Abstract
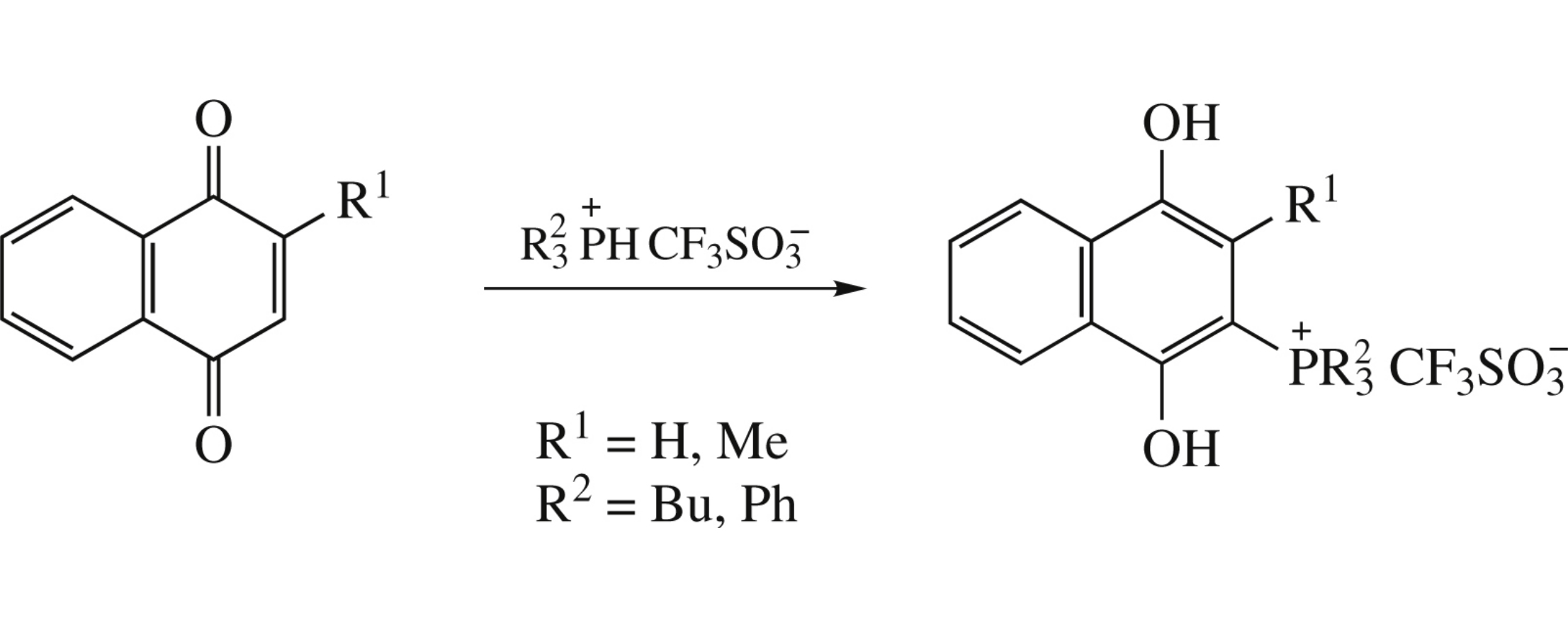
(1,4-Dihydroxynaphthalen-2-yl)phosphonium salts were obtained by reaction of P–H phosphonium salts with substituted 1,4-naphthoquinones. Some representatives of this series possess high activity against Gram-positive bacteria (Staphylococcus aureus ATCC 209p, Bacillus cereus ATCC 8035).
References
1.

Lambert C., Gaschler W., Nöll G., Weber M., Schmälzlin E., Bräuchle C., Meerholz K.
Journal of the Chemical Society Perkin Transactions 2,
2001
2.
10.1016/j.mencom.2017.03.008_bib0010
He
Synthesis,
2009
3.

Hallett J.P., Welton T.
Chemical Reviews,
2011
4.

Kagan V.E., Wipf P., Stoyanovsky D., Greenberger J.S., Borisenko G., Belikova N.A., Yanamala N., Samhan Arias A.K., Tungekar M.A., Jiang J.
Advanced Drug Delivery Reviews,
2009
5.

Wu T., Xie A., Tan S., Cai X.
Colloids and Surfaces B: Biointerfaces,
2011
6.

Xue Y., Xiao H., Zhang Y.
International Journal of Molecular Sciences,
2015
7.

