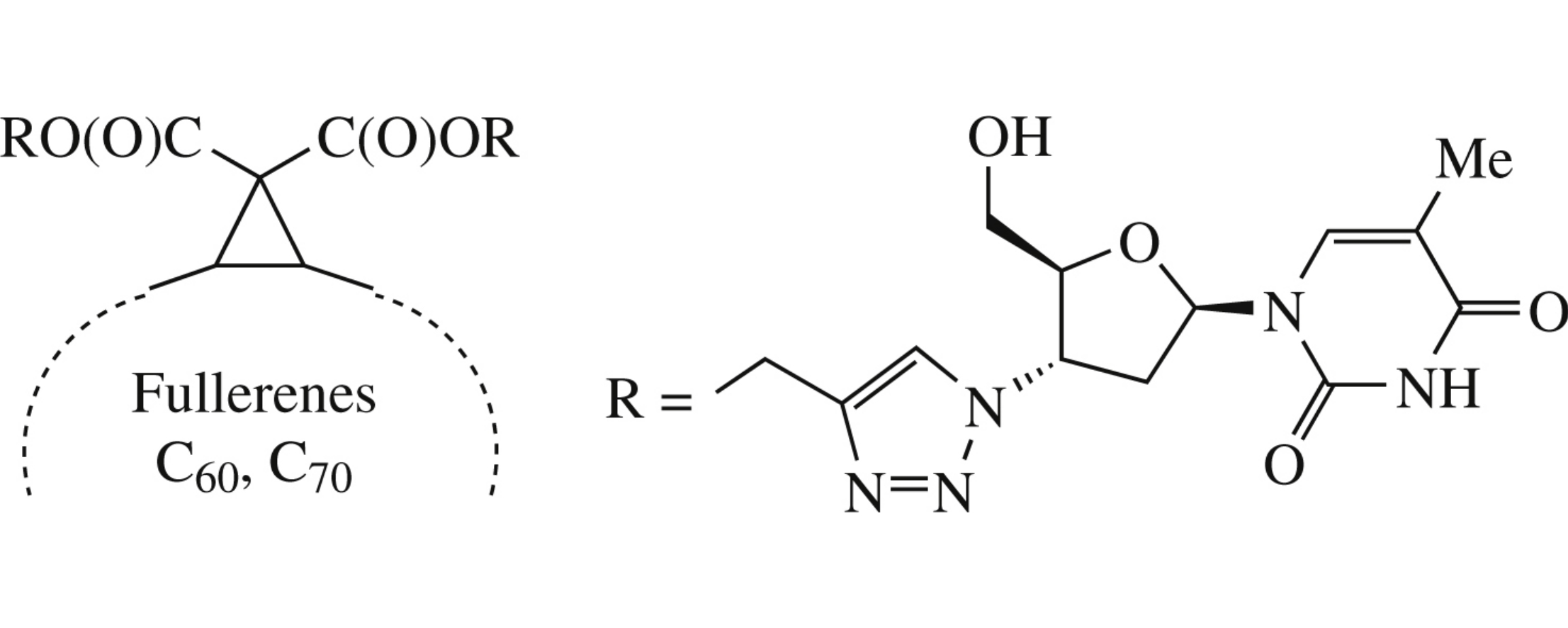
Abstract
New derivatives of fullerenes C60 and C70 containing triazole and deoxythymidine fragments are obtained by the Bingel–Hirsch and ‘click-chemistry’ reactions from acetylene methanofullerene and azido thymidine derivatives.
References
1.
10.1016/j.mencom.2017.03.033_bib0005
Piotrovsky
Fullereny v biologii (Fullerenes in Biology),
2006
2.

Gubskaya V.P., Berezhnaya L.S., Gubaidullin A.T., Faingold I.I., Kotelnikova R.A., Konovalova N.P., Morozov V.I., Litvinov I.A., Nuretdinov I.A.
Organic and Biomolecular Chemistry,
2007
3.
![Influence of water-soluble derivatives of [60]fullerene on therapeutically important targets related to neurodegenerative diseases](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Kotelnikova R.A., Smolina A.V., Grigoryev V.V., Faingold I.I., Mischenko D.V., Rybkin A.Y., Poletayeva D.A., Vankin G.I., Zamoyskiy V.L., Voronov I.I., Troshin P.A., Kotelnikov A.I., Bachurin S.O.
MedChemComm,
2014
4.
10.1016/j.mencom.2017.03.033_bib0020
Studentsov
Izv. SPbGTI (TU),
2009
5.
10.1016/j.mencom.2017.03.033_bib0025
Bakry
Int. J. Nanomed.,
2007
6.
10.1016/j.mencom.2017.03.033_bib0030
Orlova
Onkogematologiya,
2013
7.

Melnikova N.B., Korobko V.M., Gulenova M.V., Gubskaya V.P., Fazlleeva G.M., Zhiltsova O.E., Kochetkov E.N., Poddel’sky A.I., Nuretdinov I.A.
Colloids and Surfaces B: Biointerfaces,
2015
8.

