Abstract
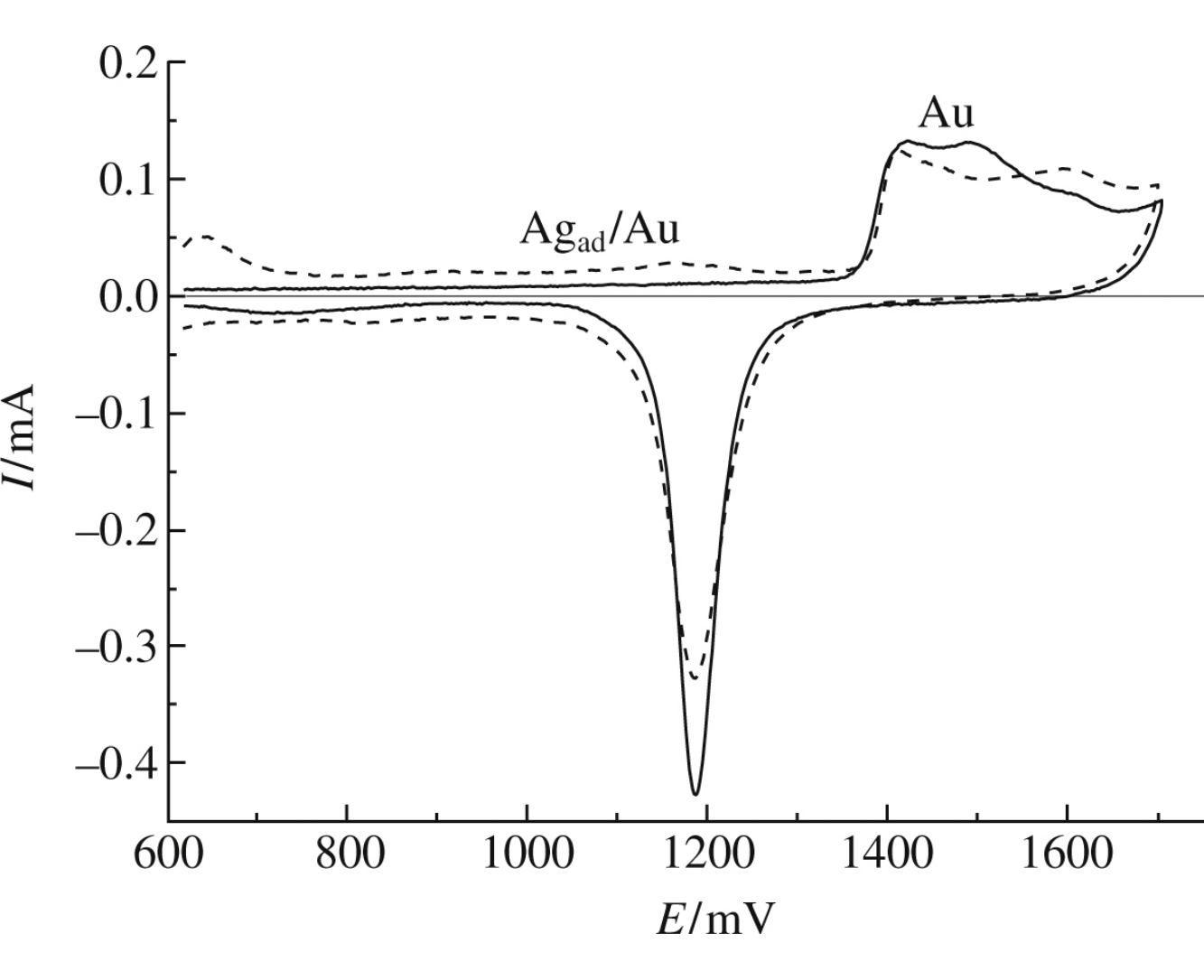
Optimal conditions are found for estimating the electrochemically active surface area of polycrystalline gold based on a monolayer coverage with Agad.
References
1.

Trasatti S., Petrii O.A.
Journal of Electroanalytical Chemistry,
1992
2.
A. A. Michri, A.G. Pshchenichnikov and R. K. Burstein, Sov. Electrochem., 1972, 8, 351 (Elektrokhimiya, 1972, 8, 364).
3.
R. Woods, in Electroanalytical Chemistry, ed. A. J. Bard, Marcel Dekker, New York, 1976, vol. 9.
4.

Swathirajan S., Mizota H., Bruckenstein S.
The Journal of Physical Chemistry,
1982
5.

Santos M.C., Mascaro L.H., Machado S.A.
Electrochimica Acta,
1998
6.

Podlovchenko B.I., Maksimov Y.M., Maslakov K.I.
Electrochimica Acta,
2014
7.

Rand D.A., Woods R.
Journal of Electroanalytical Chemistry and Interfacial Electrochemistry,
1972
8.

Perdriel C.L., Arvia A.J., Ipohorski M.
Journal of Electroanalytical Chemistry and Interfacial Electrochemistry,
1986
9.

Chen C.H., Vesecky S.M., Gewirth A.A.
Journal of the American Chemical Society,
1992
10.

Mrozek P., Sung Y., Han M., Gamboa-aldeco M., Wieckowski A., Chen C., Gewirth A.A.
Electrochimica Acta,
1995
11.

Ogaki K., Itaya K.
Electrochimica Acta,
1995
12.
Podlovchenko B.I., Gladysheva T.D., Filatov A.Y.
Mendeleev Communications,
2015
13.
D. M. Kolb, in Advances in Electrochemistry and Electrochemical Engineering, eds. H. Gerischer and C. W. Tobias, Wiley, New York, 1978, vol. 11, p. 125.
14.

Hamelin A.
Journal of Electroanalytical Chemistry and Interfacial Electrochemistry,
1986
15.

Hamelin A.
Journal of Electroanalytical Chemistry and Interfacial Electrochemistry,
1988
16.

Manzhos R.A., Krivenko A.G., Doronin S.V., Choba M.A., Safonov V.A.
Journal of Electroanalytical Chemistry,
2013
17.

Safonov V.A., Choba M.A., Krivenko A.G., Manzhos R.A., Maksimov Y.M.
Electrochimica Acta,
2012
18.

Herrero E., Buller L.J., Abruña H.D.
Chemical Reviews,
2001
19.

Yu Y., Hu Y., Liu X., Deng W., Wang X.
Electrochimica Acta,
2009
20.

Horányi G., Rizmayer E.M., Joó P.
Journal of Electroanalytical Chemistry and Interfacial Electrochemistry,
1983
21.

Juodkazis K., Juodkazyt J., Šebeka B., Lukinskas A.
Electrochemistry Communications,
1999