Abstract
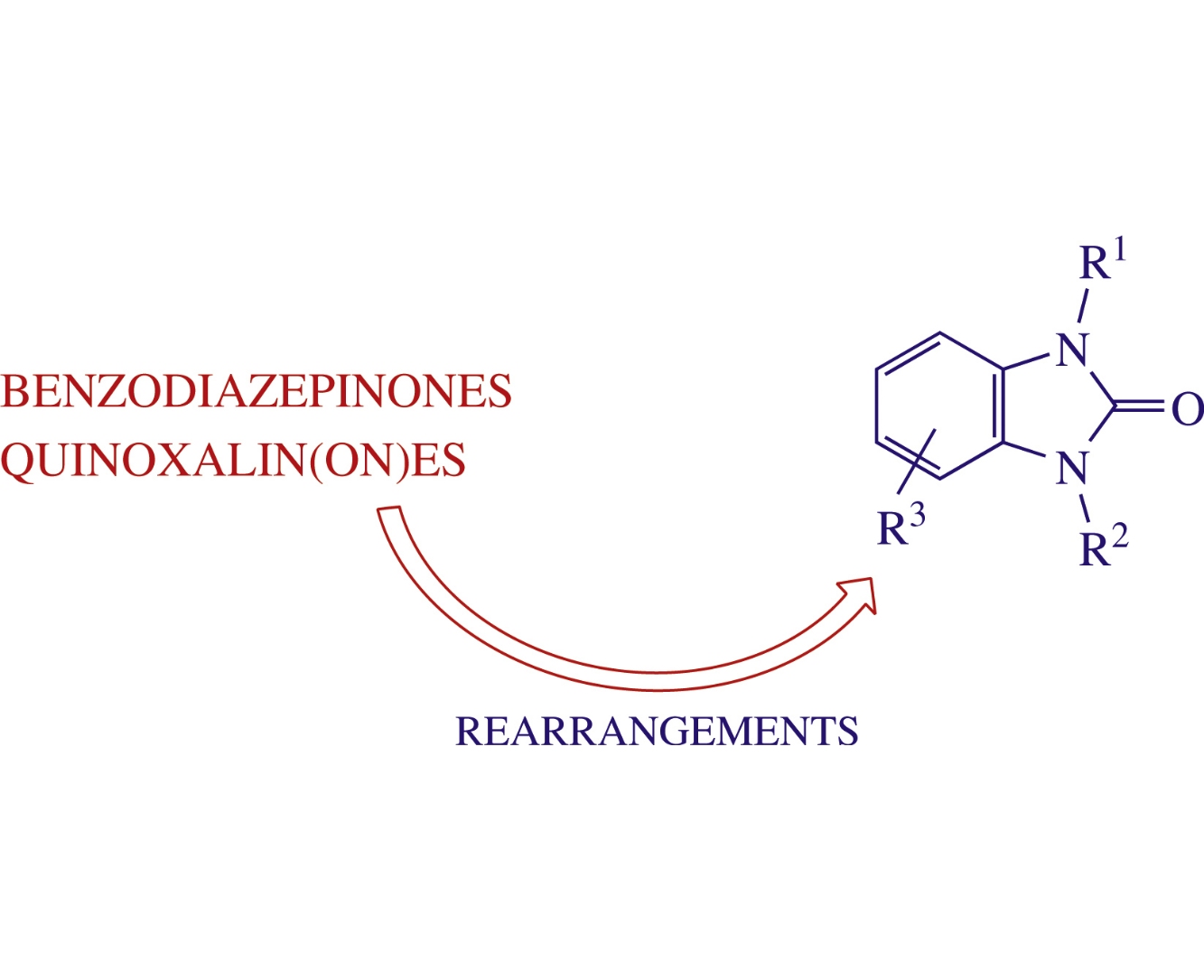
The review critically analyzes methods for the preparation of benzimidazolones proceeding via the rearrangements of benzodiazepinones and quinoxalin(on)es, highlights recent advances in the synthesis of bi- and tri-heterocyclic systems with the benzimidazolone moiety, and the advantages and major challenges in their biomedical applications.
References
1.

