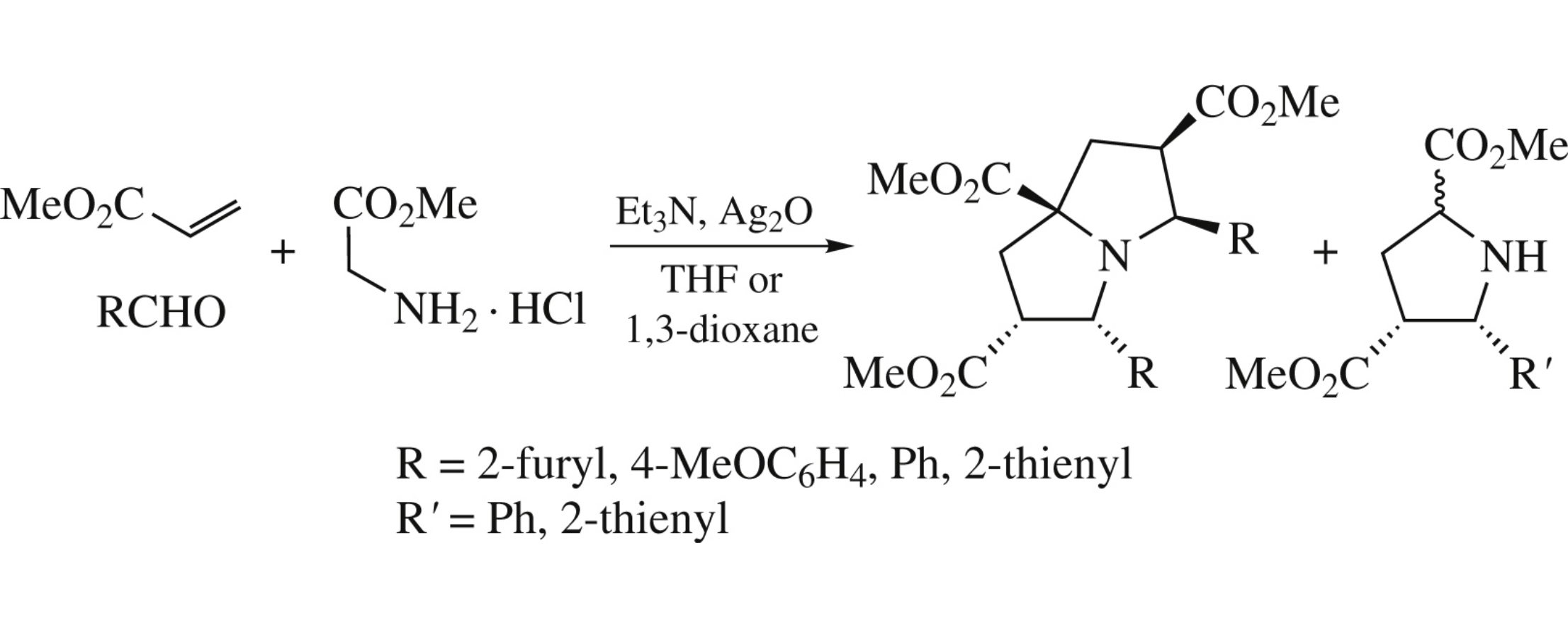
Abstract
A one-pot synthesis of new multi-functional pyrrolizidines from simple starting compounds was carried out by a double 1,3-dipolar cycloaddition protocol.
References
1.

Fletcher M.T., Hayes P.Y., Somerville M.J., De Voss J.J.
Journal of Agricultural and Food Chemistry,
2011
2.

Bianco A., Sonksen C.P., Roepstorff P., Briand J.
Journal of Organic Chemistry,
2000
3.

Gothelf K.V., Jørgensen K.A.
Chemical Reviews,
1998
4.
10.1016/j.mencom.2017.03.019_sbref0020a
Brock
Org. Lett., 2011, 13,
1594
5.

Ponpandian T., Muthusubramanian S.
Tetrahedron,
2013
6.

Lahiri R., Palanivel A., Kulkarni S.A., Vankar Y.D.
Journal of Organic Chemistry,
2014
7.

Sengupta T., Khamarui S., Samanta S., Maiti D.K.
Chemical Communications,
2013
8.

Mancebo-Aracil J., Nájera C., Sansano J.M.
Chemical Communications,
2013
9.

Mancebo-Aracil J., Nájera C., Castelló L.M., Sansano J.M., Larrañaga O., de Cózar A., Cossío F.P.
Tetrahedron,
2015
10.

Khlebnikov A.F., Novikov M.S., Kostikov R.R.
Russian Chemical Reviews,
2005
11.

Fukuzawa S., Oki H.
Organic Letters,
2008
12.

Singh M.S., Chowdhury S., Koley S.
Tetrahedron,
2016
13.
![One-pot double [3 + 2] cycloaddition for diastereoselective synthesis of tetracyclic pyrrolidine compounds](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Lu Q., Song G., Jasinski J.P., Keeley A.C., Zhang W.
Green Chemistry,
2012
14.
Zlotin S.G., Churakov A.M., Luk’yanov O.A., Makhova N.N., Sukhorukov A.Y., Tartakovsky V.A.
Mendeleev Communications,
2015
15.
Mikhaylov A.A., Zhmurov P.A., Naumova A.S., Khoroshutina Y.A., Sukhorukov A.Y., Ioffe S.L.
Mendeleev Communications,
2015
16.

Lim A.D., Codelli J.A., Reisman S.E.
Chemical Science,
2013
17.
10.1016/j.mencom.2017.03.019_bib0050
Sheldrick
Acta Crystallogr.,
2014