Abstract
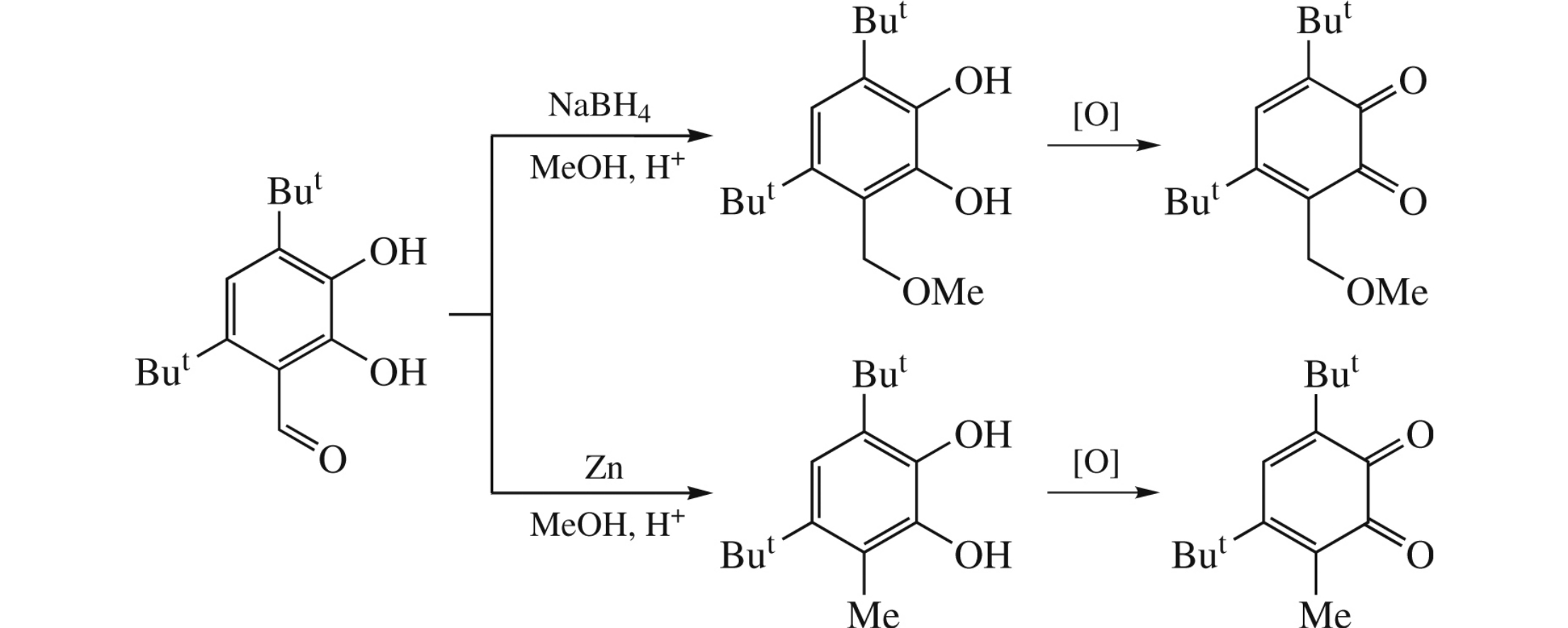
Reduction of aldehyde group in 4,6-di-tert-butyl-2,3-dihydroxybenzaldehyde leads to methoxymethyl (NaBH4, MeOH) or methyl (Zn, MeOH) analogues, which were further oxidized into the corresponding o-benzoquinones. Their photostability in benzene increases substantially on replacement of the 6-positioned Me substituent with the CH2OMe group.
References
1.

Pierpont C.G., Buchanan R.M.
Coordination Chemistry Reviews,
1981
2.

Pierpont C.
Coordination Chemistry Reviews,
2001
3.

Zanello P., Corsini M.
Coordination Chemistry Reviews,
2006
4.

Pierpont C.G., Lange C.W.
Progress in Inorganic Chemistry,
1994
5.
10.1016/j.mencom.2016.11.032_sbref0005e
Pierpont
Coord. Chem. Rev.,
2001
6.

Sato O., Tao J., Zhang Y.
Angewandte Chemie - International Edition,
2007
7.

Poddel'sky A.I., Cherkasov V.K., Abakumov G.A.
Coordination Chemistry Reviews,
2009
8.

Shushunova N.Y., Chesnokov S.A.
Polymer Science - Series B,
2009
9.

Chesnokov S.A., Cherkasov V.K., Abakumov G.A., Mamysheva O.N., Zakharina M.Y., Shushunova N.Y., Chechet Y.V., Kuropatov V.A.
Polymer Science - Series B,
2014
10.
10.1016/j.mencom.2016.11.032_sbref0010c
Abakumov
RF Patent 2138070,
1999
11.
10.1016/j.mencom.2016.11.032_sbref0010d
Abakumov
RF Patent 2472189,
2011
12.
10.1016/j.mencom.2016.11.032_sbref0010e
Chesnokov
RF Patent 2525908,
2013
13.
10.1016/j.mencom.2016.11.032_sbref0015a
Abakumov
Metalloorg. Khim.,
1991
14.

