Abstract
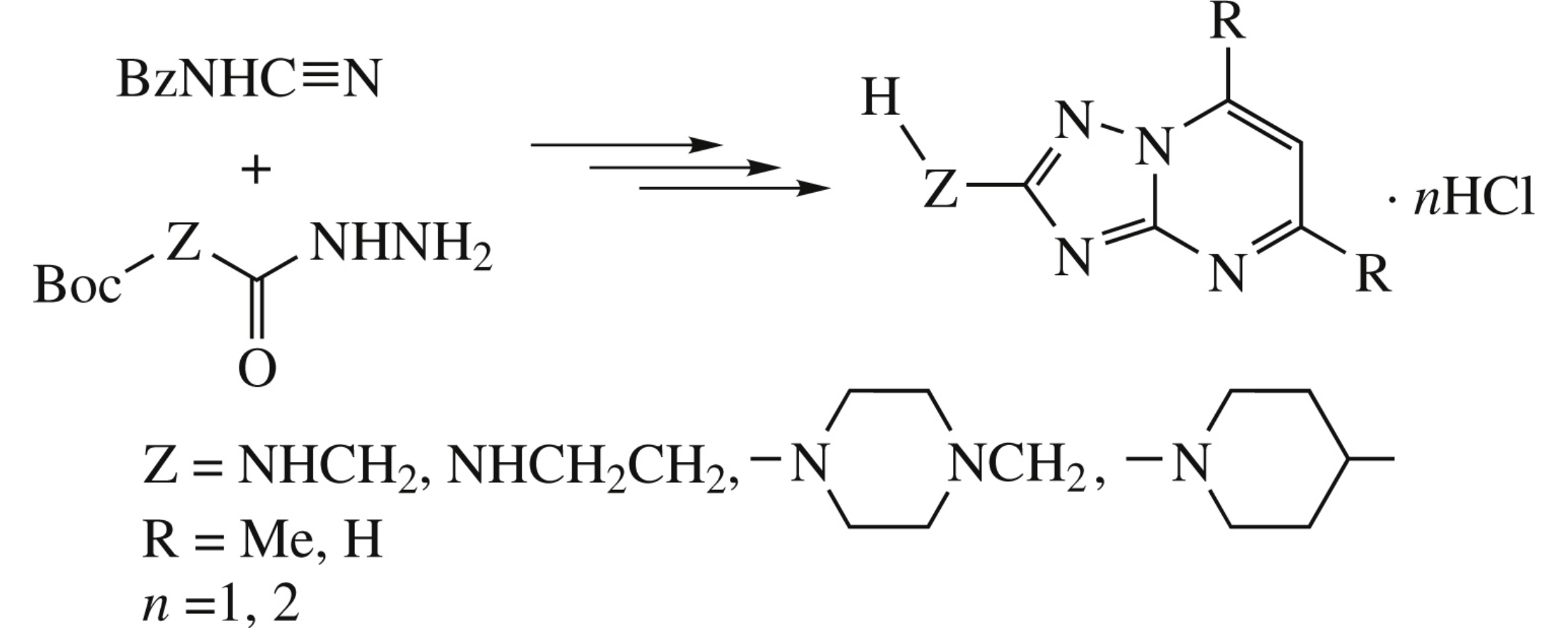
Nickel(ii) acetylacetonate-promoted cycloaddition between Boc-protected amino acid hydrazides and benzoylcyanamide followed by deprotection gives 2-amino-5-aminoalkyl-1,2,4-triazoles, whose reaction with 1,3-dielectrophiles affords the title compounds.
References
1.

Glanville A.R., Scott A.I., Morton J.M., Aboyoun C.L., Plit M.L., Carter I.W., Malouf M.A.
Journal of Heart and Lung Transplantation,
2005
2.
Chudinov M.V., Matveev A.V., Prutkov A.N., Konstantinova I.D., Fateev I.V., Prasolov V.S., Smirnova O.A., Ivanov A.V., Galegov G.A., Deryabin P.G.
Mendeleev Communications,
2016
3.

Cha R., Sobel J.D.
Expert Review of Anti-Infective Therapy,
2004
4.
Bel’skaya N.P., Demina M.A., Sapognikova S.G., Fan Z., Zhang H., Dehaen W., Bakulev V.A.
Arkivoc,
2008
5.
Shagun L.G., Dorofeev I.A., Zhilitskaya L.V., Larina L.I., Yarosh N.O.
Mendeleev Communications,
2015
6.
F. Calo, M. Kordes, H. Kraus, T. Mietzner, T. Seitz, K. Kreuz, M. Pasternak, T.W. Newton and D. Massa, Patent WO 007564 A1, 2015.
7.
K. Baumann, A. Floh, E. Goetschi, H. Jacobsen, S. Jolidon and T. Luebbers, Patent US 20090215759 A1, 2009.
8.

