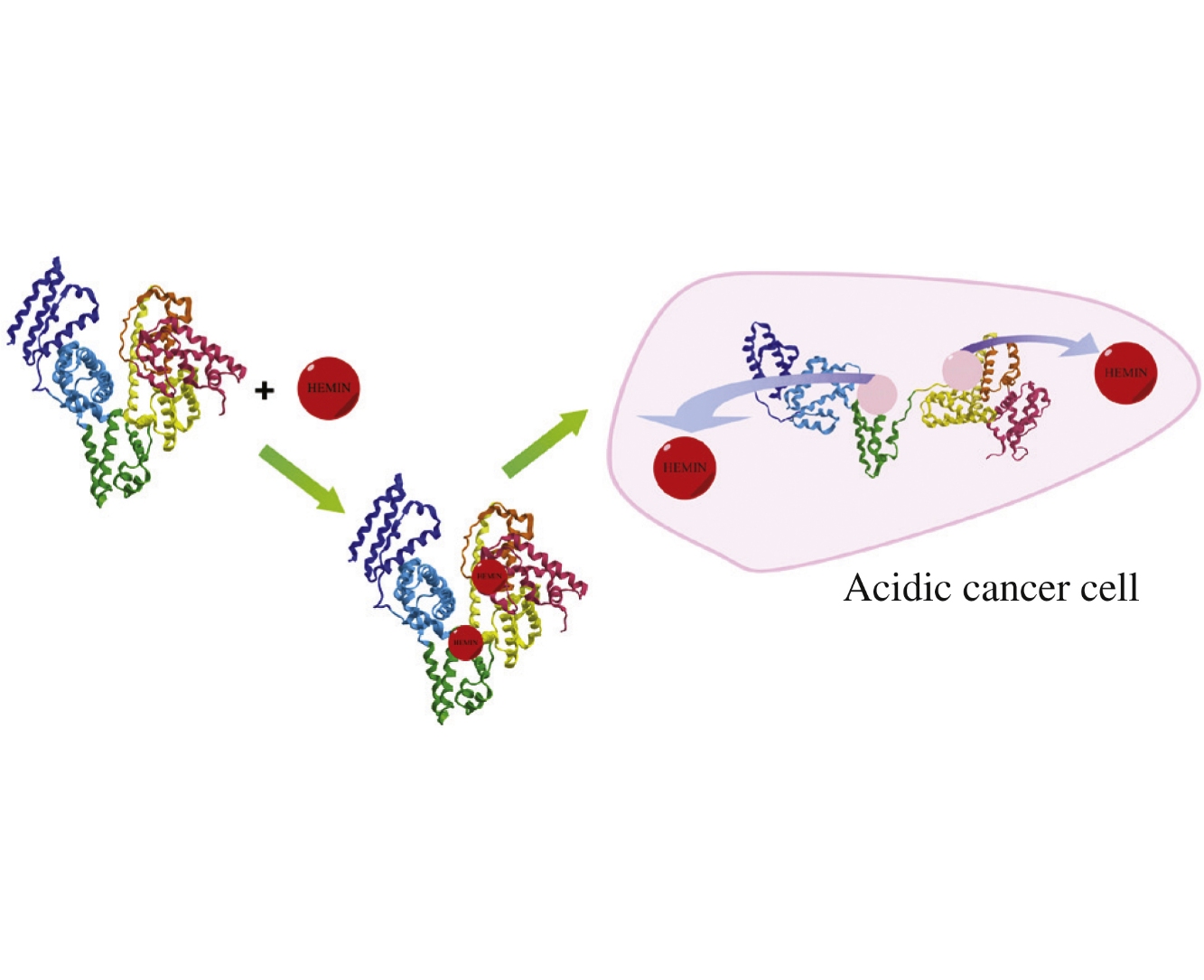
Abstract
The possibility of creating a pH-controllable protein container for the delivery of hydrophobic porphyrins as photosensitizers in cancer cells has been demonstrated.
References
1.

Inguscio V., Panzarini E., Dini L.
Cells,
2012
2.

Moan J., Berg K., Bommer J.C., Western A.
Photochemistry and Photobiology,
1992
3.

Deda D., Pavani C., Caritá E., Baptista M., Toma H., Araki K.
Journal of Biomedical Nanotechnology,
2013
4.

Pavani C., Uchoa A.F., Oliveira C.S., Iamamoto Y., Baptista M.S.
Photochemical and Photobiological Sciences,
2009
5.

Uchoa A.F., Oliveira C.S., Baptista M.S.
Journal of Porphyrins and Phthalocyanines,
2010
6.
Glidden M.D., Celli J.P., Massodi I., Rizvi I., Pogue B.W., Hasan T.
Theranostics,
2012
7.

Celli J.P., Solban N., Liang A., Pereira S.P., Hasan T.
Lasers in Surgery and Medicine,
2011
8.

Kessel D., Vicente M.G., Reiners J.J.
Lasers in Surgery and Medicine,
2006
9.

Kessel D., Castelli M., Reiners J.J.
Cell Death and Differentiation,
2005
10.

Bacellar I., Tsubone T., Pavani C., Baptista M.
International Journal of Molecular Sciences,
2015
11.
![Greatly enhanced binding of a cationic porphyrin towards bovine serum albumin by cucurbit[8]uril](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Lei W., Jiang G., Zhou Q., Zhang B., Wang X.
Physical Chemistry Chemical Physics,
2010
12.

Sheng N., Zong S., Cao W., Jiang J., Wang Z., Cui Y.
ACS applied materials & interfaces,
2015
13.

Bragina N.A., Zhdanova K.A., Mironov A.F.
Russian Chemical Reviews,
2016
14.

Singh S., Aggarwal A., Bhupathiraju N.V., Arianna G., Tiwari K., Drain C.M.
Chemical Reviews,
2015
15.

Baldassarre F., Foglietta F., Vergaro V., Barbero N., Capodilupo A.L., Serpe L., Visentin S., Tepore A., Ciccarella G.
Journal of Photochemistry and Photobiology B: Biology,
2016
16.
Lebedeva N.S., Gubarev Y.A., Koifman O.I.
Mendeleev Communications,
2015
17.

Lebedeva N.S., Gubarev Y.A., Vyugin A.I., Koifman O.I.
Journal of Luminescence,
2015
18.

Curry S., Mandelkow H., Brick P., Franks N.
Nature Structural Biology,
1998
19.

Carter D.C., Ho J.X.
Advances in Protein Chemistry,
1994
20.
10.1016/j.mencom.2017.01.014_bib0100
Peters
All About Albumin: Biochemistry, Genetics, and Medical Applications,
1995
21.

Spiro T.G., Strekas T.C.
Journal of the American Chemical Society,
1974
22.
10.1016/j.mencom.2017.01.014_bib0110
Lebedeva
Uchenye Zapiski Petrozavodskogo Gosudarstvennogo Universiteta,
2014
23.

Ascenzi P., Bocedi A., Notari S., Menegatti E., Fasano M.
Biochemical and Biophysical Research Communications,
2005
24.

25.

Baroni S., Mattu M., Vannini A., Cipollone R., Aime S., Ascenzi P., Fasano M.
FEBS Journal,
2001
26.

Zunszain P.A., Ghuman J., Komatsu T., Tsuchida E., Curry S.
BMC Structural Biology,
2003
27.

28.
10.1016/j.mencom.2017.01.014_bib0140
Beynon
Buffer Solutions: The Basics,
1996
29.

Guo M., Zou J., Yi P., Shang Z., Hu G., Yu Q.
Analytical Sciences,
2004