Abstract
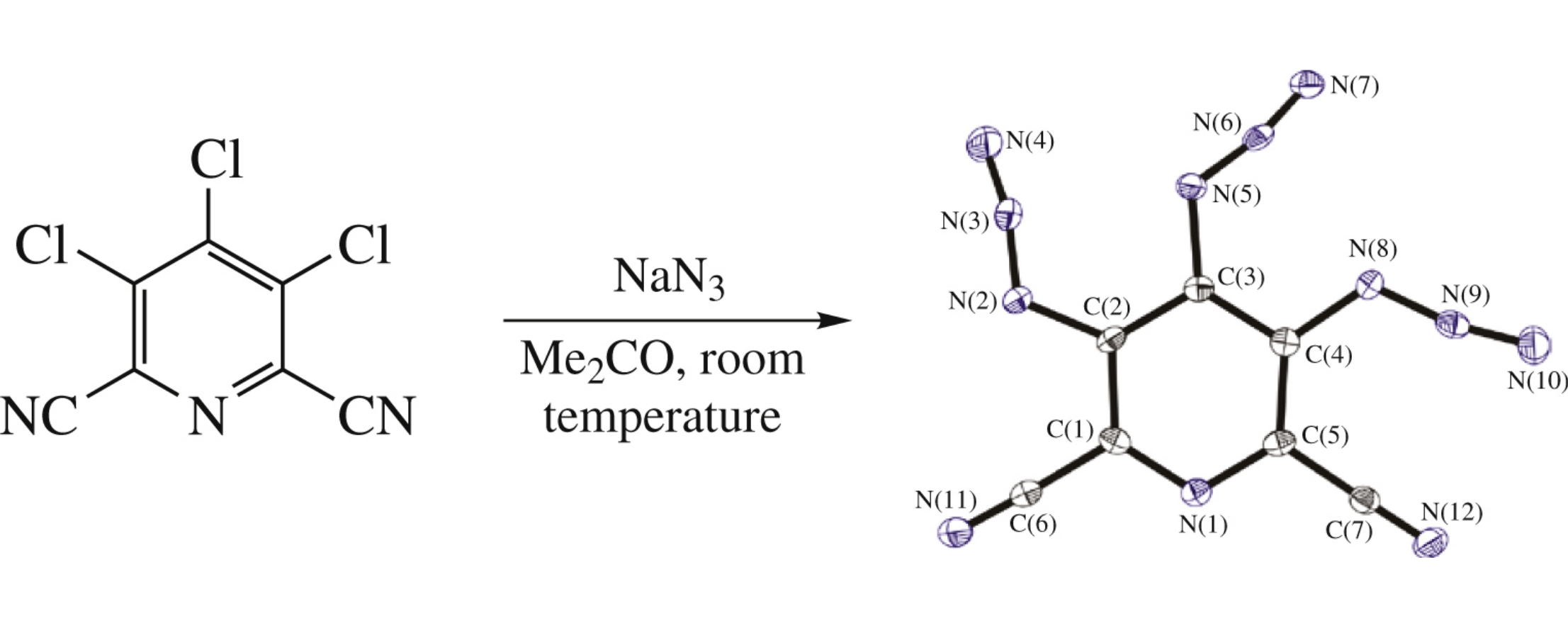
3,4,5-Triazidopyridine-2,6-dicarbonitrile, the first aromatic triazide with three adjacent azido groups in the ring, possessing the record positive heat of formation, was synthesized by azidation of 3,4,5-trichloropyridine-2,6-dicarbonitrile and characterized with X-ray analysis, thermogravimetry, differential scanning calorimetry and spectroscopic methods.
References
1.

Chapyshev S.
Molecules,
2015
2.

Finger H.
Journal für praktische Chemie,
1907
3.

4.

M. Heravi M., Tamimi M., Yahyavi H., Hosseinnejad T.
Current Organic Chemistry,
2015
5.

Hein J.E., Fokin V.V.
Chemical Society Reviews,
2010
6.
10.1016/j.mencom.2017.03.002_sbref0100c
Chapyshev
Synlett,
2009
7.