Bell K., Sunose M., Ellard K., Cansfield A., Taylor J., Miller W., Ramsden N., Bergamini G., Neubauer G.
Bioorganic and Medicinal Chemistry Letters,
2012
9.

Gregory T.F., Wright J.L., Wise L.D., Meltzer L.T., Serpa K.A., Konkoy C.S., Whittemore E.R., Woodward R.M.
Bioorganic and Medicinal Chemistry Letters,
2000
10.
![Synthesis and Anti-tumor Activities of Novel [1,2,4]triazolo[1,5-a]pyrimidines](/storage/images/resized/MjH1ITP7lMYGxeqUZfkt2BnVLgjkk413jwBV97XX_small_thumb.webp)
Zhao X., Zhao Y., Guo S., Song H., Wang D., Gong P.
Molecules,
2007
11.
![Regioselective synthesis of 6-aryl-benzo[h][1,2,4]-triazolo[5,1-b]quinazoline-7,8-diones as potent antitumoral agents.](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
wu L., Zhang C., Li W.
Bioorganic and Medicinal Chemistry Letters,
2013
12.
![Synthesis, characterization, crystal structures and in vitro antistaphylococcal activity of organotin(IV) derivatives with 5,7-disubstituted-1,2,4-triazolo[1,5-a]pyrimidine](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Girasolo M.A., Canfora L., Sabatino P., Schillaci D., Foresti E., Rubino S., Ruisi G., Stocco G.
Journal of Inorganic Biochemistry,
2012
13.
![Platinum(II) complexes with 5,7-disubstituted-1,2,4-triazolo [1,5-a]pyrimidines: Spectroscopical characterization and cytotoxic activity in vitro](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Łakomska I., Fandzloch M., Popławska B., Sitkowski J.
Spectrochimica Acta - Part A: Molecular and Biomolecular Spectroscopy,
2012
14.
J. M. H. Sheridan, J.R. Heal, W.D. O. Hamilton and I. Pike, Patent WO 2012080729 A2, 2012.
15.

Kokel D., Bryan J., Laggner C., White R., Cheung C.Y., Mateus R., Healey D., Kim S., Werdich A.A., Haggarty S.J., MacRae C.A., Shoichet B., Peterson R.T.
Nature Chemical Biology,
2010
16.

Yu W., Goddard C., Clearfield E., Mills C., Xiao T., Guo H., Morrey J.D., Motter N.E., Zhao K., Block T.M., Cuconati A., Xu X.
Journal of Medicinal Chemistry,
2011
17.
10.1016/j.mencom.2017.03.021_bib0080
Pandeya
J. Indian Chem. Soc.,
1980
18.
![A convenient method for the synthesis of 7-amino-substituted 1,2,4-triazolo[1,5-a][1,3,5]triazin-5-amines](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Dolzhenko A.V., Pastorin G., Dolzhenko A.V., Chui W.K.
Tetrahedron Letters,
2008
19.

Liu F., Li F., Ma A., Dobrovetsky E., Dong A., Gao C., Korboukh I., Liu J., Smil D., Brown P.J., Frye S.V., Arrowsmith C.H., Schapira M., Vedadi M., Jin J., et. al.
Journal of Medicinal Chemistry,
2013
20.

Bonacorso H.G., Bortolotto G.P., Navarini J., Porte L.M., Wiethan C.W., Zanatta N., Martins M.A., Flores A.F.
Journal of Fluorine Chemistry,
2010
21.

Mamalis P., Green J., Mchale D.
Journal of the Chemical Society (Resumed),
1960
22.
G. Mitchell, Patent WO 144234 A1, 2013.
23.

Dorokhov V.A., Gordeev M.F., Dem'yanets Z.K., Bochkareva M.N., Bogdanov V.S.
Russian Chemical Bulletin,
1989
24.

Dorokhov V.A., Gordeev M.F., Bogdanov V.S.
Russian Chemical Bulletin,
1988
25.

Dorokhov V.A., Gordeev M.F., Shashkova E.M., Komkov A.V., Bogdanov V.S.
Russian Chemical Bulletin,
1991
26.

Prezent M.A., Daeva E.D., Baranin S.V., Dorokhov V.A.
Russian Chemical Bulletin,
2015
27.
![Differentiation between [1,2,4]triazolo[1,5-a] pyrimidine and [1,2,4]triazolo[4,3-a]- pyrimidine regioisomers by 1H15N HMBC experiments](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Salgado A., Varela C., García Collazo A.M., Pevarello P.
Magnetic Resonance in Chemistry,
2010
28.

Crowther A.F., Curd F.H., Rose F.L.
Journal of the Chemical Society (Resumed),
1948
29.

Poojari S., Parameshwar Naik P., Krishnamurthy G.
Tetrahedron Letters,
2014
30.

Kumar D., Patel G., Chavers A.K., Chang K., Shah K.
European Journal of Medicinal Chemistry,
2011