Khasiyatullina N.R., Mironov V.F., Bogdanov A.V., Zobov V.V., Voloshina A.D., Kulik N.V., Konovalov A.I.
Pharmaceutical Chemistry Journal,
2009
8.

Strobykina I.Y., Belenok M.G., Semenova M.N., Semenov V.V., Babaev V.M., Rizvanov I.K., Mironov V.F., Kataev V.E.
Journal of Natural Products,
2015
9.

Millard M., Pathania D., Shabaik Y., Taheri L., Deng J., Neamati N.
PLoS ONE,
2010
10.

Spivak A.Y., Nedopekina D.A., Shakurova E.R., Khalitova R.R., Gubaidullin R.R., Odinokov V.N., Dzhemilev U.M., Bel’skii Y.P., Bel’skaya N.V., Stankevich S.A., Korotkaya E.V., Khazanov V.A.
Russian Chemical Bulletin,
2013
11.

Suin S., Shrivastava N.K., Maiti S., Khatua B.B.
Applied Clay Science,
2014
12.

Gómez R., Segura J.L., Martín N.
Journal of Organic Chemistry,
2000
13.

Beletskaya I.P., Kazankova M.A.
Russian Journal of Organic Chemistry,
2002
14.
Odinets I.L.
Organophosphorus Chemistry,
2010
15.

Arisawa M., Yamaguchi M.
ACS Symposium Series,
2007
16.
Khasiyatullina N.R., Mironov V.F., Bogdanov A.V., Krivolapov D.B., Litvinov I.A.
Mendeleev Communications,
2011
17.

Khasiyatullina N.R., Mironov V.F.
Russian Journal of Organic Chemistry,
2015
18.

Mironov V.F., Khasiyatullina N.R., Krivolapov D.B.
Tetrahedron Letters,
2015
19.
![Synthesis of the acidic dihydrogen complexes trans-[M(H2)(CN)L2]+ and trans-[M(H2)(CNH)L2]2+ where M = Fe, Ru, Os and L = dppm, dppe, dppp, depe, and dihydrogen substitution by the trifluoromethanesulfonate anion to give trans-[Ru(OTf )(CN)L2] or trans-[Ru(OTf )(CNH)L2]OTf †](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Fong T.P., Forde C.E., Lough A.J., Morris R.H., Rigo P., Rocchini E., Stephan T.
Journal of the Chemical Society Dalton Transactions,
1999
20.

Allen F.H., Kennard O., Watson D.G., Brammer L., Orpen A.G., Taylor R.
Journal of the Chemical Society Perkin Transactions 2,
1987
21.
![Self-assembling systems based on quaternized derivatives of 1,4-diazabicyclo[2.2.2]octane in nutrient broth as antimicrobial agents and carriers for hydrophobic drugs](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Pashirova T.N., Lukashenko S.S., Zakharov S.V., Voloshina A.D., Zhiltsova E.P., Zobov V.V., Souto E.B., Zakharova L.Y.
Colloids and Surfaces B: Biointerfaces,
2015
22.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007
23.
10.1016/j.mencom.2017.03.008_bib0070
Sheldrick
SHELXL, Version 2014/7,
2014
24.

Farrugia L.J.
Journal of Applied Crystallography,
1999
25.
10.1016/j.mencom.2017.03.008_bib0080
APEX (Version 2.1), SAINTPlus (Version 7.31A), Bruker Advanced X-ray Solutions,
2006
26.

Spek A.L.
Journal of Applied Crystallography,
2003
27.

Macrae C.F., Bruno I.J., Chisholm J.A., Edgington P.R., McCabe P., Pidcock E., Rodriguez-Monge L., Taylor R., van de Streek J., Wood P.A.
Journal of Applied Crystallography,
2008
28.
10.1016/j.mencom.2017.03.008_sbref0095a
Methods for Dilution Antimicrobial Susceptibility. Tests for Bacteria that Grow Aerobically. Approved Standard M7-A5,
2000
29.
10.1016/j.mencom.2017.03.008_sbref0095b
Reference Method for Broth Dilution Antifungal Susceptibility Testing of Conidium-Forming Filamentous Fungi. Proposed Standard M38-P,
1998
30.

Semenov V.E., Mikhailov A.S., Voloshina A.D., Kulik N.V., Nikitashina A.D., Zobov V.V., Kharlamov S.V., Latypov S.K., Reznik V.S.
European Journal of Medicinal Chemistry,
2011