Abstract
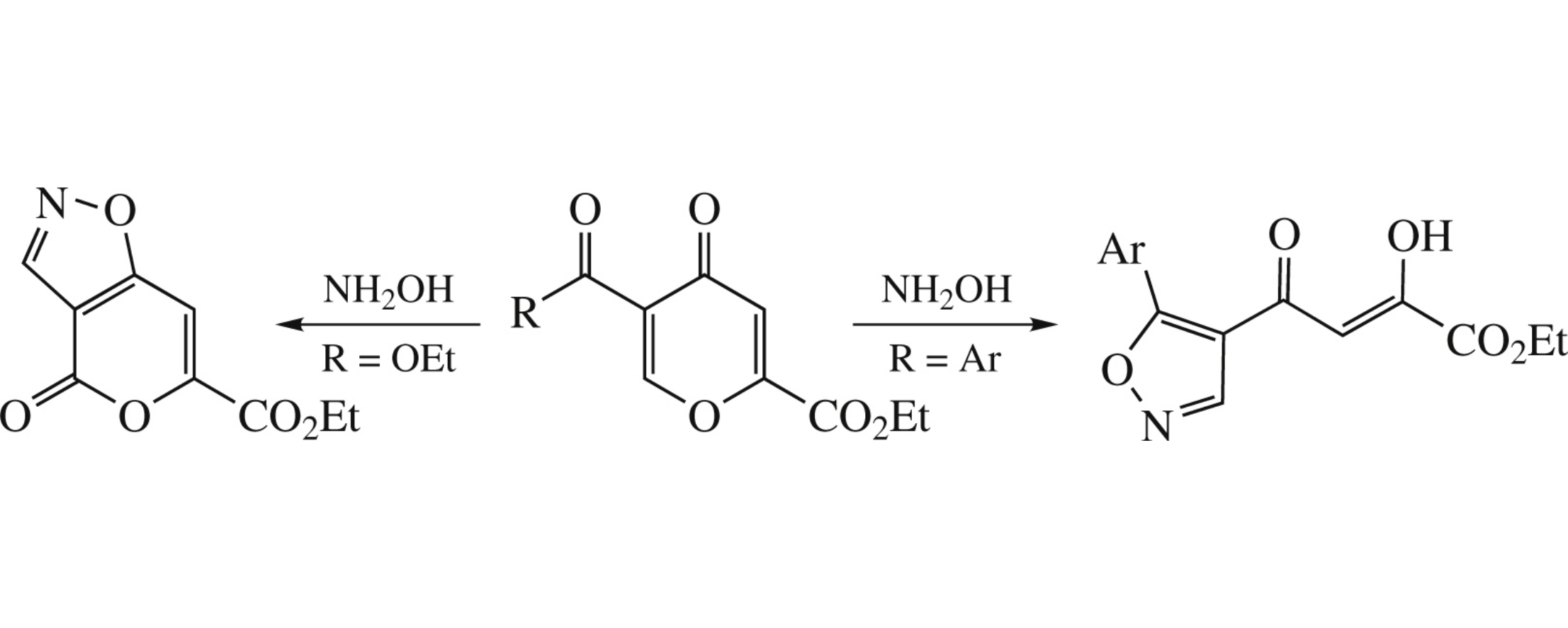
Ethyl 5-aroyl-4-pyrone-2-carboxylates react with hydroxylamine in ethanol at –20°C for 30 days to produce ethyl 4-(5-arylisoxazol-4-yl)-2,4-dioxobutanoates (yields 20–67%). The similar reaction of diethyl 4-pyrone-2,5-dicarboxylate gives ethyl 4-oxo-4H-pyrano[3,4-d]isoxazole-6-carboxylate in 37% yield.
References
1.

Di Santo R.
Journal of Medicinal Chemistry,
2013
2.

Summa V., Petrocchi A., Bonelli F., Crescenzi B., Donghi M., Ferrara M., Fiore F., Gardelli C., Gonzalez Paz O., Hazuda D.J., Jones P., Kinzel O., Laufer R., Monteagudo E., Muraglia E., et. al.
Journal of Medicinal Chemistry,
2008
3.

Johns B.A., Kawasuji T., Weatherhead J.G., Taishi T., Temelkoff D.P., Yoshida H., Akiyama T., Taoda Y., Murai H., Kiyama R., Fuji M., Tanimoto N., Jeffrey J., Foster S.A., Yoshinaga T., et. al.
Journal of Medicinal Chemistry,
2013
4.

Cuzzucoli Crucitti G., Métifiot M., Pescatori L., Messore A., Madia V.N., Pupo G., Saccoliti F., Scipione L., Tortorella S., Esposito F., Corona A., Cadeddu M., Marchand C., Pommier Y., Tramontano E., et. al.
Journal of Medicinal Chemistry,
2015
5.

Hazuda D.J., Felock P., Witmer M., Wolfe A., Stillmock K., Grobler J.A., Espeseth A., Gabryelski L., Schleif W., Blau C., Miller M.D.
Science,
2000
6.
![Inhibition of HIV-1 Ribonuclease H by a Novel Diketo Acid, 4-[5-(Benzoylamino)thien-2-yl]-2,4-dioxobutanoic Acid](/storage/images/resized/OuA6WPZekwEiUM99mP2pSqj4yHPxHezM6xgLhAjO_small_thumb.webp)
Shaw-Reid C.A., Munshi V., Graham P., Wolfe A., Witmer M., Danzeisen R., Olsen D.B., Carroll S.S., Embrey M., Wai J.S., Miller M.D., Cole J.L., Hazuda D.J.
Journal of Biological Chemistry,
2003
7.

Di Santo R.
Current Medicinal Chemistry,
2011
8.

Obydennov D.L., Röschenthaler G., Sosnovskikh V.Y.
Tetrahedron Letters,
2014
9.

Obydennov D.L., Sosnovskikh V.Y.
Chemistry of Heterocyclic Compounds,
2014
10.

Yates P., Jorgenson M.J., Roy S.K.
Canadian Journal of Chemistry,
1962
11.

Soliman G., El-Kholy I.E.
Journal of the Chemical Society (Resumed),
1954
12.

El-Sayed El-Kholy I., Rafla F.K., Soliman G.
Journal of the Chemical Society (Resumed),
1962
13.

El-Kholy I.E., Marei M.G., Mishrikey M.M.
Journal of Heterocyclic Chemistry,
1979
14.

Marei M.G., El-Ghanam M.
Bulletin of the Chemical Society of Japan,
1992
15.

Strah S., Golobič A., Golič L., Stanovnik B.
Journal of Heterocyclic Chemistry,
1997
16.

Mo D., Wink D.A., Anderson L.L.
Organic Letters,
2012
17.

Obydennov D.L., Usachev B.I.
Journal of Fluorine Chemistry,
2012
18.

Obydennov D.L., Sidorova E.S., Usachev B.I., Sosnovskikh V.Y.
Tetrahedron Letters,
2013
19.

Obydennov D.L., Röschenthaler G., Sosnovskikh V.Y.
Tetrahedron Letters,
2013
20.

Imagawa T., Haneda A., Kawanisi M.
Magnetic Resonance in Chemistry,
1980
21.
10.1016/j.mencom.2017.03.022_bib0075
Becker
Synthesis,
2005
22.

F. Abdel-Wahab B., Abdel-Gawad H., A. Mohamed H., M. Dawood K.
Heterocycles,
2010
23.

Nolsöe J.M., Weigelt D.
Journal of Heterocyclic Chemistry,
2009
24.

Eftekhari-Sis B., Zirak M.
Chemical Reviews,
2014