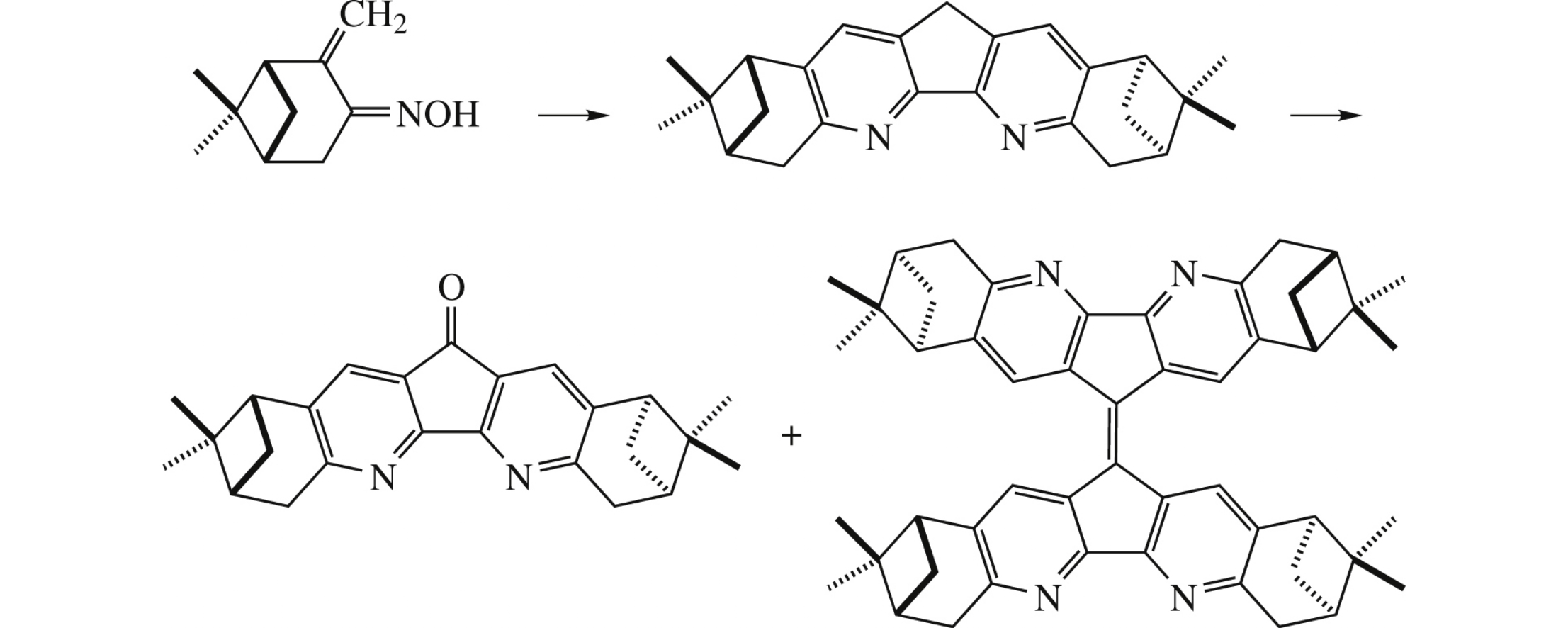
Abstract
Reaction of (+)-pinocarvone oxime with 2-morpholinocyclopent-2-enone and FeCl3·6H2O affords chiral C2-symmetric fused derivative of 4,5-diazafluorene, whose oxidation with SeO2 gives nopinane-annelated derivatives of 4,5-diaza-9Hfluoren- 9-one (C2-symmetry) and of 9,9’-bi-4,5-diazafluorenylidene (D2-symmetry). Structures of the compounds synthesized were proved by X-ray crystallography.
References
1.

Mamula O.
Coordination Chemistry Reviews,
2003
2.

Chelucci G., Thummel R.P.
Chemical Reviews,
2002
3.

Argent S.P., Adams H., Riis-Johannessen T., Jeffery J.C., Harding L.P., Mamula O., Ward M.D.
Inorganic Chemistry,
2006
4.

Chen J., Takenaka N.
Chemistry - A European Journal,
2009
5.

Denmark S.E., Fan Y.
Tetrahedron Asymmetry,
2006
6.

Malkov A.V., Bell M., Castelluzzo F., Kočovský P.
Organic Letters,
2005
7.

Malkov A.V., Orsini M., Pernazza D., Muir K.W., Langer V., Meghani P., Kočovský P.
Organic Letters,
2002
8.

Malkov A.V., Kočovský P.
European Journal of Organic Chemistry,
2006
9.

Vasilyev E.S., Agafontsev A.M., Tkachev A.V.
Synthetic Communications,
2014
10.

Kokina T.E., Glinskaya L.A., Tkachev A.V., Plyusnin V.F., Tsoy Y.V., Bagryanskaya I.Y., Vasilyev E.S., Piryazev D.A., Sheludyakova L.A., Larionov S.V.
Polyhedron,
2016
11.

Chibiryaev A.M., De Kimpe N., Tkachev A.V.
Tetrahedron Letters,
2000
12.

Sato K., Inoue S., Kitagawa T., Takahashi T.
Journal of Organic Chemistry,
1973
13.
Vasilyev E.S., Agafontsev A.M., Kolesnik V.D., Gatilov Y.V., Tkachev A.V.
Mendeleev Communications,
2011
14.
![Synthesis, Structure and Chemistry of a Twisted Olefinic Bis-didentate Proligand: 5,5′-Bi-5H-cyclopenta[2,1-b : 3,4-b′]dipyridinylidene](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Riklin M., von Zelewsky A., Bashall A., McPartlin M., Baysal A., Connor J., Wallis J.
Helvetica Chimica Acta,
1999
15.

Caldwell A.G.
Journal of the Chemical Society (Resumed),
1952
16.
10.1016/j.mencom.2017.03.006_bib0050
Sheldrick
SHELX-97, Programs for Crystal Structure Analysis (Release 97-2),
1997
17.
10.1016/j.mencom.2017.03.006_sbref0055a
Spek
PLATON, A Multipurpose Crystallographic Tool (Version 10M),
2003
18.

Spek A.L.
Journal of Applied Crystallography,
2003