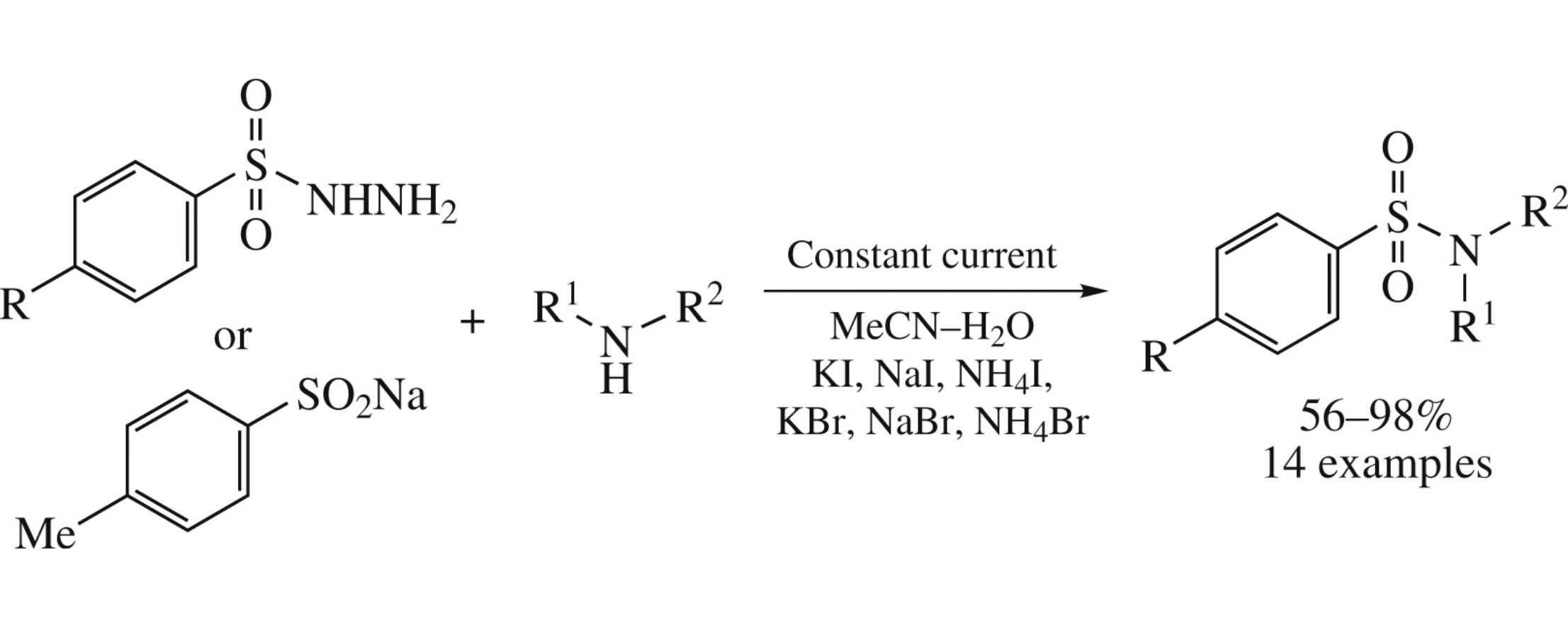
Abstract
An efficient electrochemical synthesis of sulfonamides (yields 56–98%) from arenesulfonohydrazides or sodium p-methylbenzenesulfinate and amines was performed in an undivided cell with graphite anode and iron cathode in MeCN–H2O medium using halides as redox mediators and supporting electrolytes.
References
1.

Wilden J.D.
Journal of Chemical Research,
2010
2.

Scozzafava A., Owa T., Mastrolorenzo A., Supuran C.
Current Medicinal Chemistry,
2003
3.

Supuran C.T., Casini A., Scozzafava A.
Medicinal Research Reviews,
2003
4.

Roell D., Baniahmad A.
Molecular and Cellular Endocrinology,
2011
5.

