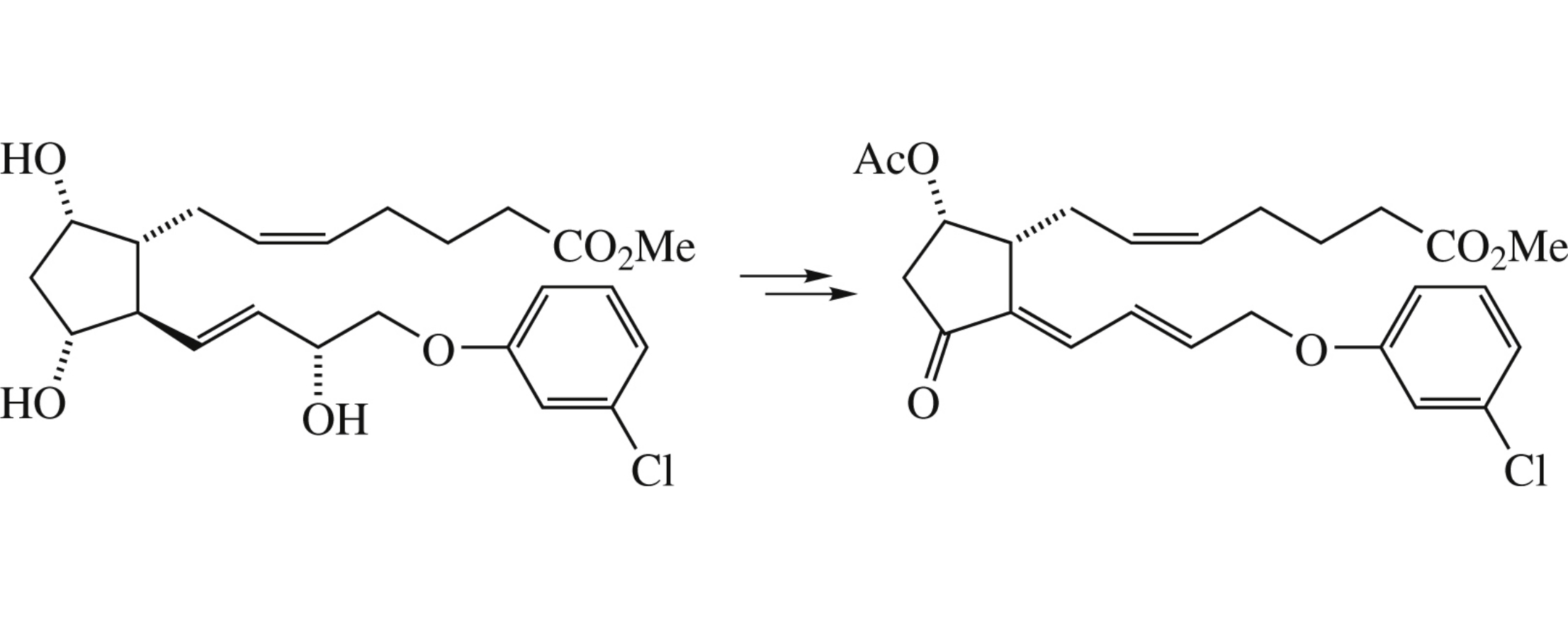
Abstract
The title compound, a possible precursor of metabolically stable corresponding PGJ2, was obtained from cloprostenol in seven steps. The compound demonstrated cytotoxicity against the HEK 293, Hep G2 and Jurkat cell lines.
References
1.

Straus D.S., Glass C.K.
Medicinal Research Reviews,
2001
2.

Fukushima S., Kishimoto S., Takeuchi Y., Fukushima M.
Advanced Drug Delivery Reviews,
2000
3.

Rossi A., Kapahi P., Natoli G., Takahashi T., Chen Y., Karin M., Santoro M.G.
Nature,
2000
4.

Pande V., Ramos M.J.
Bioorganic and Medicinal Chemistry Letters,
2005
5.

Suzuki M., Mori M., Niwa T., Hirata R., Furuta K., Ishikawa T., Noyori R.
Journal of the American Chemical Society,
1997
6.

Furuta K., Tomokiyo K., Satoh T., Watanabe Y., Suzuki M.
ChemBioChem,
2000
7.

Shibata T., Kondo M., Osawa T., Shibata N., Kobayashi M., Uchida K.
Journal of Biological Chemistry,
2002
8.

Forman B.M., Tontonoz P., Chen J., Brun R.P., Spiegelman B.M., Evans R.M.
Cell,
1995
9.

Satoh T., Furuta K., Suzuki M., Watanabe Y.
Biochemical and Biophysical Research Communications,
1999
10.

Fitzpatrick F.A., Wynalda M.A.
Journal of Biological Chemistry,
1983
11.

Kitade M., Tanaka H., Oe S., Iwashima M., Iguchi K., Takahashi T.
Chemistry - A European Journal,
2006
12.

Tanaka H., Hasegawa T., Kita N., Nakahara H., Shibata T., Oe S., Ojika M., Uchida K., Takahashi T.
Chemistry - An Asian Journal,
2006
13.

Fukushima S., Takeuchi Y., Kishimoto S., Yamashita S., Uetsuki K., Shirakawa S., Suzuki M., Furuta K., Noyori R., Sasaki H., Kikuchi Y., Kita T., Yamori T., Sawada J., Kojima M., et. al.
Anti-Cancer Drugs,
2003
14.

Furuta K., Maeda M., Hirata Y., Shibata S., Kiuchi K., Suzuki M.
Bioorganic and Medicinal Chemistry Letters,
2007
15.

Żurawiński R., Mikołajczyk M., Cieślak M., Królewska K., Kaźmierczak-Barańska J.
Organic and Biomolecular Chemistry,
2015
16.

Vostrikov N.S., Lobko I.F., Miftakhov M.S.
Tetrahedron Letters,
2014
17.
G. A. Tolstikov, M.S. Miftakhov, N.S. Vostrikov, O.M. Kuznetsov, V. Kh. Singizova, E.K. Starostin, M.E. Adler, F.A. Valeev and N. A. Danilova, Patent SU 1512065 A1, 1989.
18.
10.1016/j.mencom.2017.03.005_bib0090
Tolstikov
Zh. Org. Khim.,
1992
19.

Mosmann T.
Journal of Immunological Methods,
1983