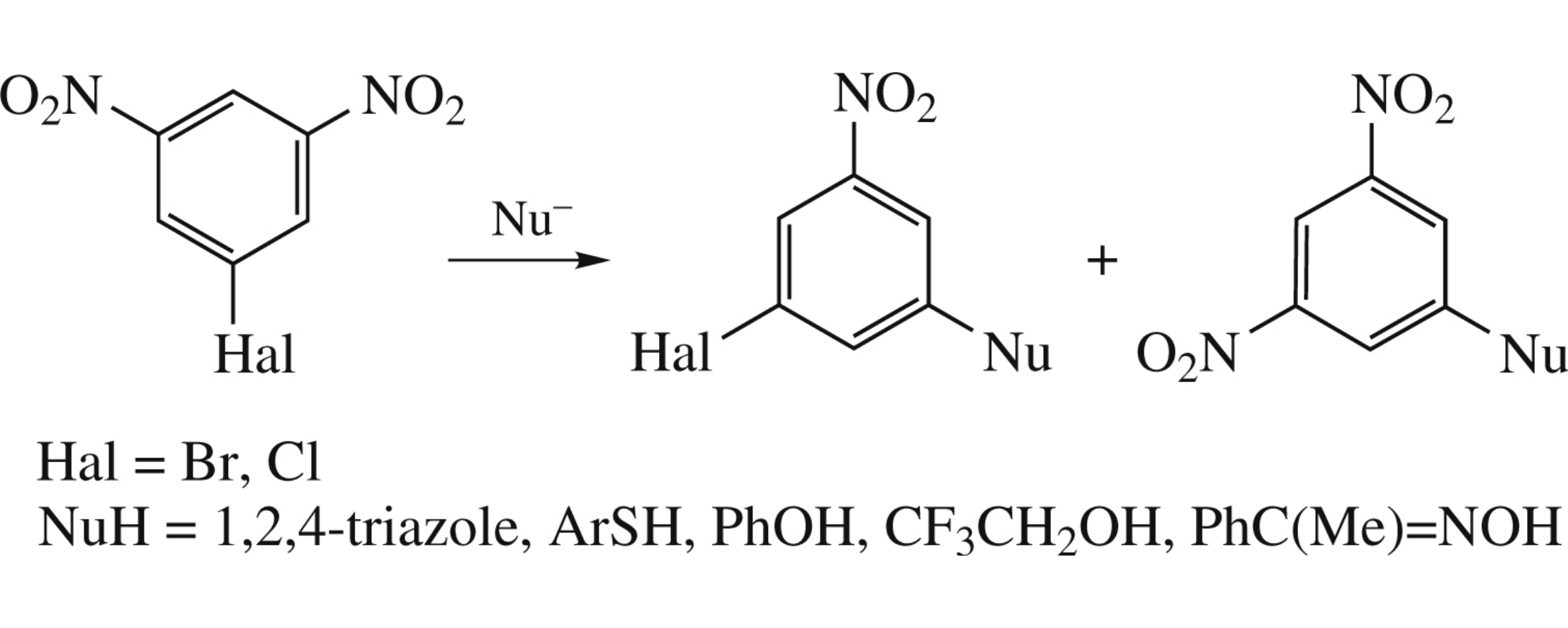
Abstract
Aromatic nucleophilic substitution reactions in 1-halo-3,5-dinitrobenzenes (where the halogen is bromine or chlorine) can occur with replacement of either a nitro group or a halogen atom, depending on the nature of anionic nucleophile Nu– as a Lewis base (hard, soft or intermediate), as well as on the polarity of the dipolar aprotic solvent.
References
1.
Shevelev S.A., Dutov M.D., Vatsadze I.A., Serushkina O.V., Rusanov A.L., Andrievskii A.M.
Mendeleev Communications,
1995
2.
Shevelev S.A., Dutov M.D., Korolev M.A., Sapozhnikov O.Y., Rusanov A.L.
Mendeleev Communications,
1998
3.

Kokurkina G.V., Dutov M.D., Shevelev S.A., Popkov S.V., Zakharov A.V., Poroikov V.V.
European Journal of Medicinal Chemistry,
2011
4.

Serushkina O.V., Dutov M.D., Sapozhnikov O.Y., Ugrak B.I., Shevelev S.A.
Russian Journal of Organic Chemistry,
2002
5.
Sapozhnikov O.Y., Dutov M.D., Korolev M.A., Kachala V.V., Shevelev S.A.
Mendeleev Communications,
2001
6.
Dutov M.D., Serushkina O.V.
Mendeleev Communications,
2013
7.
10.1016/j.mencom.2017.03.018_bib0035
Miller
Nucleophilic Aromatic Substitution,
1968
8.
10.1016/j.mencom.2017.03.018_sbref0040a
Terrier
Nucleophilic Aromatic Displacement. The Influence of the Nitro Group,
1991
9.
10.1016/j.mencom.2017.03.018_sbref0040b
Terrier
Modern Nucleophilic Aromatic Substitution,
2013
10.

Vlasov V.M.
Russian Chemical Reviews,
2003
11.

Bevan C.W., Foley A.J., Hirst J., Uwamu W.O.
Journal of the Chemical Society B Physical Organic,
1970
12.
10.1016/j.mencom.2017.03.018_bib0050
Knyazev
Russ. J. Org. Chem.,
1976
13.

Martin P.
Helvetica Chimica Acta,
1988
14.

Hameed P S., Patil V., Solapure S., Sharma U., Madhavapeddi P., Raichurkar A., Chinnapattu M., Manjrekar P., Shanbhag G., Puttur J., Shinde V., Menasinakai S., Rudrapatana S., Achar V., Awasthy D., et. al.
Journal of Medicinal Chemistry,
2014
15.

Miroshnikova O.V., Hudson T.H., Gerena L., Kyle D.E., Lin A.J.
Journal of Medicinal Chemistry,
2007
16.

Effenberger F., Koch M., Streicher W.
Chemische Berichte,
1991
17.

Belfield A.J., Brown G.R., Foubister A.J., Ratcliffe P.D.
Tetrahedron,
1999
18.
10.1016/j.mencom.2017.03.018_bib0080
Pearson
Hard and Soft Acids and Bases,
1973
19.

Guillot-Edelheit G., Laloi-Diard M., Eisenstin O.
Tetrahedron,
1978
20.

Buncel E., Cannes C., Chatrousse A., Terrier F.
Journal of the American Chemical Society,
2002
21.

Laloi-Diard M., Verchere J., Gosselin P., Terrier F.
Tetrahedron Letters,
1984
22.

Terrier F., Rodriguez-Dafonte P., Le Guével E., Moutiers G.
Organic and Biomolecular Chemistry,
2006
23.

Pratt D.A., Pesavento R.P., van der Donk W.A.
Organic Letters,
2005
24.

Serushkina O.V., Dutov M.D., Solkan V.N., Shevelev S.A.
Russian Chemical Bulletin,
2001
25.

Shevelev S.A., Dutov M.D., Serushkina O.V.
Russian Chemical Bulletin,
1995
26.
Shevelev S.A., Vatsadze I.A., Dutov M.D.
Mendeleev Communications,
2002