Abstract
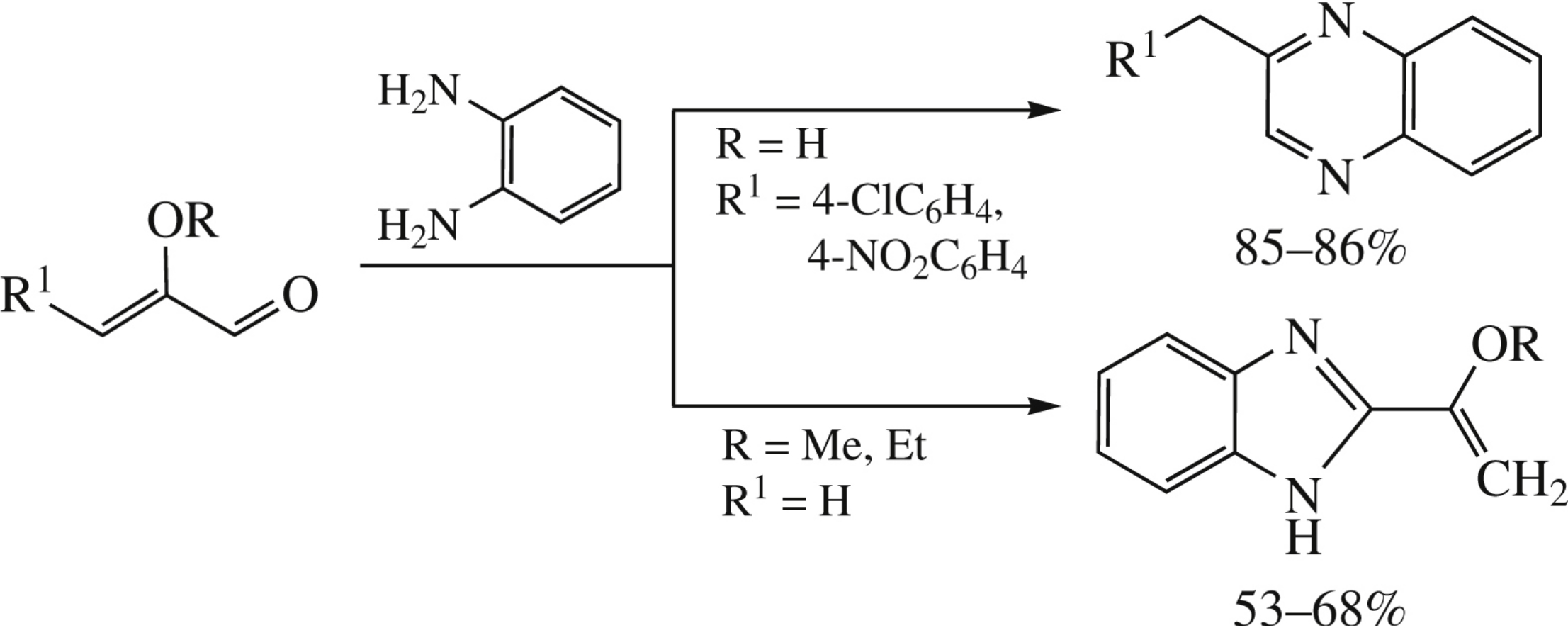
The reaction of o-phenylenediamine with 2-alkoxypropenals at room temperature affords 2-(1-alkoxyvinyl)benzimidazoles (53–68%), while 2-hydroxy-3-arylpropenals give 2-(arylmethyl) quinoxalines (85–86%).
References
1.

Keiko N.A., Vchislo N.V.
Asian Journal of Organic Chemistry,
2016
2.

Keiko N.A., Vchislo N.V.
Asian Journal of Organic Chemistry,
2016
3.

Kim M.K., Shin H., Park K., Kim H., Park J., Kim K., Nam J., Choo H., Chong Y.
Journal of Medicinal Chemistry,
2015
4.

Huang S., Hsei I., Chen C.
Bioorganic and Medicinal Chemistry,
2006
5.

Li Z., Li L., Sun T., Liu L., Xie Z.
Dyes and Pigments,
2016
6.

Shin Y., Suchomel J., Cardozo M., Duquette J., He X., Henne K., Hu Y., Kelly R.C., McCarter J., McGee L.R., Medina J.C., Metz D., San Miguel T., Mohn D., Tran T., et. al.
Journal of Medicinal Chemistry,
2015
7.
Gvozdev V.D., Shavrin K.N., Egorov M.P., Nefedov O.M.
Mendeleev Communications,
2016
8.
![Advances in the synthesis of imidazo[1,5-a]- and imidazo[1,2-a]quinoxalines](/storage/images/resized/9Mus3KG1Tkd7Bwaurt8H3RwWh0CxRlGoO6ng9UK1_small_thumb.webp)
Mamedov V.A., Kalinin A.A.
Russian Chemical Reviews,
2014
9.

Hu B., Unwalla R.J., Goljer I., Jetter J.W., Quinet E.M., Berrodin T.J., Basso M.D., Feingold I.B., Nilsson A.G., Wilhelmsson A., Evans M.J., Wrobel J.E.
Journal of Medicinal Chemistry,
2010
10.

Smits R.A., Lim H.D., Hanzer A., Zuiderveld O.P., Guaita E., Adami M., Coruzzi G., Leurs R., de Esch I.J.
Journal of Medicinal Chemistry,
2008
11.

Benitez D., Cabrera M., Hernández P., Boiani L., Lavaggi M.L., Di Maio R., Yaluff G., Serna E., Torres S., Ferreira M.E., Vera de Bilbao N., Torres E., Pérez-Silanes S., Solano B., Moreno E., et. al.
Journal of Medicinal Chemistry,
2011
12.

Keiko N.A., Vchislo N.V., Larina L.I.
Russian Journal of Organic Chemistry,
2013
13.
Keiko N.A., Vchislo N.V., Verochkina E.A., Chuvashev Y.A., Larina L.I.
Mendeleev Communications,
2016
14.

Chikashita H., Nishida S., Miyazaki M., Morita Y., Itoh K.
Bulletin of the Chemical Society of Japan,
1987
15.

Alloum A.B., Bougrin K., Soufiaoui M.
Tetrahedron Letters,
2003
16.

Lin S., Yang L.
Tetrahedron Letters,
2005