Meldal M., Tornøe C.W.
Chemical Reviews,
2008
8.

9.

Bräse S., Gil C., Knepper K., Zimmermann V.
Angewandte Chemie - International Edition,
2005
10.

Scriven E.F., Turnbull K.
Chemical Reviews,
1988
11.

Gillan E.G.
Chemistry of Materials,
2000
12.

Kroke E., Schwarz M., Buschmann V., Miehe G., Fuess H., Riedel R.
Advanced Materials,
1999
13.

Utschig T., Schwarz M., Miehe G., Kroke E.
Carbon,
2004
14.

Sato T., Narazaki A., Kawaguchi Y., Niino H., Bucher G., Grote D., Wolff J.J., Wenk H.H., Sander W.
Journal of the American Chemical Society,
2004
15.

Wasserman E., Schueller K., Yager W.A.
Chemical Physics Letters,
1968
16.

Chapyshev S.V., Ushakov E.N., Neuhaus P., Sander W.
Journal of Organic Chemistry,
2014
17.

Chapyshev S.V., Walton R., Sanborn J.A., Lahti P.M.
Journal of the American Chemical Society,
2000
18.

Chapyshev S.V., Grote D., Finke C., Sander W.
Journal of Organic Chemistry,
2008
19.

Misochko E.Y., Akimov A.V., Chapyshev S.V.
Journal of Chemical Physics,
2008
20.

Chapyshev S.V., Neuhaus P., Grote D., Sander W.
Journal of Physical Organic Chemistry,
2009
21.

Misochko E.Y., Akimov A.V., Masitov A.A., Korchagin D.V., Yakushchenko I.K., Chapyshev S.V.
Journal of Chemical Physics,
2012
22.

Chapyshev S.V., Korchagin D.V., Neuhaus P., Sander W.
Beilstein Journal of Organic Chemistry,
2013
23.

Chapyshev S.V., Misochko E.Y., Akimov A.V., Dorokhov V.G., Neuhaus P., Grote D., Sander W.
Journal of Organic Chemistry,
2009
24.

Misochko E.Y., Akimov A.V., Masitov A.A., Korchagin D.V., Aldoshin S.M., Chapyshev S.V.
Journal of Chemical Physics,
2013
25.

Misochko E.Y., Masitov A.A., Akimov A.V., Korchagin D.V., Chapyshev S.V.
Journal of Physical Chemistry A,
2014
26.

Akimov A., Masitov A., Korchagin D., Chapyshev S., Misochko E., Savitsky A.
Journal of Chemical Physics,
2015
27.
10.1016/j.mencom.2017.03.002_sbref0060a
Matyáš
Primary Explosives,
2013
28.

Yan Q., Zeman S., Zhang J., Qi X., Li T., Musil T.
Journal of Physical Chemistry C,
2015
29.

Huynh M.V., Hiskey M.A., Hartline E.L., Montoya D.P., Gilardi R.
Angewandte Chemie - International Edition,
2004
30.

Mikhailov Y.M., Chapyshev S.V., Nedel’ko V.V.
Russian Chemical Bulletin,
2009
31.

Goralski C.T., Evans T.E.
Journal of Organic Chemistry,
1975
32.

Kuzina S.I., Korchagin D.V., Shilov G.V., Chapyshev S.V., Mikhailov A.I., Aldoshin S.M.
Doklady Physical Chemistry,
2008
33.

Chapyshev S.V., Chernyak A.V., Ushakov E.N.
Magnetic Resonance in Chemistry,
2016
34.

Montgomery J.A., Frisch M.J., Ochterski J.W., Petersson G.A.
Journal of Chemical Physics,
2000
35.

Göbel M., Klapötke T.M.
Advanced Functional Materials,
2009
36.

Pan Y., Zhu W., Xiao H.
Journal of Molecular Modeling,
2012
37.
10.1016/j.mencom.2017.03.002_bib0095
Sheldrick
SHELXTL v. 6.14 Structure Determination Software Suite,
2000