Theberge C.R., Bednar R.A., Bell I.M., Corcoran H.A., Fay J.F., Hershey J.C., Johnston V.K., Kane S.A., Mosser S., Salvatore C.A., Williams T.M., Zartman C.B., Zhang X., Graham S.L., Vacca J.P., et. al.
Bioorganic and Medicinal Chemistry Letters,
2008
2.

Wang W., Cao H., Wolf S., Camacho-Horvitz M.S., Holak T.A., Dömling A.
Bioorganic and Medicinal Chemistry,
2013
3.

Omura H., Kawai M., Shima A., Iwata Y., Ito F., Masuda T., Ohta A., Makita N., Omoto K., Sugimoto H., Kikuchi A., Iwata H., Ando K.
Bioorganic and Medicinal Chemistry Letters,
2008
4.

Palin R., Bom A., Clark J.K., Evans L., Feilden H., Houghton A.K., Jones P.S., Montgomery B., Weston M.A., Wishart G.
Bioorganic and Medicinal Chemistry,
2007
5.

Palin R., Clark J.K., Evans L., Houghton A.K., Jones P.S., Prosser A., Wishart G., Yoshiizumi K.
Bioorganic and Medicinal Chemistry,
2008
6.

Monforte A., Logoteta P., Luca L.D., Iraci N., Ferro S., Maga G., De Clercq E., Pannecouque C., Chimirri A.
Bioorganic and Medicinal Chemistry,
2010
7.
![Synthesis and SAR of 1-Hydroxy-1H-benzo[d]imidazol-2(3H)-ones as Inhibitors of d-Amino Acid Oxidase](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Berry J.F., Ferraris D.V., Duvall B., Hin N., Rais R., Alt J., Thomas A.G., Rojas C., Hashimoto K., Slusher B.S., Tsukamoto T.
ACS Medicinal Chemistry Letters,
2012
8.

Yu K., Wang X.A., Civiello R.L., Trehan A.K., Pearce B.C., Yin Z., Combrink K.D., Gulgeze H.B., Zhang Y., Kadow K.F., Cianci C.W., Clarke J., Genovesi E.V., Medina I., Lamb L., et. al.
Bioorganic and Medicinal Chemistry Letters,
2006
9.

Sivendran S., Jones V., Sun D., Wang Y., Grzegorzewicz A.E., Scherman M.S., Napper A.D., McCammon J.A., Lee R.E., Diamond S.L., McNeil M.
Bioorganic and Medicinal Chemistry,
2010
10.

Hammach A., Barbosa A., Gaenzler F.C., Fadra T., Goldberg D., Hao M., Kroe R.R., Liu P., Qian K.C., Ralph M., Sarko C., Soleymanzadeh F., Moss N.
Bioorganic and Medicinal Chemistry Letters,
2006
11.
10.1016/j.mencom.2017.01.001_bib0055
Vora
Der Pharma Chem.,
2010
12.

Liu W., Lau F., Liu K., Wood H.B., Zhou G., Chen Y., Li Y., Akiyama T.E., Castriota G., Einstein M., Wang C., McCann M.E., Doebber T.W., Wu M., Chang C.H., et. al.
Journal of Medicinal Chemistry,
2011
13.

Terefenko E.A., Kern J., Fensome A., Wrobel J., Zhu Y., Cohen J., Winneker R., Zhang Z., Zhang P.
Bioorganic and Medicinal Chemistry Letters,
2005
14.

Wang J.H., Hou Q.Q., Tang K., Cheng X.L., Dong L.H., Liu Y.J., Liu C.B.
SAR and QSAR in Environmental Research,
2011
15.

Guillaume M.
Organic Process Research and Development,
2006
16.

Li Q., Li T., Woods K.W., Gu W., Cohen J., Stoll V.S., Galicia T., Hutchins C., Frost D., Rosenberg S.H., Sham H.L.
Bioorganic and Medicinal Chemistry Letters,
2005
17.

Nardi Antonio, Calderone Vincenzo, Chericoni Silvio, Morelli Ivano
Planta Medica,
2003
18.

Meanwell N.A., Sit S., Gao J., Boissard C.G., Lum-Ragan J., Dworetzky S.I., Gribkoff V.K.
Bioorganic and Medicinal Chemistry Letters,
1996
19.