Buina N.A., Fazleeva G.M., Sadreeva R.R., Nuretdinov I.A., Konovalova N.P., D'yachkovskaya R.F., Volkova L.N.
Pharmaceutical Chemistry Journal,
1993
9.
![Synthesis and Characterization of Nitrogen-Bridged [C60]Fullerene/3′-Deoxythimidine Conjugates](/storage/images/resized/xqixcltwJYe6H8Uco2JbAFfIOzt7UNKH0OcPOPzO_small_thumb.webp)
Ungurenasu C., Pinteala M., Simionescu B.C.
Synthesis,
2005
10.
10.1016/j.mencom.2017.03.033_sbref0050a
Krayevsky
Molek. Biol.,
1999
11.

Pokrovsky A.G., Pronayeva T.R., Fedyuk N.V., Shirokova E.A., Khandazhinskaya A.L., Tarusova N.B., Karpenko I.L., Krayevsky A.A.
Nucleosides, Nucleotides and Nucleic Acids,
2001
12.
![1,2,3-Triazole-[2,5-Bis-O-(tert-butyldimethylsilyl)-.beta.-D-ribofuranosyl]-3'-spiro-5''-(4''-amino-1'',2''-oxathiole 2'',2''-dioxide) (TSAO) Analogs: Synthesis and Anti-HIV-1 Activity](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Alvarez R., Velazquez S., San-Felix A., Aquaro S., Clercq E.D., Perno C., Karlsson A., Balzarini J., Camarasa M.J.
Journal of Medicinal Chemistry,
1994
13.

Kotelnikov A.I., Rybkin A.Y., Khakina E.A., Kornev A.B., Barinov A.V., Goryachev N.S., Ivanchikhina A.V., Peregudov A.S., Martynenko V.M., Troshin P.A.
Organic and Biomolecular Chemistry,
2013
14.

15.
![Novel water-soluble methanofullerenes C60[C13H18O4(OH)4]6 and C60[C9H10O4(OH)4]6: Promising uncouplers of respiration and phosphorylation](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Kalacheva N.V., Gubskaya V.P., Fazleeva G.M., Igtisamova G.R., Nuretdinov I.A., Rizvanov A.A., Cherepnev G.V.
Bioorganic and Medicinal Chemistry Letters,
2015
16.

Lucafò M., Gerdol M., Pallavicini A., Pacor S., Zorzet S., Da Ros T., Prato M., Sava G.
Toxicology,
2013
17.

Ershova E.S., Sergeeva V.A., Chausheva A.I., Zheglo D.G., Nikitina V.A., Smirnova T.D., Kameneva L.V., Porokhovnik L.N., Kutsev S.I., Troshin P.A., Voronov I.I., Khakina E.A., Veiko N.N., Kostyuk S.V.
Mutation Research - Genetic Toxicology and Environmental Mutagenesis,
2016
18.
Kornev A.B., Peregudov A.S., Martynenko V.M., Guseva G.V., Sashenkova T.E., Rybkin A.Y., Faingold I.I., Mishchenko D.V., Kotelnikova R.A., Konovalova N.P., Balzarini J., Troshin P.A.
Mendeleev Communications,
2013
19.

Lucafò M., Pacor S., Fabbro C., Da Ros T., Zorzet S., Prato M., Sava G.
Journal of Nanoparticle Research,
2012
20.
![Antimicrobial photodynamic therapy with decacationic monoadducts and bisadducts of [70]fullerene: in vitro and in vivo studies](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Huang L., Wang M., Dai T., Sperandio F.F., Huang Y., Xuan Y., Chiang L.Y., Hamblin M.R.
Nanomedicine,
2013
21.

Perrone D., Bortolini O., Fogagnolo M., Marchesi E., Mari L., Massarenti C., Navacchia M.L., Sforza F., Varani K., Capobianco M.L.
New Journal of Chemistry,
2013
22.
![Modification of nucleic acids using [3 + 2]-dipolar cycloaddition of azides and alkynes](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Ustinov A.V., Stepanova I.A., Dubnyakova V.V., Zatsepin T.S., Nozhevnikova E.V., Korshun V.A.
Russian Journal of Bioorganic Chemistry,
2010
23.
10.1016/j.mencom.2017.03.033_bib0110
Severov
Efferent. Fiz.-Khim. Meditsina,
2012
24.
Akhmetov A.R., Tuktarov A.R., Popod’ko N.R., Dzhemilev U.M.
Mendeleev Communications,
2015
25.
Zhang Q., Jin B., Peng R., Lei S., Chu S.
Mendeleev Communications,
2015
26.
Biglova Y.N., Mikheev V.V., Torosyan S.A., Biglova R.Z., Miftakhov M.S.
Mendeleev Communications,
2015
27.

Gubskaya V.P., Latypov S.K., Sibgatullina F.G., Balandina A.A., Zhelonkina T.A., Fazleeva G.M., Islamova L.N., Sharafutdinova D.R., Nuretdinov I.A., Sinyashin O.G.
Russian Chemical Bulletin,
2012
28.

Yin J., Jin L., Liu R., Li Q., Fan C., Li Y., Li W., Chen Q.
Journal of Organic Chemistry,
2006
29.

Wang F., Fu H., Jiang Y., Zhao Y.
Green Chemistry,
2008
30.
10.1016/j.mencom.2017.03.033_sbref0145a
Derome
Modern NMR Techniques for Chemistry Research,
1988
31.
10.1016/j.mencom.2017.03.033_sbref0145b
Atta-ur-Rahman
One and Two Dimensional NMR Spectroscopy,
1989