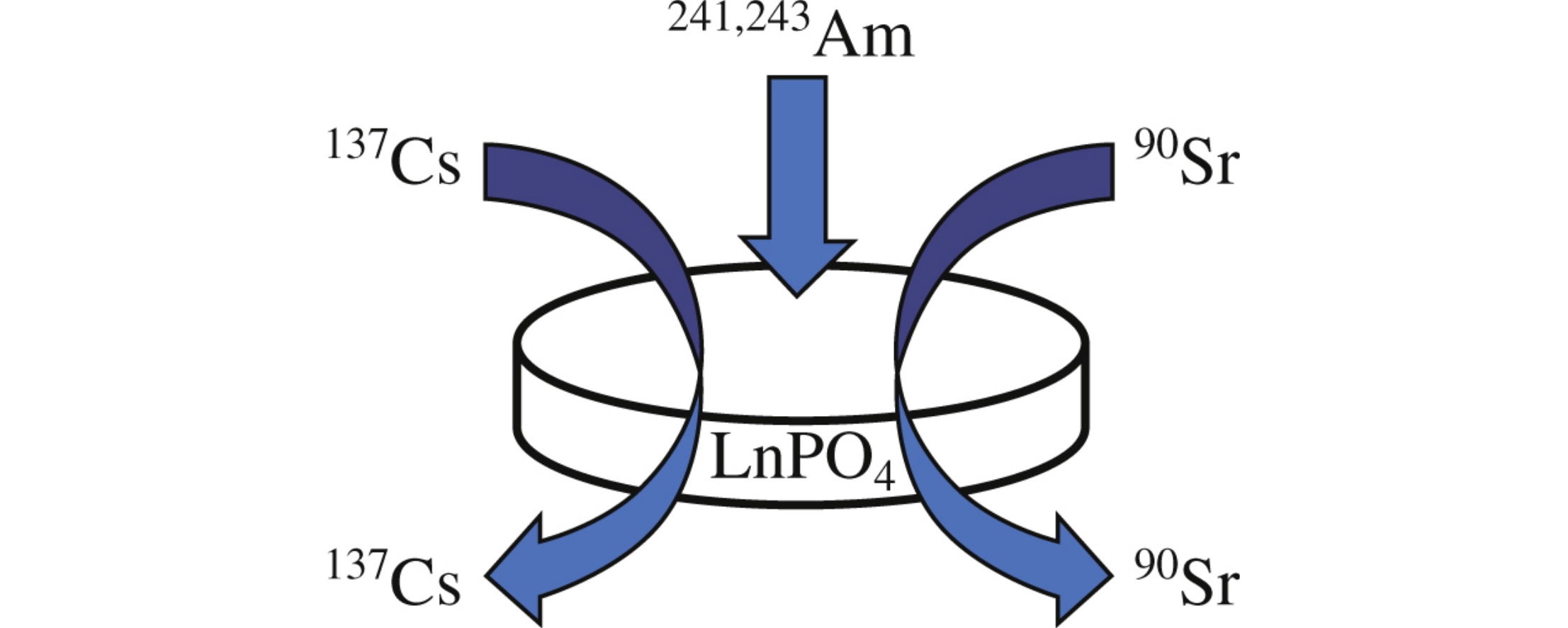
Abstract
The effect of ionic strength, pH value, and particles morphology on the Amiii sorption onto LnPO4 with different crystalline structures, i.e., monazite, xenotime, and rhabdophane, was established. The sorption capacity of the LnPO4 was found to be independent of its crystal structure and equal to ca. 1 μmol m–2.
References
1.
Myasoedov B.F., Kalmykov S.N.
Mendeleev Communications,
2015
2.

Alyapyshev M.Y., Babain V.A., Ustynyuk Y.A.
Russian Chemical Reviews,
2016
3.

Drot R., Simoni E.
Langmuir,
1999
4.

Ordoñez-Regil E., Drot R., Simoni E., Ehrhardt J.J.
Langmuir,
2002
5.

Drot R., Lindecker C., Fourest B., Simoni E.
New Journal of Chemistry,
1998
6.

Qian L., Hu P., Jiang Z., Geng Y., Wu W.
Science China Chemistry,
2010
7.

Almazan-Torres M.G., Drot R., Mercier-Bion F., Catalette H., Den Auwer C., Simoni E.
Journal of Colloid and Interface Science,
2008
8.

Drot R., Simoni E., Alnot M., Ehrhardt J.J.
Journal of Colloid and Interface Science,
1998
9.

Ordoñez-Regil E., Drot R., Simoni E.
Journal of Colloid and Interface Science,
2003
10.

Boatner L.A.
Reviews in Mineralogy and Geochemistry,
2002
11.

Meyssamy H., Riwotzki K., Kornowski A., Naused S., Haase M.
Advanced Materials,
1999
12.

Onoda H., Nariai H., Moriwaki A., Maki H., Motooka I.
Journal of Materials Chemistry A,
2002
13.

Yu C., Yu M., Li C., Liu X., Yang J., Yang P., Lin J.
Journal of Solid State Chemistry,
2009
14.

Zhang Y., Yan Z., You L., Si R., Yan C.
European Journal of Inorganic Chemistry,
2003
15.

Kijkowska R.
Journal of Materials Science,
2003
16.
10.1016/j.mencom.2017.03.028_bib0080
Boakye
J. Am. Ceram. Soc.,
2005
17.

Gavrichev K.S., Ryumin M.A., Tyurin A.V., Khoroshilov A.V., Mezentseva L.P., Osipov A.V., Ugolkov V.L., Gusarov V.V.
Journal of Thermal Analysis and Calorimetry,
2010
18.

Dong H., Liu Y., Yang P., Wang W., Lin J.
Solid State Sciences,
2010
19.

Fang Y., Xu A., Song R., Zhang H., You L., Yu J.C., Liu H.
Journal of the American Chemical Society,
2003
20.

Guan H., Zhang Y.
Journal of Solid State Chemistry,
2004
21.

Ma J., Wu Q.
Journal of Applied Crystallography,
2010
22.

Yan Z., Zhang Y., You L., Si R., Yan C.
Journal of Crystal Growth,
2004
23.

Kijkowska R.
Thermochimica Acta,
2003
24.

Mooney R.C.
Acta Crystallographica,
1950
25.

Hikichi Y., Sasaki T., Suzuki S., Murayama K., Miyamoto M.
Journal of the American Ceramic Society,
1988
26.

Celebi A.S., Kolis J.W.
Journal of the American Ceramic Society,
2004
27.
10.1016/j.mencom.2017.03.028_bib0135
Bryukhanova
Nanosystems: Phys. Chem. Math.,
2016
28.

Goldberg S., Criscenti L.J., Turner D.R., Davis J.A., Cantrell K.J.
Vadose Zone Journal,
2007