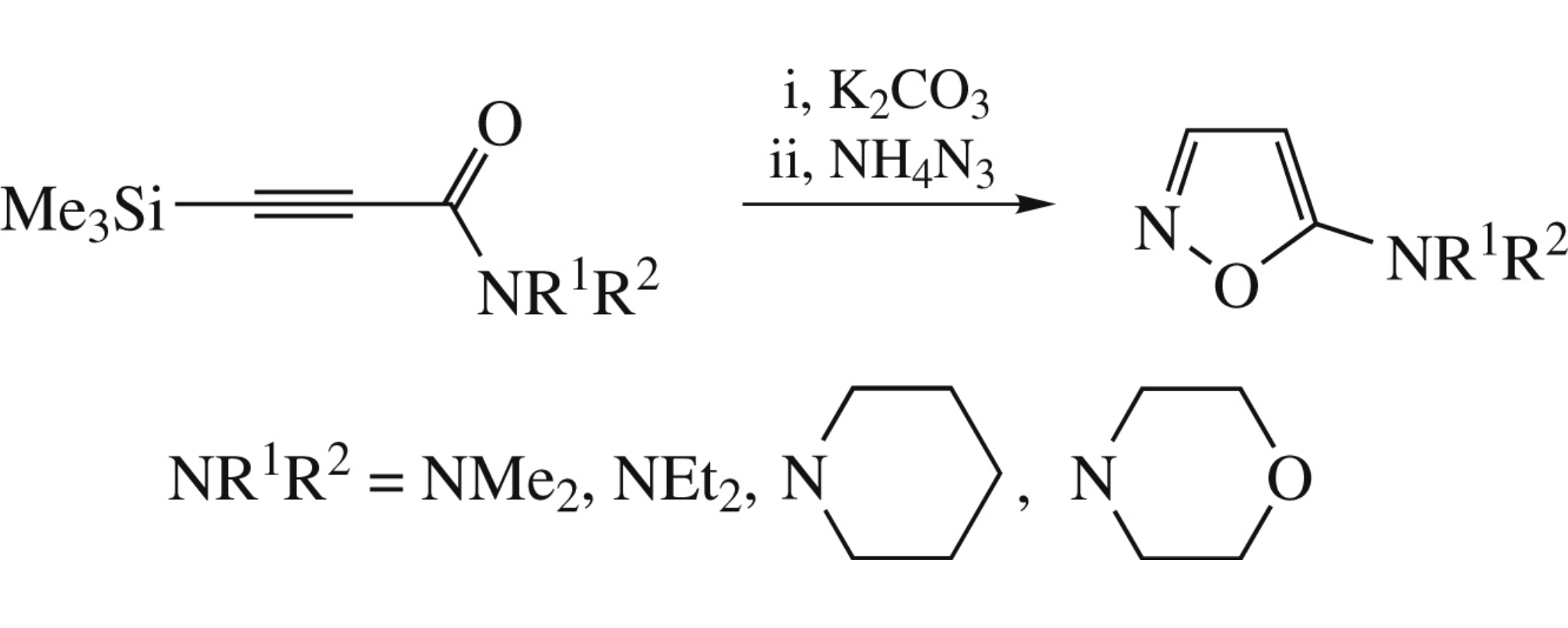
Abstract
Reaction between N,N-disubstituted 3-trimethylsilylprop-2-ynamides and ammonium azide under mild conditions affords hitherto unknown 5-aminoisoxazole derivatives in good yields.
References
1.
10.1016/j.mencom.2017.03.023_sbref0005a
Sperry
Curr. Opin. Drug Discov. Dev.,
2005
2.

Waldo J.P., Larock R.C.
Journal of Organic Chemistry,
2007
3.
10.1016/j.mencom.2017.03.023_sbref0005c
Kumar
Int. J. Pharm. Chem. Biol. Sci.,
2013
4.

Grecian S., Fokin V.
Angewandte Chemie - International Edition,
2008
5.
Kislyi V.P., Danilova E.B., Semenov V.V.
Mendeleev Communications,
2012
6.

Galenko A.V., Khlebnikov A.F., Novikov M.S., Pakalnis V.V., Rostovskii N.V.
Russian Chemical Reviews,
2015
7.

Hu F., Szostak M.
Advanced Synthesis and Catalysis,
2015
8.
Zlotin S.G., Churakov A.M., Luk’yanov O.A., Makhova N.N., Sukhorukov A.Y., Tartakovsky V.A.
Mendeleev Communications,
2015
9.
Ananikov V.P., Galkin K.I., Egorov M.P., Sakharov A.M., Zlotin S.G., Redina E.A., Isaeva V.I., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2016
10.
Vereshchagin A.N., Elinson M.N., Anisina Y.E., Ryzhkov F.V., Goloveshkin A.S., Bushmarinov I.S., Zlotin S.G., Egorov M.P.
Mendeleev Communications,
2015
11.

