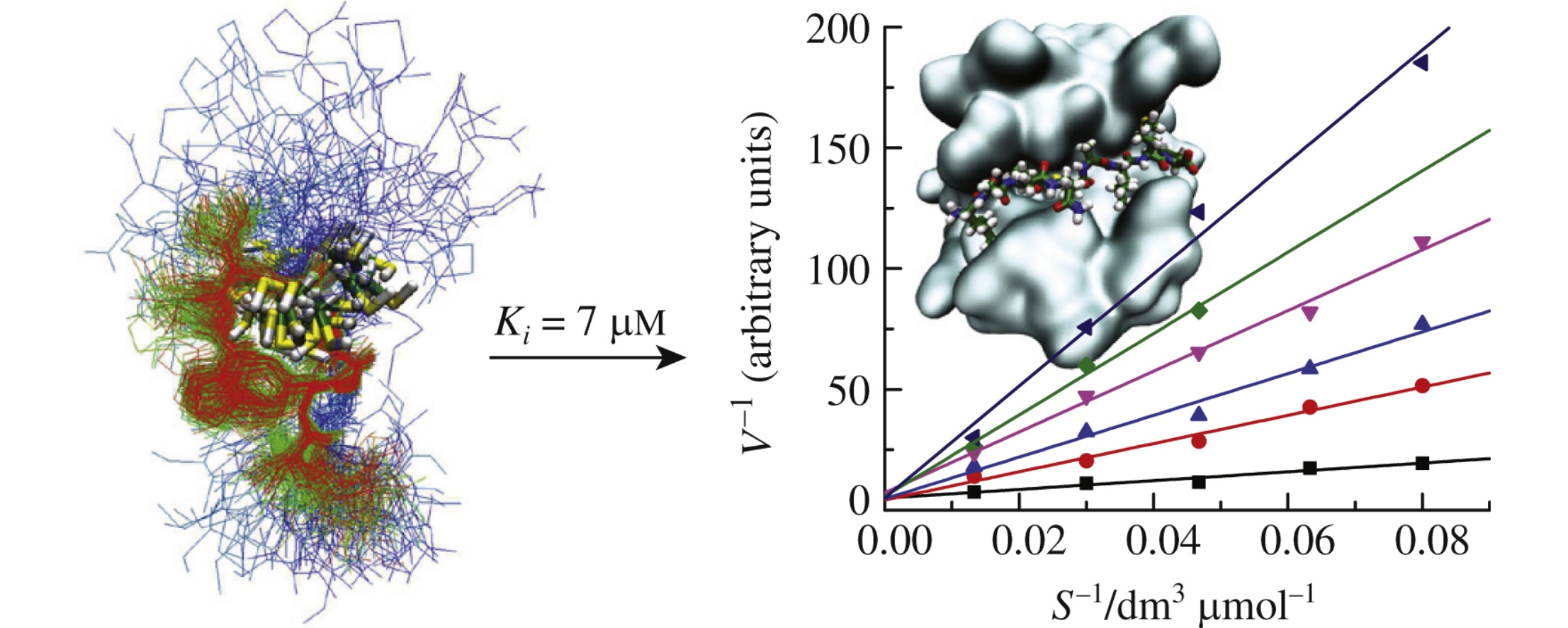
Abstract
Taking matrix metalloproteinase MMP-2 as an example, we demonstrate that the rational design of oligopeptide-based inhibitors by molecular modeling should involve both a study of interactions in the active sites of the target enzyme and the conformational dynamics of the oligopeptide in solution.
References
1.

Vandenbroucke R.E., Libert C.
Nature Reviews Drug Discovery,
2014
2.

Brinckerhoff C.E., Matrisian L.M.
Nature Reviews Molecular Cell Biology,
2002
3.

Overall C.M., López-Otín C.
Nature Reviews Cancer,
2002
4.

Ndinguri M., Bhowmick M., Tokmina-Roszyk D., Robichaud T., Fields G.
Molecules,
2012
5.

Higashi S., Miyazaki K.
Journal of Biological Chemistry,
2003
6.

Hashimoto H., Takeuchi T., Komatsu K., Miyazaki K., Sato M., Higashi S.
Journal of Biological Chemistry,
2011
7.

Vasilevskaya T., Khrenova M.G., Nemukhin A.V., Thiel W.
Journal of Computational Chemistry,
2015
8.

Pelmenschikov V., Siegbahn P.E.
Inorganic Chemistry,
2002
9.

Díaz N., Suárez D., Suárez E.
Proteins: Structure, Function and Genetics,
2009
10.

Vasilevskaya T., Khrenova M.G., Nemukhin A.V., Thiel W.
Journal of Computational Chemistry,
2016
11.
Vasilevskaya T., Khrenova M.G., Nemukhin A.V., Thiel W.
Mendeleev Communications,
2016
12.

Khrenova M.G., Savitsky A.P., Topol I.A., Nemukhin A.V.
Journal of Physical Chemistry B,
2014
13.

Khrenova M.G., Nemukhin A.V., Savitsky A.P.
Journal of Physical Chemistry B,
2014
14.
Khrenova M.G., Solovyev I.D., Azev V.N., Lapshin G.D., Savitsky A.P.
Mendeleev Communications,
2016
15.

Miyazaki K., Hasegawa M., Funahashi K., Umeda M.
Nature,
1993
16.

Netzel-Arnett S., Mallya S.K., Nagase H., Birkedal-Hansen H., Van Wart H.E.
Analytical Biochemistry,
1991
17.

Murphy G., Nguyen Q., Cockett M.I., Atkinson S.J., Allan J.A., Knight C.G., Willenbrock F., Docherty A.J.
Journal of Biological Chemistry,
1994
18.
10.1016/j.mencom.2017.03.017_bib0090
Voevodin
Otkrytye Sistemy,
2012