Abstract
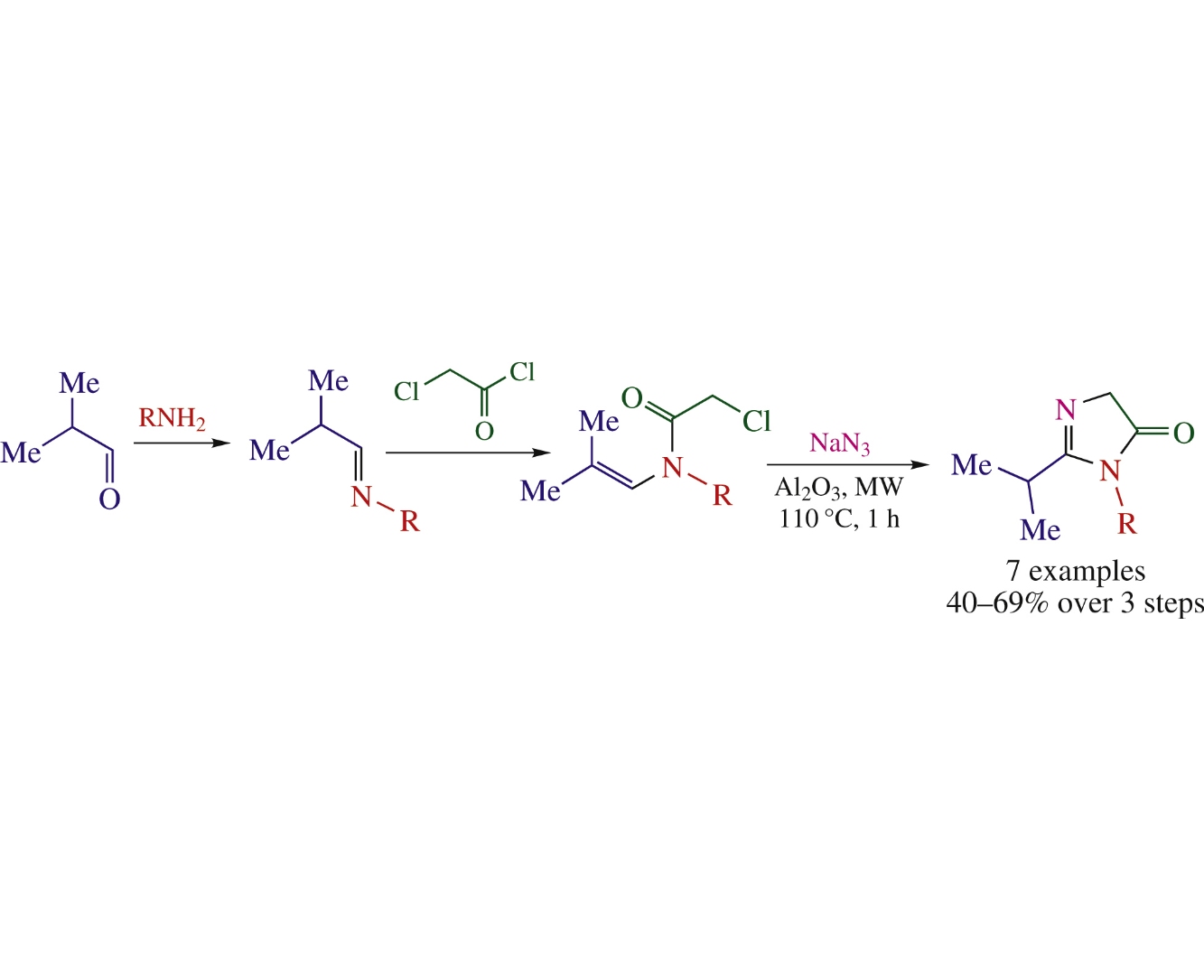
A novel method to prepare biologically relevant 1H-imidazol-5(4H)-ones from aliphatic amines, isobutyraldehyde, chloroacetyl chloride and sodium azide under microwave irradiation has been developed.
References
1.

