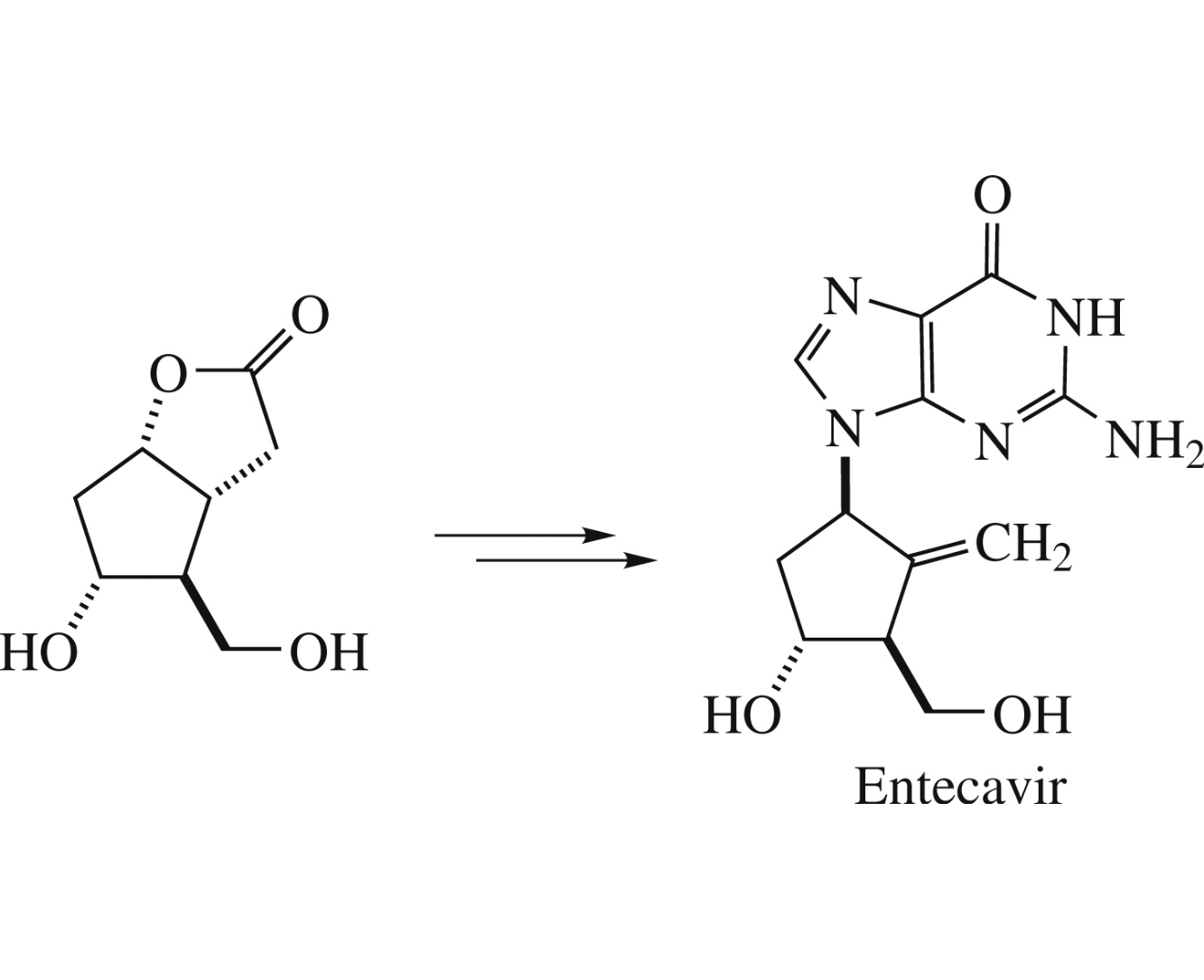
Abstract
Racemic Entecavir was prepared from Corey lactone diol benzylidene acetal in four steps in 24% overall yield.
References
1.