Ma M., Cheng Y., Xu Z., Xu P., Qu H., Fang Y., Xu T., Wen L.
European Journal of Medicinal Chemistry,
2007
6.

Chohan Z.H., Youssoufi M.H., Jarrahpour A., Ben Hadda T.
European Journal of Medicinal Chemistry,
2010
7.

Reddy N.S., Mallireddigari M.R., Cosenza S., Gumireddy K., Bell S.C., Reddy E.P., Reddy M.V.
Bioorganic and Medicinal Chemistry Letters,
2004
8.

Natarajan A., Guo Y., Harbinski F., Fan Y., Chen H., Luus L., Diercks J., Aktas H., Chorev M., Halperin J.A.
Journal of Medicinal Chemistry,
2004
9.

Supuran C., Innocenti A., Mastrolorenzo A., Scozzafava A.
Mini-Reviews in Medicinal Chemistry,
2004
10.

Siddiqui N., Pandeya S.N., Khan S.A., Stables J., Rana A., Alam M., Arshad M.F., Bhat M.A.
Bioorganic and Medicinal Chemistry Letters,
2007
11.

12.

Stranix B.R., Lavallée J., Sévigny G., Yelle J., Perron V., LeBerre N., Herbart D., Wu J.J.
Bioorganic and Medicinal Chemistry Letters,
2006
13.

Lakrout S., K'tir H., Amira A., Berredjem M., Aouf N.
RSC Advances,
2014
14.

Gioiello A., Rosatelli E., Teofrasti M., Filipponi P., Pellicciari R.
ACS Combinatorial Science,
2013
15.

Sridhar R., Srinivas B., Kumar V.P., Narender M., Rao K.R.
Advanced Synthesis and Catalysis,
2007
16.

Shi F., Tse M.K., Zhou S., Pohl M., Radnik J., Hübner S., Jähnisch K., Brückner A., Beller M.
Journal of the American Chemical Society,
2009
17.

Zhu M., Fujita K., Yamaguchi R.
Organic Letters,
2010
18.

Cui X., Shi F., Zhang Y., Deng Y.
Tetrahedron Letters,
2010
19.

Yin J., Buchwald S.L.
Organic Letters,
2000
20.

Yin J., Buchwald S.L.
Journal of the American Chemical Society,
2002
21.

He H., Wu Y.
Tetrahedron Letters,
2003
22.

Lam P.Y., Vincent G., Clark C.G., Deudon S., Jadhav P.K.
Tetrahedron Letters,
2001
23.
![N-Arylation of imidazoles, imides, amines, amides and sulfonamides with boronic acids using a recyclable Cu(OAc)2·H2O/[bmim][BF4] system](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Kantam M.L., Neelima B., Reddy C.V., Neeraja V.
Journal of Molecular Catalysis A Chemical,
2006
24.

Kalita B., Lamar A.A., Nicholas K.M.
Chemical Communications,
2008
25.

Powell D.A., Fan H.
Journal of Organic Chemistry,
2010
26.

Moon S., Nam J., Rathwell K., Kim W.
Organic Letters,
2014
27.

Song Z., Antonchick A.P.
Organic and Biomolecular Chemistry,
2016
28.

Tang X., Huang L., Qi C., Wu X., Wu W., Jiang H.
Chemical Communications,
2013
29.

Zhao J., Xu J., Chen J., Wang X., He M.
RSC Advances,
2014
30.

Yotphan S., Sumunnee L., Beukeaw D., Buathongjan C., Reutrakul V.
Organic and Biomolecular Chemistry,
2016
31.
Kokorekin V.A., Yanilkin V.V., Morozov V.I., Tomilov Y.V., Platonov D.N., Petrosyan V.A.
Mendeleev Communications,
2014
32.
Lyalin B.V., Sigacheva V.L., Kokorekin V.A., Petrosyan V.A.
Mendeleev Communications,
2015
33.
10.1016/j.mencom.2016.11.027_bib0165
Ogibin
Russ. Chem. Rev.,
2009