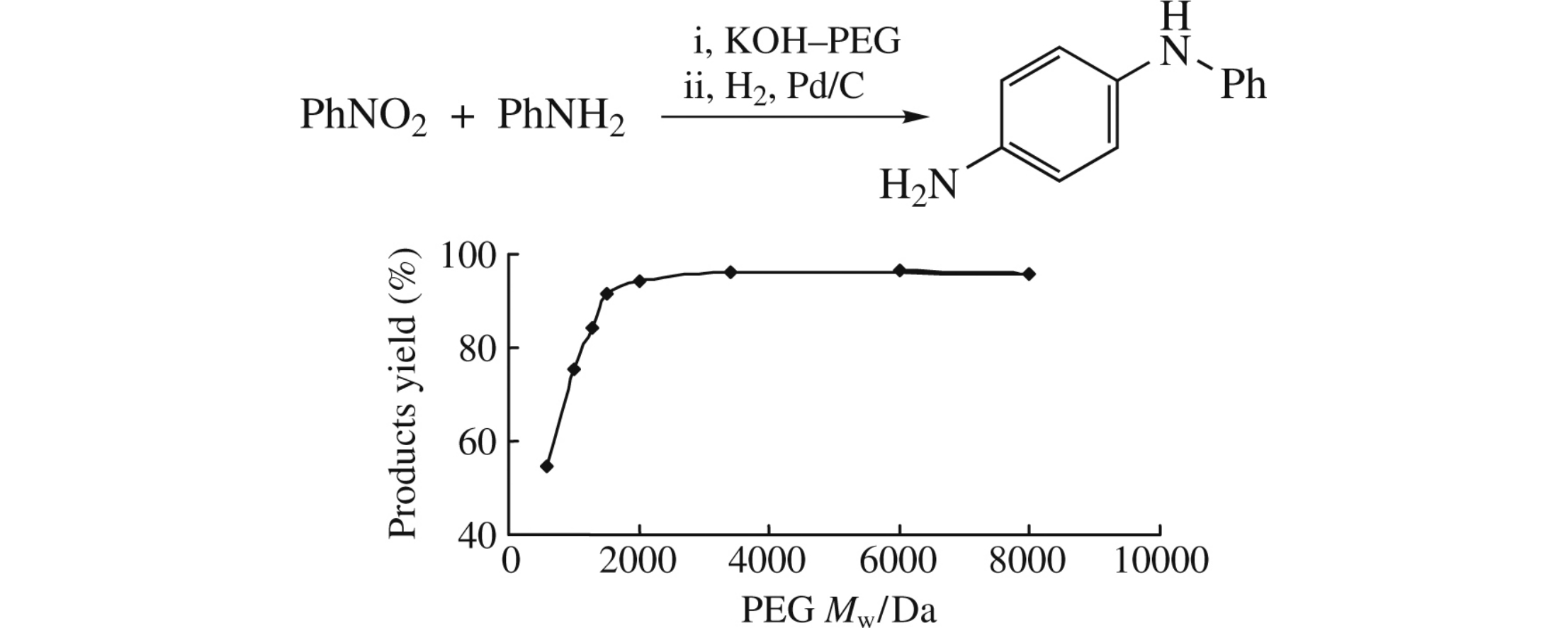
Abstract
A novel protocol for efficient coupling of nitrobenzene and aniline in poly(ethylene glycol) medium in the presence of KOH giving diphenylamine derivatives has been developed, to enable the exclusion of the toxic tetramethylammonium hydroxide catalyst commonly used in the rubber antidegradant industry.
References
1.

Talma A.G., Datta S., Huntink N.M., Datta R.N.
Rubber Chemistry and Technology,
2007
2.
10.1016/j.mencom.2016.11.033_sbref0010a
Wedemeyer
US Patent 4034042 A,
1977
3.

Rondestvedt C.S.
Journal of Organic Chemistry,
1977
4.

Buchwald S., Mauger C., Mignani G., Scholz U.
Advanced Synthesis and Catalysis,
2006
5.
J. Haider, U. Scholz and A. Sicheneder, Patent EP 1437340 A1, 2004, and references cited herein.
6.
A. Giatti, F. Korpel, R.K. Rains and G. J. Boerman, Patent US 6770189 B2, 2004, and references cited herein.
7.
R. D. Triplett and R. K. Rains, Patent US 6395933, 2002.
8.

Stern M.K., Cheng B.K., Hileman F.D., Allman J.M.
Journal of Organic Chemistry,
1994
9.

Gokel G.W., Goli D.M., Schultz R.A.
Journal of Organic Chemistry,
1983
10.

11.

Stern M.K., Hileman F.D., Bashkin J.K.
Journal of the American Chemical Society,
1992
12.

Troxler L., Wipff G.
Journal of the American Chemical Society,
1994
13.

Neumann R., Sasson Y.
Journal of Organic Chemistry,
1984
14.

Voskoboynikov A.Z., Agarkov A.Y., Chemyshev E.A., Beletskaya I.P., Churakov A.V., Kuz'mina L.G.
Journal of Organometallic Chemistry,
1997
15.

Totten G.E., Clinton N.A.
Journal of Macromolecular Science Part C- Polymer Reviews,
1998
16.

Zhang C., Gamble S., Ainsworth D., Slawin A.M., Andreev Y.G., Bruce P.G.
Nature Materials,
2009
17.

van Truong N., Norris A.R., Shin H.S., Buncel E., Bannard R.A., Purdon J.G.
Inorganica Chimica Acta,
1991
18.

Hortal A.R., Hurtado P., Martínez-Haya B., Arregui A., Bañares L.
Applied Physics A: Materials Science and Processing,
2008
19.
10.1016/j.mencom.2016.11.033_bib0085
Inoue
Cation Binding by Macrocycles,
1990
20.

Jeon S., Sawyer D.T.
Inorganic Chemistry,
1990
21.
10.1016/j.mencom.2016.11.033_sbref0095a
Blaser
Fine Chemicals through Heterogeneous Catalysis,
2001
22.

Blaser H., Steiner H., Studer M.
ChemCatChem,
2009