Dömling A., Ugi I.
Angewandte Chemie - International Edition,
2000
2.
10.1016/j.mencom.2017.01.031_bib0010
Ugi
Angew. Chem.,
1959
3.

Biginelli P.
Berichte der deutschen chemischen Gesellschaft,
1891
4.

Hantzsch A.
Berichte der deutschen chemischen Gesellschaft,
1881
5.

Krasavin M., Dar’in D.
Tetrahedron Letters,
2016
6.

Sharma U.K., Sharma N., Vachhani D.D., Van der Eycken E.V.
Chemical Society Reviews,
2015
7.

Ivachtchenko A.V., Ivanenkov Y.A., Kysil V.M., Krasavin M.Y., Ilyin A.P.
Russian Chemical Reviews,
2010
8.
![Convenient access to novel functionalized pyrazino[1,2-b]isoquinolin-6-one and diazepino[1,2-b]isoquinolin-7-one scaffolds via the Cushman multicomponent reaction followed by post-condensation](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
9.

Cook G.R., Barta N.S., Stille J.R.
Journal of Organic Chemistry,
1992
10.

Padwa A., Heidelbaugh T.M., Kuethe J.T., McClure M.S., Wang Q.
Journal of Organic Chemistry,
2002
11.

Lesniak S., Pasternak B.
Synthetic Communications,
2002
12.
![Rhodium-catalysed hydroformylation of N-(2-propenyl)-β-lactams as a key step in the synthesis of functionalised N-[4-(2-oxoazetidin-1-yl)but-1-enyl]acetamides](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Dekeukeleire S., D'hooghe M., Müller C., Vogt D., De Kimpe N.
New Journal of Chemistry,
2010
13.
![Synthesis of 1-substituted 1,2,3,4-tetrahydrobenz[g]isoquinoline-5,10-diones](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Shinkevich E., Deblander J., Matthijs S., Jacobs J., De Kimpe N., Tehrani K.A.
Organic and Biomolecular Chemistry,
2011
14.

Kulyashova A., Krasavin M.
Tetrahedron Letters,
2016
15.

Huisgen R., Szeimies G., Möbius L.
Chemische Berichte,
1967
16.
10.1016/j.mencom.2017.01.031_bib0080
Moonen
Synthesis,
2005
17.

Mooney C.R., Horke D.A., Chatterley A.S., Simperler A., Fielding H.H., Verlet J.R.
Chemical Science,
2013
18.

Baranov M.S., Solntsev K.M., Baleeva N.S., Mishin A.S., Lukyanov S.A., Lukyanov K.A., Yampolsky I.V.
Chemistry - A European Journal,
2014
19.

Huang H., Suslov N.B., Li N., Shelke S.A., Evans M.E., Koldobskaya Y., Rice P.A., Piccirilli J.A.
Nature Chemical Biology,
2014
20.

Rogers T.A., Andrews G.E., Jaeger L., Grabow W.W.
ACS Synthetic Biology,
2014
21.

Takeuchi H., Hagiwara S., Eguchi S.
Tetrahedron,
1989
22.

Ortiz Barbosa Y.A., Hart D.J., Magomedov N.A.
Tetrahedron,
2006
23.

Jones L.H., Dupont T., Mowbray C.E., Newman S.D.
Organic Letters,
2006
24.

Rottmann A., Liebscher J.
Journal of Heterocyclic Chemistry,
1996
25.
![Hydroxylation of 5H-3-Oxo-2,3,6,7,8,9-hexahydroimidazo[1,2-a]azepine derivatives by molecular oxygen](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Hydroxylation of 5H-3-Oxo-2,3,6,7,8,9-hexahydroimidazo[1,2-a]azepine derivatives by molecular oxygen
Campagna F., Carotti A., Casini G.
Journal of Heterocyclic Chemistry,
1990
26.

Lee C., Chen Y., Lin H., Jhong Y., Chang C., Tsai C., Kao C., Chien T.
Tetrahedron,
2012
27.
López H.S., Enciso J.E., Ochoa-Terán A., Velazquez J.I., Sarmiento J.I.
Mendeleev Communications,
2016