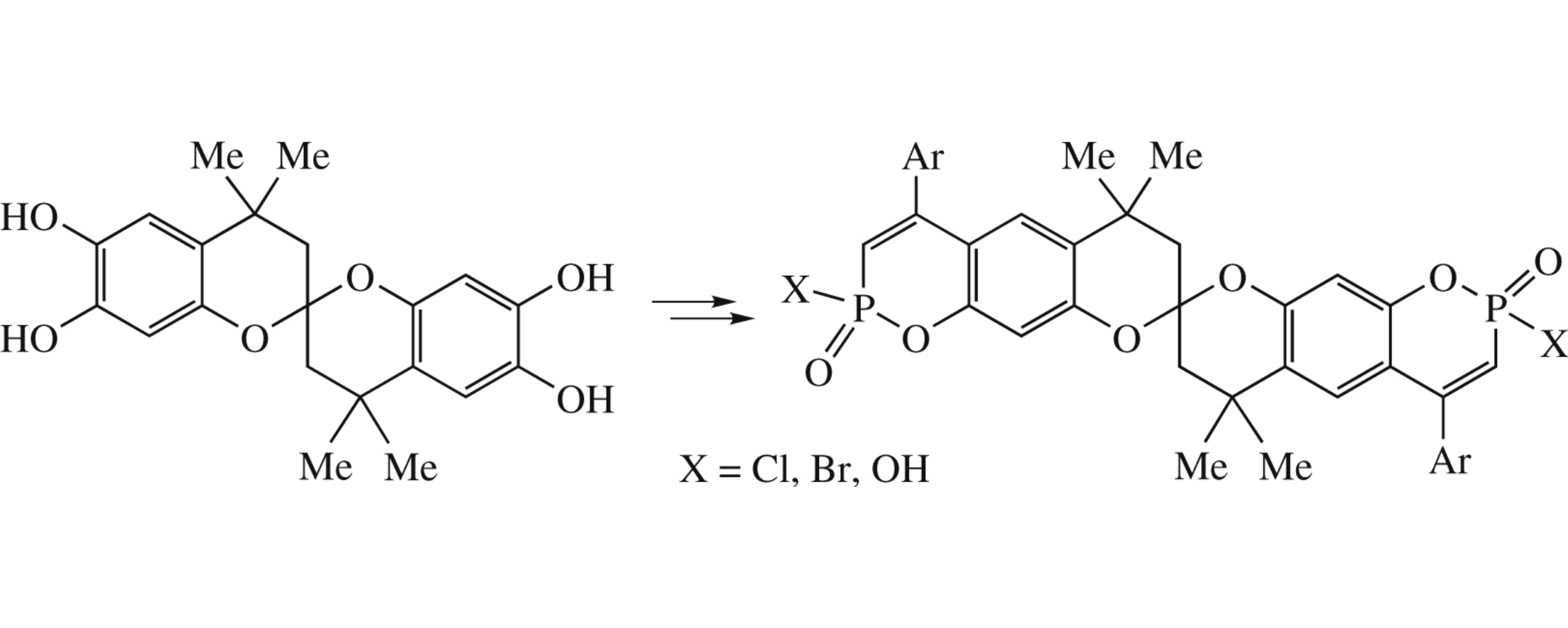
Abstract
8,8’-Spirobi(chromano-1,2-oxaphosphinines) were obtained by the reaction between phosphorylated derivatives of spirodichromane and arylacetylenes with a high chemoselectivity.
References
1.
10.1016/j.mencom.2017.03.007_bib0005
Nifant’ev
Dokl. Chem.,
1997
2.

Fritsch D., Bengtson G., Carta M., McKeown N.B.
Macromolecular Chemistry and Physics,
2011
3.

Gryko D.T., Pia̧tek P., Jurczak J.
Tetrahedron,
1997
4.

McKeown N.B., Makhseed S., Budd P.M.
Chemical Communications,
2002
5.
Tuktarov A.R., Khalilov L.M., Babievskii K.K., Akhmetov A.R., Sokolov V.I., Dzhemilev U.M.
Mendeleev Communications,
2015
6.

Audorff H., Walker R., Kador L., Schmidt H.
Chemistry - A European Journal,
2011
7.

Mironov V.F., Zyablikova T.A., Konovalova I.V., Khanipova M.G., Musin R.A.
Russian Chemical Bulletin,
1997
8.

Li B., Zhou B., Lu H., Ma L., Peng A.
European Journal of Medicinal Chemistry,
2010
9.

Baker W., Besly D.M.
Journal of the Chemical Society (Resumed),
1939
10.

Mironov V.F., Gubaidullin A.T., Petrov R.R., Litvinov I.A., Shtyrlina A.A., Zyablikova T.A., Azancheev N.M., Konovalov A.I., Musin R.Z.
Phosphorus, Sulfur and Silicon and the Related Elements,
1999
11.

Mironov V.F., Nemtarev A.V.
Review Journal of Chemistry,
2011