Abakumov G.A., Cherkasov V.K., Abakumova L.G., Nevodchikov V.I., Druzhkov N.O., Makarenko N.P., Kursky J.A.
Journal of Organometallic Chemistry,
1995
15.

Abakumov G.A., Nevodchikov V.I., Zaitova N.V., Druzhkov N.O., Abakumova L.G., Kurskii Y.A., Cherkasov V.K.
Russian Chemical Bulletin,
1997
16.

Abakumov G.A., Nevodchikov V.I., Zaitova N.V., Druzhkov N.O., Abakumova L.G., Kurskii Y.A., Cherkasov V.K.
Russian Chemical Bulletin,
1997
17.

Klaić L., Morimoto R.I., Silverman R.B.
ACS Chemical Biology,
2012
18.

Camelio A.M., Johnson T.C., Siegel D.
Journal of the American Chemical Society,
2015
19.

Schaller F., Rahalison L., Islam N., Potterat O., Hostettmann K., Stoeckli-Evans H., Mavi S.
Helvetica Chimica Acta,
2000
20.

Sanchez A.J., Konopelski J.P.
Journal of Organic Chemistry,
1994
21.

22.

Martin H., Magauer T., Mulzer J.
Angewandte Chemie - International Edition,
2010
23.

Sengoku T., Xu S., Ogura K., Emori Y., Kitada K., Uemura D., Arimoto H.
Angewandte Chemie - International Edition,
2014
24.

Majetich G., Zou G.
Organic Letters,
2007
25.

Diterpenes from rosemary (Rosmarinus officinalis): Defining their potential for anti-cancer activity
Petiwala S.M., Johnson J.J.
Cancer Letters,
2015
26.

Marrero J.G., Moujir L., Andrés L.S., Montaño N.P., Araujo L., Luis J.G.
Journal of Natural Products,
2009
27.
![6,6'-[piperazine-1,4-diylbis(methylene)]bis[3,5-di(tert-butyl)-1,2- benzoquinone]: Synthesis and properties](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Sayapin Y.A., Tupaeva I.O., Tkachev V.V., Shilov G.V.
Russian Journal of Organic Chemistry,
2016
28.

Arsenyev M.V., Baranov E.V., Chesnokov S.A., Cherkasov V.K., Abakumov G.A.
Russian Chemical Bulletin,
2013
29.

Singh M.S., Nagaraju A., Anand N., Chowdhury S.
RSC Advances,
2014
30.

Dorrestijn E., Kranenburg M., Ciriano M.V., Mulder P.
Journal of Organic Chemistry,
1999
31.
10.1016/j.mencom.2016.11.032_bib0065
Bukharov
Zh. Org. Khim.,
2004
32.

Gorden A.E., Xu J., Raymond K.N., Durbin P.
Chemical Reviews,
2003
33.
Zhang Q., Jin B., Peng R., Lei S., Chu S.
Mendeleev Communications,
2015
34.
Arsenyev M.V., Baranov E.V., Fedorov A.Y., Chesnokov S.A., Abakumov G.A.
Mendeleev Communications,
2015
35.
10.1016/j.mencom.2016.11.032_sbref0080a
Barltop
Excited States in Organic Chemistry,
1975
36.

Chesnokov S.A., Cherkasov V.K., Chechet Y.V., Nevodchikov N.I., Abakumov G.A., Mamysheva O.N.
Russian Chemical Bulletin,
2000
37.

Shurygina M.P., Kurskii Y.A., Druzhkov N.O., Chesnokov S.A., Abakumov G.A.
High Energy Chemistry,
2010
38.

Klement’eva S.V., Fukin G.K., Baranov E.V., Cherkasov V.K., Abakumov G.A.
High Energy Chemistry,
2011
39.

Vol’eva V.B., Belostotskaya I.S., Komissarova N.L., Starikova Z.A., Kurkovskaya L.N.
Russian Journal of Organic Chemistry,
2006
40.

Mishchenko O.G., Maslennikov S.V., Spirina I.V., Druzhkov N.O., Kurskii Y.A., Maslennikov V.P.
Russian Journal of General Chemistry,
2007
41.

Fukin G.K., Cherkasov A.V., Shurygina M.P., Druzhkov N.O., Kuropatov V.A., Chesnokov S.A., Abakumov G.A.
Structural Chemistry,
2010
42.
Data Collection. Reduction and Correction Program. CrysAlisPro Software Package, Agilent Technologies, Oxford, UK, 2012.
43.
10.1016/j.mencom.2016.11.032_bib0100
Sheldrick
SADABS-2014/2. Bruker/Siemens Area Detector Absorption Correction Program,
2012
44.
SCALE3 ABSPACK: Empirical Absorption Correction, CrysAlisPro Software Package, Agilent Technologies, Oxford, UK, 2012.
45.
10.1016/j.mencom.2016.11.032_bib0110
Sheldrick
2015
46.
G. M. Sheldrick, SHELXTL v. 6.14, Structure Determination Software Suite, Bruker AXS, Madison, USA, 2003.