Kraska J., Boruszczak Z.
Dyes and Pigments,
1990
20.

Boruszczak Z., Kraska J.
Dyes and Pigments,
1994
21.

Baur R., Macholdt H.
Journal of Electrostatics,
1997
22.

Pacios L.F., Lazar Z., Benali B.
Journal of Molecular Structure THEOCHEM,
2002
23.

Shang X., Li B., Li C., Wang X., Zhang T., Jiang S.
Dyes and Pigments,
2013
24.

Bell I.M., Bednar R.A., Fay J.F., Gallicchio S.N., Hochman J.H., McMasters D.R., Miller-Stein C., Moore E.L., Mosser S.D., Pudvah N.T., Quigley A.G., Salvatore C.A., Stump C.A., Theberge C.R., Wong B.K., et. al.
Bioorganic and Medicinal Chemistry Letters,
2006
25.

Yu K., Sin N., Civiello R.L., Wang X.A., Combrink K.D., Gulgeze H.B., Venables B.L., Wright J.J., Dalterio R.A., Zadjura L., Marino A., Dando S., D’Arienzo C., Kadow K.F., Cianci C.W., et. al.
Bioorganic and Medicinal Chemistry Letters,
2007
26.

Kawamoto H., Nakashima H., Kato T., Arai S., Kamata K., Iwasawa Y.
Tetrahedron,
2001
27.

Miah A.H., Abas H., Begg M., Marsh B.J., O’Flynn D.E., Ford A.J., Percy J.M., Procopiou P.A., Richards S.A., Rumley S.
Bioorganic and Medicinal Chemistry,
2014
28.

Sams A.G., Larsen K., Mikkelsen G.K., Hentzer M., Christoffersen C.T., Jensen K.G., Frederiksen K., Bang-Andersen B.
Bioorganic and Medicinal Chemistry Letters,
2012
29.
10.1016/j.mencom.2017.01.001_bib0145
Cheeseman
Chemistry of Heterocyclic Compounds. Vol. 35. Condensed Pyrazines,
1979
30.

Mamedov V.A.
RSC Advances,
2016
31.
10.1016/j.mencom.2017.01.001_bib0155
Mamedov
Quinoxalines: Synthesis, Reactions, Mechanisms and Structure,
2016
32.
10.1016/j.mencom.2017.01.001_bib0160
Grimmett
Imidazole and Benzimidazole Synthesis,
1997
33.

Mamedov V.A., Murtazina A.M.
Russian Chemical Reviews,
2011
34.

Sexton W.A.
Journal of the Chemical Society (Resumed),
1942
35.

Hinsberg O., Koller P.
Berichte der deutschen chemischen Gesellschaft,
1896
36.
10.1016/j.mencom.2017.01.001_bib0180
Monti
Gazzetta,
1940
37.

Davoll J.
Journal of the Chemical Society (Resumed),
1960
38.

Ried W., Stahlhofen P.
Chemische Berichte,
1957
39.

Rossi A., Hunger A., Kebrle J., Hoffmann K.
Helvetica Chimica Acta,
1960
40.

Israel M., Jones L.C., Modest E.J.
Tetrahedron Letters,
1968
41.
![Nouvelle synthese de l' (∝-methyl vinyl)-9 methyl-3s-triazolo[4,3-a]benzimidazole a partir de la methyl-4benzodiazepine-1,5 one-2.](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Achour R., Essassi E.M., Zniber R.
Tetrahedron Letters,
1988
42.

Branco P.S., Prabhakar S., Lobo A.M., Williams D.J.
Tetrahedron,
1992
43.

Ruediger E.H., Gandhi S.S., Gibson M.S., Fărcaşiu D., Uncuţa C.
Canadian Journal of Chemistry,
1986
44.

Gibson M.S., Green M.
Tetrahedron,
1965
45.

Kametani T., Sota K., Shio M.
Journal of Heterocyclic Chemistry,
1970
46.

Kost A.N., Solomko Z.F., Budylin V.A., Semenova T.S.
Chemistry of Heterocyclic Compounds,
1972
47.

Howard H., Sarges R., Siegel T., Beyer T.
European Journal of Medicinal Chemistry,
1992
48.