Micetich R.G., Raap R., Chin C.G.
Journal of Medicinal Chemistry,
1971
12.

Tomita K., Tsuzuki Y., Shibamori K., Tashima M., Kajikawa F., Sato Y., Kashimoto S., Chiba K., Hino K.
Journal of Medicinal Chemistry,
2002
13.
10.1016/j.mencom.2017.03.023_sbref0020c
Mączyński
Acta Pol. Pharm.,
2014
14.

Hamama W.S., Ibrahim M.E., Zoorob H.H.
Synthetic Communications,
2013
15.
![A CONVENIENT APPROACH TO THE SYNTHESIS OF NEW SUBSTITUTED ISOXAZOLO[5,4-D] PYRIMIDIN-4(5H)-ONES](/storage/images/resized/3SpVxcYL33bOvPq4sHxJLH2NeKNeDloahSUpNiO4_small_thumb.webp)
Davoodnia A., Bakavoli M., Pooryaghoobi N., Roshani M.
Heterocyclic Communications,
2007
16.
10.1016/j.mencom.2017.03.023_sbref0025c
Yu
Synlett,
2010
17.
Alyabiev S.B., Kravchenko D.V., Ivachtchenko A.V.
Mendeleev Communications,
2008
18.
Muravyova E.A., Tkachenko V.V., Desenko S.M., Sen’ko Y.V., Müller T.J., Vashchenko E.V., Chebanov V.A.
Arkivoc,
2013
19.
10.1016/j.mencom.2017.03.023_sbref0030a
Johnson
Synthesis,
2013
20.

Khalafy J., Akbari Dilmaghani K., Soltani L., Poursattar-Marjani A.
Chemistry of Heterocyclic Compounds,
2008
21.

Sobenina L.N., Drichkov V.N., Mikhaleva A.I., Petrova O.V., Ushakov I.A., Trofimov B.A.
Tetrahedron,
2005
22.
Lesiv A.V., Ioffe S.L., Strelenko Y.A., Bliznets I.V., Tartakovsky V.A.
Mendeleev Communications,
2002
23.

Saad A., Vaultier M., Derdour A.
Molecules,
2004
24.

Himbert G., Kuhn H., Barz M.
European Journal of Organic Chemistry,
1990
25.

Medvedeva A.S., Andreev M.V., Safronova L.P.
Russian Journal of Organic Chemistry,
2010
26.

Demina M.M., Nguyen T.L., Shaglaeva N.S., Mareev A.V., Medvedeva A.S.
Russian Journal of Organic Chemistry,
2012
27.

Medvedeva A.S., Demina M.M., Nguyen T.L., Vu T.D., Bulanov D.A., Novokshonov V.V.
Russian Journal of Organic Chemistry,
2013
28.

Demina M.M., Novopashin P.S., Sarapulova G.I., Larina L.I., Smolin A.S., Fundamenskii V.S., Kashaev A.A., Medvedeva A.S.
Russian Journal of Organic Chemistry,
2004
29.
Medvedeva A.S., Demina M.M., Vu T.D., Andreev M.V., Shaglaeva N.S., Larina L.I.
Mendeleev Communications,
2016
30.

Pinho e Melo T.M., Lopes C.S., Rocha Gonsalves A.M., Storr R.C.
Synthesis,
2002
31.
10.1016/j.mencom.2017.03.023_sbref0070b
Banert
in Organic Azides: Syntheses and Applications,
2010
32.

Andreev M.V., Medvedeva A.S., Safronova L.P.
Russian Journal of Organic Chemistry,
2013
33.
10.1016/j.mencom.2017.03.023_bib0080
1976
34.

Coppola G.M., Damon R.E.
Synthetic Communications,
1993
35.

Undheim K., Riege L.A.
Journal of the Chemical Society Perkin Transactions 1,
1975
36.
10.1016/j.mencom.2017.03.023_bib0090
Raphael
Acetylenic Compounds in Organic Synthesis,
1955
37.

Andreev M.V., Safronova L.P., Medvedeva A.S.
Russian Journal of Organic Chemistry,
2011
38.
![Synthesis of Amide-Substituted Isoxazoles and Pyrazoles via Regioselective [3+2] Cycloadditions of Terminally Unsubstituted Ynamides](/storage/images/resized/SovkqI6gX6to8o5XBnS9RNxEOYVicn7hatkMzrvl_small_thumb.webp)
P. Hsung R., Li H., You L., Zhang X., L. Johnson W., Figueroa R.
Heterocycles,
2007
39.

GRUNDMANN C.
Synthesis,
1970
40.

R. Katritzky A., Jiang R., K. Singh S.
Heterocycles,
2004