Yang Q., Kang J., Zheng L., Wang X., Wan N., Wu J., Qiao Y., Niu P., Wang S., Peng Y., Wang Q., Yu W., Chang J.
Journal of Medicinal Chemistry,
2015
2.

Bisacchi G.S., Chao S.T., Bachard C., Daris J.P., Innaimo S., Jacobs G.A., Kocy O., Lapointe P., Martel A., Merchant Z., Slusarchyk W.A., Sundeen J.E., Young M.G., Colonno R., Zahler R., et. al.
Bioorganic and Medicinal Chemistry Letters,
1997
3.

Innaimo S.F., Seifer M., Bisacchi G.S., Standring D.N., Zahler R., Colonno R.J.
Antimicrobial Agents and Chemotherapy,
1997
4.

5.

Zhou B., Li Y.
Tetrahedron Letters,
2012
6.

Liu X., Jiao X., Wu Q., Tian C., Li R., Xie P.
Tetrahedron Letters,
2012
7.

Velasco J., Ariza X., Badía L., Bartra M., Berenguer R., Farràs J., Gallardo J., Garcia J., Gasanz Y.
Journal of Organic Chemistry,
2013
8.

Ziegler F.E., Sarpong M.A.
Tetrahedron,
2003
9.

Ruediger E., Martel A., Meanwell N., Solomon C., Turmel B.
Tetrahedron Letters,
2004
10.

Campian M., Putala M., Sebesta R.
Current Organic Chemistry,
2014
11.

Carbocyclic nucleoside analogues: classification, target enzymes, mechanisms of action and synthesis
Matyugina E.S., Khandazhinskaya A.P., Kochetkov S.N.
Russian Chemical Reviews,
2012
12.
10.1016/j.mencom.2017.01.002_bib0025
Tolstikov
Zh. Org. Khim.,
1984
13.

Rigby J.H., Payen A., Warshakoon N.
Tetrahedron Letters,
2001
14.
Valiullina Z.R., Akhmet’yanova V.A., Vostrikov N.S., Miftakhov M.S.
Mendeleev Communications,
2016
15.
T.-C. Hu and H.-T. Huang, Patent US 20110201809 A1, 2011.
16.
D. Alberico, J. Clayton, C. Dixon and B. Gorin, Patent WO 2011150512 A1, 2011.
17.
D. Alberico, B. Gorin, R. Beharrilall, C. Dixon, J. Clayton and V. Rexon, Patent US 20130066071 A1, 2013.
18.
H. Kang and J. Chen, Faming Zhuanli Shenging, Patent CN 101693713 A2 0100414, 2010.
19.
Y. W. Ye, J. Yuan, J. Nie, D. Xu and C. Chen, Patent US 8937076 B2, 2015.
20.

Ros F., Molina M.T.
European Journal of Organic Chemistry,
1999
21.

Chickos J.S., Garin D.L., Hitt M., Schilling G.
Tetrahedron,
1981
22.
![[Physicochemical properties of suplatast tosilate racemate and enantiomers].](/storage/images/resized/KBGMujzjwr0rGFxg8Kz0qvYdFHkcBo5w0GTtfiiU_small_thumb.webp)
USHIO T., ENDO K., YAMAMOTO K.
Yakugaku Zasshi,
2017
23.

Vostrikov N.S., Vasikov V.Z., Miftakhov M.S.
Russian Journal of Organic Chemistry,
2004
24.

Wang X.J., Wiehler H., Ching C.B.
Journal of Chemical & Engineering Data,
2003
25.
10.1016/j.mencom.2017.01.002_sbref0050f
Navrátilová
Enantiomer,
2001
26.

Che Q., Quan P., Mu M., Zhang X., Zhao H., Zhang Y., You S., Xiao Y., Fang L.
Expert Opinion on Drug Delivery,
2014