Khodarahmi G.A., Chen C.S., Hakimelahi G.H., Tseng C.T., Chern J.W.
Journal of the Iranian Chemical Society,
2005
49.

Barchet V.R., Merz K.W.
Tetrahedron Letters,
1964
50.

Eleftheriadis N., Neochoritis C.G., Tsoleridis C.A., Stephanidou-Stephanatou J., Iakovidou-Kritsi Z.
European Journal of Medicinal Chemistry,
2013
51.

Rossi A., Hunger A., Kebrle J., Hoffmann K.
Helvetica Chimica Acta,
1960
52.

Shaw A.Y., Medda F., Hulme C.
Tetrahedron Letters,
2012
53.

Xu Z., De Moliner F., Cappelli A.P., Hulme C.
Angewandte Chemie - International Edition,
2012
54.

Xu Z., Martinez-Ariza G., Cappelli A.P., Roberts S.A., Hulme C.
Journal of Organic Chemistry,
2015
55.
56.
![�ber die Synthese von 4,9-Dihydro-9-(2-dimethylamino�thyl)-4-methyl-thieno[3,4-b][1,5]benzodiazepin-10-on](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Hromatka O., Binder D., Eichinger K.
Monatshefte fur Chemie,
1975
57.

Mamedov V.A., Murtazina A.M., Zhukova N.A., Beschastnova T.N., Rizvanov I.K., Latypov S.K.
Tetrahedron,
2014
58.

Mamedov V.A., Murtazina A.M., Adgamova D.I., Zhukova N.A., Beschastnova T.N., Kharlamov S.V., Rizvanov I.K., Latypov S.K.
Journal of Heterocyclic Chemistry,
2014
59.

Spence G.G., Taylor E.C., Buchardt O.
Chemical Reviews,
1970
60.

Haddadin M.J., Issidorides C.H.
Tetrahedron Letters,
1967
61.

Haddadin M.J., Agopian G., Issidorides C.H.
Journal of Organic Chemistry,
1971
62.

J. Haddadin M., A. Jarrar A., S. Halawi S.
Heterocycles,
1976
63.

Cordes T., Regner N., Heinz B., Borysova E., Ryseck G., Gilch P.
Journal of Photochemistry and Photobiology A: Chemistry,
2009
64.

Jarrar A.A., Fataftah Z.A.
Tetrahedron,
1977
65.

Ahmad Y., Habib M.S., Mohammady A., Bakhtiari B., Shamsi S.A.
Journal of Organic Chemistry,
1968
66.

Mamedov V.A., Saifina D.F., Gubaidullin A.T., Saifina A.F., Rizvanov I.K.
Tetrahedron Letters,
2008
67.

Mamedov V.A., Khafizova E.A., Gubaidullin A.T., Murtazina A.M., Adgamova D.I., Samigullina A.I., Litvinov I.A.
Russian Chemical Bulletin,
2011
68.

Mamedov V.A., Zhukova N.A., Syakaev V.V., Gubaidullin A.T., Beschastnova T.N., Adgamova D.I., Samigullina A.I., Latypov S.K.
Tetrahedron,
2013
69.

Kalinin A.A., Isaikina O.G., Mamedov V.A.
Chemistry of Heterocyclic Compounds,
2007
70.

Mamedov V.A., Saifina D.F., Rizvanov I.K., Gubaidullin A.T.
Tetrahedron Letters,
2008
71.
![A reaction for the synthesis of benzimidazoles and 1H-imidazo[4,5-b]pyridines via a novel rearrangement of quinoxalinones and their aza-analogues when exposed to 1,2-arylenediamines](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Mamedov V.A., Zhukova N.A., Beschastnova T.N., Gubaidullin A.T., Balandina A.A., Latypov S.K.
Tetrahedron,
2010
72.

Mamedov V.A., Kalinin A.A., Gubaidullin A.T., Gorbunova E.A., Litvinov I.A.
Russian Journal of Organic Chemistry,
2006
73.

Mamedov V.A., Zhukova N.A., Beschastnova T.N., Zakirova E.I., Kadyrova S.F., Mironova E.V., Nikonova A.G., Latypov S.K., Litvinov I.A.
Tetrahedron Letters,
2012
74.

Mamedov V.A., Zhukova N.A., Beschastnova T.N., Gubaidullin A.T., Rakov D.V., Rizvanov I.K.
Tetrahedron Letters,
2011
75.

Mamedov V.A., Saifina D.F., Gubaidullin A.T., Ganieva V.R., Kadyrova S.F., Rakov D.V., Rizvanov I.K., Sinyashin O.G.
Tetrahedron Letters,
2010
76.

Mamedov V.A., Murtazina A.M., Gubaidullin A.T., Hafizova E.A., Rizvanov I.K.
Tetrahedron Letters,
2009
77.

Mamedov V.A., Murtazina A.M., Gubaidullin A.T., Khafizova E.A., Rizvanov I.K., Litvinov I.A.
Russian Chemical Bulletin,
2010
78.

Mamedov V.A., Kadyrova S.F., Zhukova N.A., Galimullina V.R., Polyancev F.M., Latypov S.K.
Tetrahedron,
2014
79.

Hinsberg O.
Berichte der deutschen chemischen Gesellschaft,
1884
80.

Fischer E.
Berichte der deutschen chemischen Gesellschaft,
1884
81.
82.

Radziszewski B.
Berichte der deutschen chemischen Gesellschaft,
1882
83.

Japp F.R., Robinson H.H.
Berichte der deutschen chemischen Gesellschaft,
1882
84.

Tschitschibabin A.E.
Berichte der deutschen chemischen Gesellschaft (A and B Series),
1927
85.

Knorr L.
Berichte der deutschen chemischen Gesellschaft,
1884
86.
87.
10.1016/j.mencom.2017.01.001_bib0435
Hassner
Organic Syntheses Based on Name Reactions,
2012
88.
10.1016/j.mencom.2017.01.001_bib0440
Mamedov
2013
89.
![Reaction for the Synthesis of Benzimidazol-2-ones, Imidazo[5,4-b]-, and Imidazo[4,5-c]pyridin-2-ones via the Rearrangement of Quinoxalin-2-ones and Their Aza Analogues When Exposed to Enamines](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Mamedov V.A., Zhukova N.A., Zamaletdinova A.I., Beschastnova T.N., Kadyrova M.S., Rizvanov I.K., Syakaev V.V., Latypov S.K.
Journal of Organic Chemistry,
2014
90.

Allu S., Selvakumar S., Singh V.K.
Tetrahedron Letters,
2010
91.

Andrey O., Alexakis A., Bernardinelli G.
Organic Letters,
2003
92.

Harada S., Kumagai N., Kinoshita T., Matsunaga S., Shibasaki M.
Journal of the American Chemical Society,
2003
93.

Boncel S., Mączka M., Walczak K.Z.
Tetrahedron,
2010
94.

Naruse Y., Suzuki T., Inagaki S.
Tetrahedron Letters,
2005
95.

Hilt G., Treutwein J.
Angewandte Chemie - International Edition,
2007
96.

Shen R., Zhu S., Huang X.
Journal of Organic Chemistry,
2009
97.

Keck G.E., Webb R.R.
Journal of the American Chemical Society,
1981
98.

Keck G.E., Webb R.
Tetrahedron Letters,
1979
99.

Oppolzer W., Pfenninger E., Keller K.
Helvetica Chimica Acta,
1973
100.
D. F. Saifina, PhD Thesis, 2009.
101.

Mamedov V.A., Zhukova N.A., Beschastnova T.N., Syakaev V.V., Krivolapov D.B., Mironova E.V., Zamaletdinova A.I., Rizvanov I.K., Latypov S.K.
Journal of Organic Chemistry,
2015
102.
10.1016/j.mencom.2017.01.001_bib0510
Comprehensive Organic Name Reactions and Reagents,
2009
103.
10.1016/j.mencom.2017.01.001_bib0515
Smith
March's Advanced Organic Chemistry,
2001
104.
10.1016/j.mencom.2017.01.001_bib0520
The Chemistry of Enamines,
1994
105.

Hickmott P.W.
Tetrahedron,
1984
106.

Hickmott P.W.
Tetrahedron,
1982
107.

Hickmott P.W.
Tetrahedron,
1982